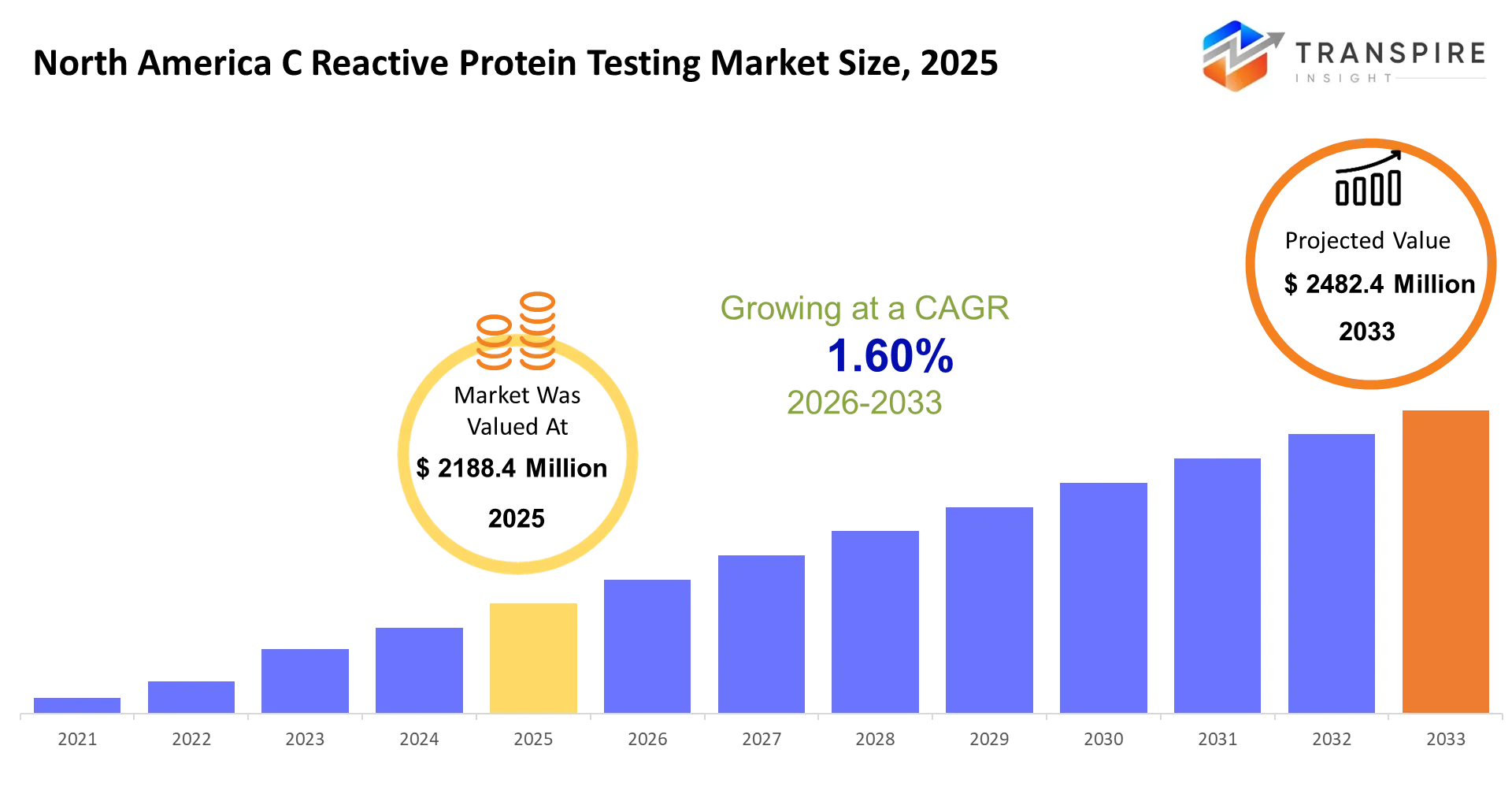
North America C Reactive Protein Testing Market Size & Forecast:
- North America C Reactive Protein Testing Market Size 2025: USD 2188.4 Million
- North America C Reactive Protein Testing Market Size 2033: USD 2482.4 Million
- North America C Reactive Protein Testing Market CAGR: 1.60%
- North America C Reactive Protein Testing Market Segments: By Type (CRP Tests, hs-CRP Tests, Rapid Tests, Lab-based Tests, Others), By Application (Cardiovascular Diseases, Infections, Inflammatory Disorders, Autoimmune Diseases, Others), By End-User (Hospitals, Diagnostic Labs, Clinics, Homecare, Others), By Technology (Immunoassay, ELISA, Turbidimetric, Rapid Kits, Others).
To learn more about this report, Download Free Sample Report
North America C Reactive Protein Testing Market Summary:
The North America C Reactive Protein Testing Market size is estimated at USD 2188.4 Million in 2025 and is anticipated to reach USD 2482.4 Million by 2033, growing at a CAGR of 1.60% from 2026 to 2033. The testing market for C-reactive protein (CRP) in North America provides essential support to doctors because it enables them to assess inflammation from infections and cardiovascular risks and chronic diseases within a short time frame. The system provides emergency departments with faster patient evaluation and outpatient facilities with enhanced patient tracking and preventive medicine programs with better methods to assess patient risk.
The evolution of testing procedures during the last three to five years has resulted in patient testing through point-of-care systems and integrated diagnostic technologies. The COVID-19 pandemic created a turning point for this shift because it made rapid biomarker testing common and led to increased funding for decentralized diagnostic systems.
Healthcare providers now prefer CRP tests that deliver quick actionable results which doctors can use for direct patient care. The testing approach decreases the time needed for diagnosis while enabling doctors to begin treatment sooner which leads to more tests being conducted at hospitals and clinics and home healthcare services and this process results in continuous market growth.
Key Market Insights
- Healthcare facilities in North America create a testing market for C Reactive Protein that experiences continuous growth because of their established medical systems which enable hospitals to conduct tests for both emergency situations and routine health assessments.
- The United States maintains its market control in the region and will achieve an 80% market share by 2025 because of its advanced diagnostic technologies and associated reimbursement systems.
- Canada will remain the most rapidly expanding area until 2030 because public health screening programs continue to grow and demand for biomarker-based diagnostics increases.
- The CRP testing market will reach its highest point through laboratory testing which currently holds 55% of the total market share because this method provides dependable results in testing environments used for clinical research.
- The point-of-care CRP testing market will experience its fastest growth rate because it needs to provide quicker results to doctors who require immediate information for their clinical choices.
- CRP testing shows its highest application rate in cardiovascular disease screening which accounts for 40% of total uses because the test helps identify risks for patients and their doctors to develop preventive healthcare plans.
- The market for infectious disease monitoring has emerged as an important segment because it enables doctors to identify bacterial and viral infections through their daily clinical activities.
- Hospitals maintain their position as the primary end-user market for medical services which accounts for more than 60% of total demand because their emergency diagnostic facilities and inpatient treatment procedures require these medical services.
- The growth of clinics and home healthcare facilities as end-user sections demonstrates how the healthcare system moves toward providing decentralized medical services that focus on patient needs.
- Companies introduce product innovation through the development of high-sensitivity CRP assays and portable analyzers which will help them meet growing demand while enhancing their diagnostic solutions.
- Strategic partnerships together with collaborations to hospitals and diagnostic networks enable businesses to enter new markets while they expand their operations in specific areas.
What are the Key Drivers, Restraints, and Opportunities in the North America C Reactive Protein Testing Market?
The clinical transition toward quick evidence-based evaluations in acute and primary care settings has become the main driving force behind the North America C Reactive Protein Testing Market expansion. The COVID-19 pandemic established actual patient biomarker testing as a standard practice which led healthcare facilities to develop more efficient testing operations. High-sensitivity CRP and point-of-care platforms now serve as essential tools for infection diagnosis and cardiovascular risk evaluation. The system directly boosts test usage during patient visits while it develops additional income sources for diagnostic companies.
Healthcare organizations face a fundamental obstacle because reimbursement methods and costs differ between their systems. The United States CRP testing market faces challenges because clinical testing value exists yet payer policies fail to provide reliable coverage for point-of-care testing. The issue remains unresolved because it requires updates from regulatory organizations together with policies from insurers and clinical guideline development to reach a solution. The process becomes slower at small medical practices and outpatient clinics which prevents them from reaching test market expansion and revenue generation through decentralized testing operations.
The integration of CRP testing into remote patient monitoring and digital health systems creates a new business opportunity. The combination of portable CRP devices with telehealth platforms enables doctors to monitor inflammation levels in patients who have chronic diseases from their homes. The integration of home-based diagnostic systems with connected care infrastructure creates new testing applications which enable higher testing rates and generate additional revenue streams for North American healthcare providers.
What Has the Impact of Artificial Intelligence Been on the North America C Reactive Protein Testing Market?
The North America C Reactive Protein Testing Market experiences operational changes because artificial intelligence and advanced digital technologies enhance laboratory work efficiency and assist doctors in making clinical choices. The laboratory information systems and immunoassay analyzers now use AI-driven automation to operate their systems which include algorithms that execute sample prioritization and identify abnormal CRP values and generate reports. The system enables laboratories to work at maximum capacity because it decreases the need for human work and provides faster results while maintaining accurate testing throughout the entire process.
The machine learning models establish predictive functions through their ability to link CRP levels with patient background information together with their existing medical conditions and various biological indicators. The models identify early signs of flare-ups which indicate inflammation or increased risk of sepsis or cardiovascular problems, thus enabling doctors to start their treatment faster. The hospital networks achieved measurable progress through these tools which helped them decrease time spent on diagnosing patients while improving their ability to categorize patients, which resulted in both reduced expenses and better treatment results.
The operational operations of digital integration provide advantages through its ability to enhance data exchanges between electronic health records, which creates better patient care and test result management capabilities. The clinical data collection process faces a major problem because it involves diverse information from multiple sources which lack standardized organization. The healthcare system faces two challenges that create data quality problems which hinder model development because they restrict model accuracy and they obstruct the implementation of AI technologies in standard CRP testing procedures.
Key Market Trends
- Since 2020, hospitals have shifted from centralized lab testing toward point-of-care CRP devices which enable testing results to be delivered within 20 minutes.
- The period from 2021 to 2025 saw outpatient clinics increasingly implement portable analyzers which demonstrated the growing trend of decentralized diagnostic methods that prioritize patient needs.
- The introduction of high-sensitivity CRP assays emerged as a response to updated cardiovascular guidelines which established preventive screening requirements that extended their application beyond acute infection testing into permanent risk assessment.
- The period from 2022 to 2024 experienced stricter reimbursement rules in the United States which caused smaller healthcare providers to postpone their implementation of modern CRP testing systems that offered clinical benefits.
- Diagnostic companies like Abbott Laboratories and Roche Diagnostics redirected their funding toward compact analyzers which demonstrate their commitment to developing swift testing systems that operate at patient locations.
- The period following 2021 experienced fast progress in electronic health record systems which enabled healthcare facilities to automatically interpret CRP results thus enhancing their ability to make clinical decisions throughout their multiple sites.
- Manufacturers learned from supply chain disruptions that occurred between 2020 and 2022 which showed they depended on imported reagents so they started to create backup plans and produce materials at local sites throughout North America.
- The year 2025 marked the beginning of telehealth platforms which added CRP testing to their remote monitoring systems for chronic disease management through persistent inflammation assessment.
- The competitive landscape transformed as businesses sought alliances with hospital networks and diagnostic chains which shifted their focus from product development to creating service-based diagnostic systems.
North America C Reactive Protein Testing Market Segmentation
By Type
The type segmentation shows a clear hierarchy which starts with lab-based CRP tests because hospitals and high-throughput diagnostic labs commonly use these tests. The clinical workflow needs standardized tests which provide high accuracy to conduct critical care tests and monitor chronic diseases, so these testing formats continue to dominate the field. Emergency departments and outpatient facilities have adopted rapid tests and point-of-care formats since 2020 because these testing methods enable quicker diagnosis.
Cardiovascular screening programs have started using high-sensitivity CRP tests which have expanded their testing capabilities through the addition of preventive diagnostic tests. The forecast period will bring incremental market share growth to rapid and hs-CRP formats which will lead manufacturers to develop portable analyzers and multiplex testing systems.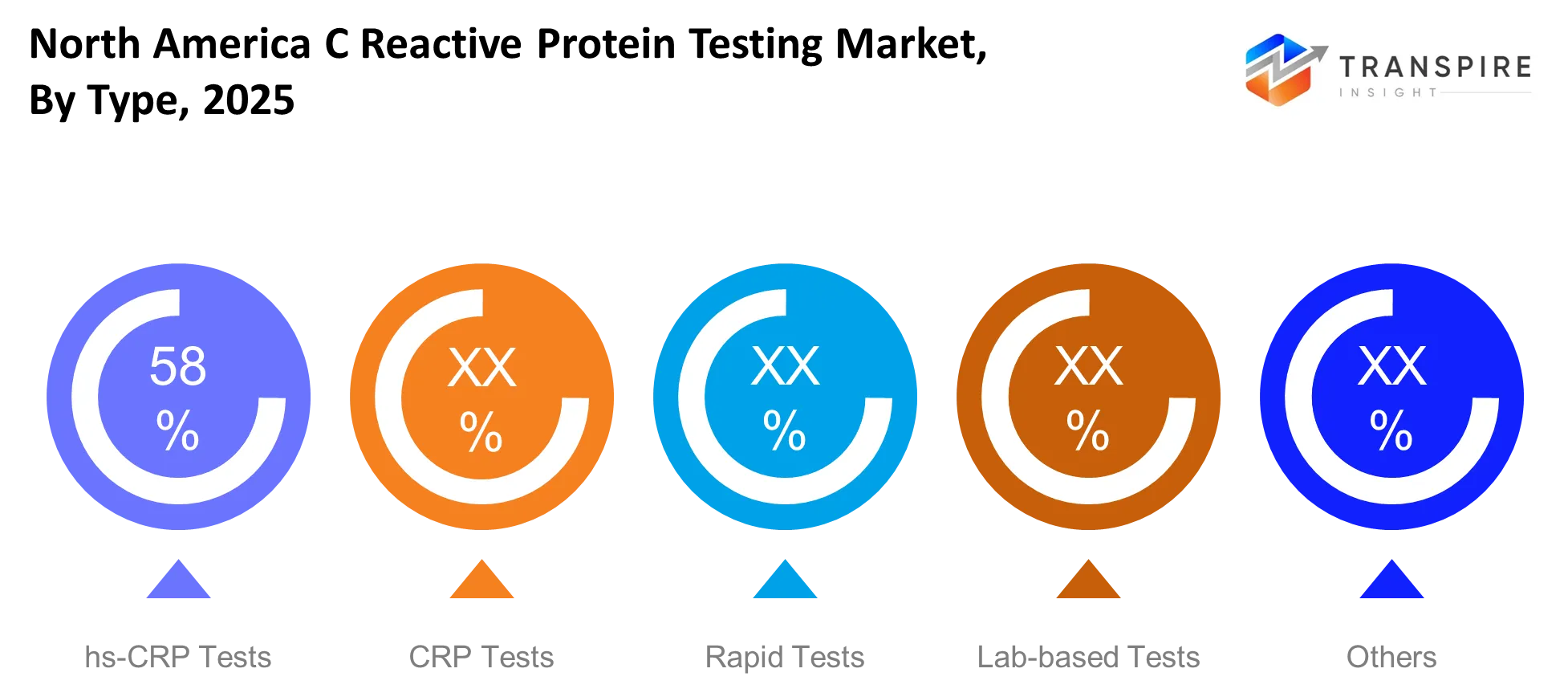
By Application
To learn more about this report, Download Free Sample Report
The current applications of application segmentation remain focused on cardiovascular diseases because they make up the largest portion of cases which use CRP markers for risk assessment and preventive treatment procedures. The American clinical guidelines which recommend early identification of cardiac risk factors have created an environment which supports this clinical practice. Infection monitoring has emerged as a strong secondary segment, especially after the pandemic highlighted the importance of distinguishing bacterial from viral conditions.
The use of inflammatory and autoimmune disorder applications continues to expand because more people get diagnosed with these disorders and need ongoing treatment for their permanent conditions. The future expansion of the business will achieve success through integrated diagnostic pathways which use CRP testing to diagnose multiple health conditions at increased testing rates and greater testing value.
By End-User
Hospitals control most of end-user segmentation because they provide essential services for acute care and emergency diagnostics and inpatient monitoring. Diagnostic laboratories follow closely, supported by centralized testing models and economies of scale in processing large sample volumes.
The expansion of clinics and homecare facilities has occurred throughout recent years because the healthcare system moved away from centralized operations toward decentralized outpatient treatment. The increasing use of portable testing devices together with telehealth-supported diagnostics drives this transformation. The growth of clinics and home-based testing will continue to develop, which will lead suppliers to create user-friendly connected devices that people can use outside of hospitals.
By Technology
The main technology segmentation of the market is driven by immunoassay-based systems which maintain their leading position because of their precise results and ability to scale operations and their seamless integration with automated laboratory systems. ELISA methods continue to play a strong role in research and specialized diagnostics, although they face competition from faster alternatives.
Clinical laboratories now use turbidimetric methods because these methods can be integrated with existing chemistry analyzers and they provide cost advantages. Healthcare providers have chosen rapid test kits as their primary option because these kits provide quick results and easy access to diagnostic tests. upcoming technology development efforts will concentrate on achieving better sensitivity results and shorter testing periods and digital data system integration, which will shape purchasing choices made by healthcare providers and diagnostic networks.
What are the Key Use Cases Driving the North America C Reactive Protein Testing Market?
The North America C Reactive Protein Testing Market exists mainly to assess cardiovascular risk through testing which has become its primary application. High-sensitivity CRP testing enables doctors to detect minor heart disease-related inflammation which they need for their preventive healthcare programs. The use case creates peak demand because it enables health facilities to conduct routine tests on entire patient groups while providing treatment for their ongoing needs.
The field of infection management has experienced rapid growth because medical professionals at hospitals and outpatient clinics now use CRP testing to differentiate between bacterial and viral infections. This method improves antibiotic stewardship because it decreases the number of prescriptions that doctors write without justification. The medical field sees continuous need for chronic inflammatory diseases because specialty clinics treat patients who require extended medical care.
Home-based monitoring systems that connect to telehealth services through CRP testing for chronic disease management represent an emerging use case. The field of diagnostics develops new possibilities through research which combines CRP testing with multiple biomarker panels to better identify complex medical conditions at their initial stages.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 2188.4 Million |
|
Market size value in 2026 |
USD 2221.3 Million |
|
Revenue forecast in 2033 |
USD 2482.4 Million |
|
Growth rate |
CAGR of 1.60% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
North America (Canada, The United States, and Mexico) |
|
Key company profiled |
Abbott, Roche, Siemens Healthineers, Beckman Coulter, Thermo Fisher, Danaher, Bio-Rad, Ortho Clinical Diagnostics, Randox, Sysmex, Mindray, Horiba, Agilent, EKF Diagnostics, Nova Biomedical |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (CRP Tests, hs-CRP Tests, Rapid Tests, Lab-based Tests, Others), By Application (Cardiovascular Diseases, Infections, Inflammatory Disorders, Autoimmune Diseases, Others), By End-User (Hospitals, Diagnostic Labs, Clinics, Homecare, Others), By Technology (Immunoassay, ELISA, Turbidimetric, Rapid Kits, Others) |
Which Regions are Driving the North America C Reactive Protein Testing Market Growth?
North America dominates the market because the United States holds the biggest market share through its effective reimbursement systems and its developed medical facilities. The federal government supports preventive healthcare initiatives and biomarker-based testing which has established routine CRP testing procedures throughout medical facilities and outpatient centers.
The combination of large hospital systems and integrated lab networks together with the common use of electronic health records systems creates an environment that enables medical facilities to maintain high rates of testing throughout their operations. The region maintains its leadership position through ongoing financial support of diagnostic research and the swift implementation of portable diagnostic systems.
Canada exists as a consistent second market which derives its stability from a government-funded healthcare system that guarantees equal access to testing for all patients. The United States operates with more private funding while this country depends on national screening initiatives and provincial healthcare regulations for its expansion. The requirement creates dependable CRP testing needs which medical facilities use for both chronic disease treatment and primary care operations. Canada maintains steady funding and integrated healthcare services which make it a dependable source of regional income that avoids the market fluctuations experienced by systems based on free-market principles.
Mexico demonstrates its highest growth rate because of recent improvements in its healthcare system and better access to diagnostic testing facilities. Government efforts to enhance early disease detection processes while expanding laboratory capabilities have resulted in faster CRP testing adoption across urban and semi-urban regions. The private health sector is now purchasing new diagnostic technologies which establishes additional revenue streams. The market between 2026 and 2033 will experience significant growth which creates business opportunities for new companies and investors who want to enter the testing market that offers affordable and mobile testing options.
Who are the Key Players in the North America C Reactive Protein Testing Market and How Do They Compete?
The North America C Reactive Protein Testing Market shows moderate competition because multinational diagnostics companies hold most of the market share while smaller businesses focus on niche markets and point-of-care testing. Companies use technology updates and complete platform solutions to maintain market share instead of competing through lower prices.
The main competitive factor evaluates assay performance through three metrics which include measurement accuracy and testing speed and testing results integration into digital clinical systems. New companies are developing testing solutions which use portable and quick testing methods to create market challenges that established companies must overcome. Abbott Laboratories develops small point-of-care testing devices which provide quick CRP test results for use in emergency departments and outpatient facilities.
Abbott uses this method to create a unique product which provides fast and easy-to-use results for testing in decentralized healthcare settings. Roche Diagnostics develops high-sensitivity tests which work with automated laboratory systems, enabling the company to meet the needs of large hospital networks that require testing at high volume with exact accuracy. Roche continues to expand its business through partnerships with hospital systems and funding for its digital laboratory development projects.
Siemens Healthineers uses its advanced automation system and complete data integration capabilities to create hospital systems that work through interconnected medical facility networks. Thermo Fisher Scientific establishes its market position through its extensive diagnostic product range and its ability to serve research-based applications which help it enter multiple market segments. Danaher Corporation uses its diagnostic companies to drive product development through ongoing research and purchasing new businesses, which helps them improve testing methods and grow their presence in medical and laboratory environments.
Company List
- Abbott
- Roche
- Siemens Healthineers
- Beckman Coulter
- Thermo Fisher
- Danaher
- Bio-Rad
- Ortho Clinical Diagnostics
- Randox
- Sysmex
- Mindray
- Horiba
- Agilent
- EKF Diagnostics
- Nova Biomedical
Recent Development News
In April 2026, Quest Diagnostics Raises 2026 Financial Outlook on Strong Diagnostic Testing Demand: Quest Diagnostics reported stronger-than-expected Q1 performance in the U.S., driven by sustained demand for routine diagnostic and inflammatory testing services, including CRP-related panels. The company also upgraded its full-year 2026 revenue and profit guidance, reflecting stable outpatient testing volumes across the U.S. healthcare system.
Source: https://www.reuters.com
In January 2026, Abbott Expands Diagnostic Portfolio with Enhanced Inflammation Testing Focus: Abbott Laboratories strengthened its diagnostics segment in the U.S. by advancing its inflammation and cardiac biomarker testing portfolio, including CRP-related assay enhancements for high-throughput hospital laboratories. The initiative is aimed at improving early detection of systemic inflammation in chronic disease patients.
Source: https://www.openpr.com
What Strategic Insights Define the Future of the North America C Reactive Protein Testing Market?
The North America C Reactive Protein Testing Market has begun to adopt a diagnostic model which combines testing at patient locations with digital medical pathways. The system moves in this direction because payers need to decrease hospital operations while clinicians require immediate medical information which they can use for patient treatment. The next five to seven years will see growth become dependent on CRP testing's success in connecting with multiple testing systems and remote patient monitoring technologies.
The development of multiplex panels and AI-enabled diagnostic systems has created a hidden danger which threatens to eliminate the need for traditional single-biomarker tests that use CRP. The use of broader testing panels by doctors will reduce the profit margins for traditional CRP tests.
A new business opportunity exists through CRP testing which can now function within home diagnostic systems that operate with telehealth reimbursement frameworks. Businesses need to establish their core business around devices which can connect with digital health systems because this will help them maintain important market presence and generate income through stable customer relationships.
North America C Reactive Protein Testing Market Report Segmentation
By Type
- CRP Tests
- hs-CRP Tests
- Rapid Tests
- Lab-based Tests
- Others
By Application
- Cardiovascular Diseases
- Infections
- Inflammatory Disorders
- Autoimmune Diseases
- Others
By End-User
- Hospitals
- Diagnostic Labs
- Clinics
- Homecare
- Others
By Technology
- Immunoassay
- ELISA
- Turbidimetric
- Rapid Kits
- Others
Frequently Asked Questions
Find quick answers to common questions.
The Approximate North America C Reactive Protein Testing Market size for the Market will be USD 2482.4 Million in 2033.
The key segments of the North America C Reactive Protein Testing Market are By Type (CRP Tests, hs-CRP Tests, Rapid Tests, Lab-based Tests, Others), By Application (Cardiovascular Diseases, Infections, Inflammatory Disorders, Autoimmune Diseases, Others), By End-User (Hospitals, Diagnostic Labs, Clinics, Homecare, Others), By Technology (Immunoassay, ELISA, Turbidimetric, Rapid Kits, Others).
Major Players in the North America C Reactive Protein Testing Market are Abbott, Roche, Siemens Healthineers, Beckman Coulter, Thermo Fisher, Danaher, Bio-Rad, Ortho Clinical Diagnostics, Randox, Sysmex, Mindray, Horiba, Agilent, EKF Diagnostics, Nova Biomedical.
The Current Market size of the North America C Reactive Protein Testing Market is USD 2188.4 Million in 2025.
The North America C Reactive Protein Testing Market CAGR is 1.60%.
- Abbott
- Roche
- Siemens Healthineers
- Beckman Coulter
- Thermo Fisher
- Danaher
- Bio-Rad
- Ortho Clinical Diagnostics
- Randox
- Sysmex
- Mindray
- Horiba
- Agilent
- EKF Diagnostics
- Nova Biomedical
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


