North America Enteric Disease Testing Market Size & Forecast:
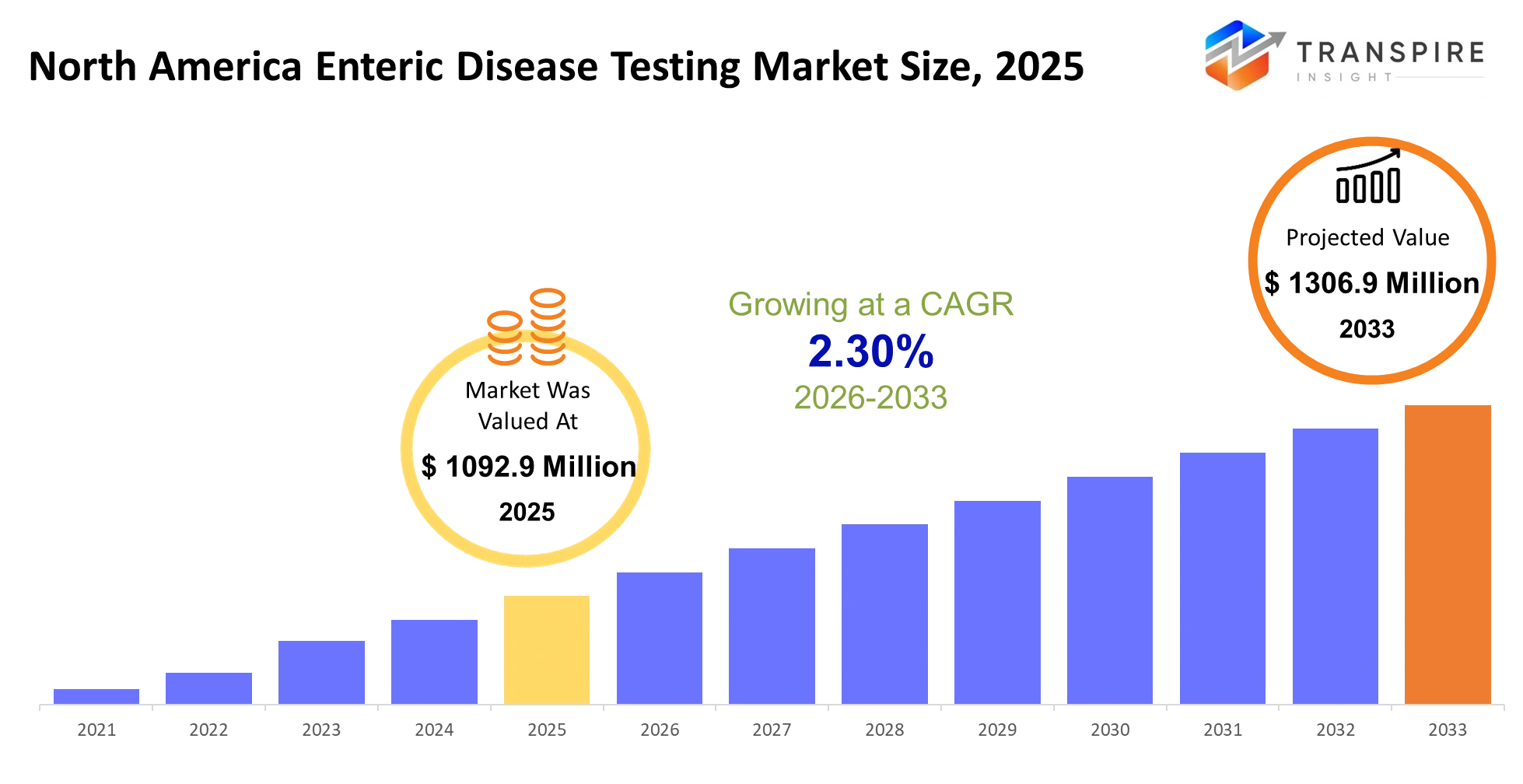
- North America Enteric Disease Testing Market Size 2025: USD 1092.9 Million
- North America Enteric Disease Testing Market Size 2033: USD 1306.9 Million
- North America Enteric Disease Testing Market CAGR: 2.30%
- North America Enteric Disease Testing Market Segments: By Type (Bacterial Testing, Viral Testing, Parasitic Testing, Molecular Diagnostics, Rapid Tests, Others), By Application (Gastroenteritis, Cholera, Salmonella, E. coli, Foodborne Diseases, Others), By End-User (Hospitals, Labs, Clinics, Research Institutes, Others), By Technology (PCR, ELISA, Culture Methods, Sequencing, Rapid Kits, Others).
To learn more about this report, Download Free Sample Report
North America Enteric Disease Testing Market Summary:
The North America Enteric Disease Testing Market size is estimated at USD 1092.9 Million in 2025 and is anticipated to reach USD 1306.9 Million by 2033, growing at a CAGR of 2.30% from 2026 to 2033. The North America Enteric Disease Testing Market is experiencing robust expansion because more people develop gastrointestinal infections and more people learn about the value of detecting diseases in their initial stages.
The demand for advanced diagnostic solutions is increasing because of three main factors which include the consumption of contaminated food and water resources together with changes in food consumption patterns and the regular occurrence of bacterial and viral infection outbreaks. The healthcare industry is moving towards using rapid testing technologies which provide accurate results because these solutions help medical professionals deliver timely patient care and drive market growth.
Molecular diagnostics and point-of-care testing technology improvements are driving fundamental changes in the North America Enteric Disease Testing Market because they enable faster testing results and more accurate results and simpler testing processes. The market experiences growth because governments are building stronger public health surveillance systems together with their increasing interest in preventive healthcare solutions. The market will continue to grow during the next years because laboratories and healthcare facilities keep improving their diagnostic systems.
What Has the Impact of Artificial Intelligence Been on the North America Enteric Disease Testing Market?
Artificial intelligence technologies create fast transformations in the North America enteric disease testing market because they improve research decision-making and speed up the development of new diagnostic tests. The North American enteric disease testing market applies artificial intelligence for advanced market research because the technology analyzes real-time data to uncover concealed patterns that demonstrate infection distribution and patient demographic information.
Predictive analytics rely on machine learning algorithms which enable healthcare providers and stakeholders to accurately predict disease outbreaks and testing requirements. The current transformation process creates more efficient strategies which help organizations deal with changing public health threats.
The north america enteric disease testing market shows operational efficiency improvements through artificial intelligence which implements automated systems and streamlines work processes. The AI-powered systems in laboratories automate all tasks which results in fewer human mistakes while increasing the productivity of diagnostic testing kit production.
AI-based supply chain optimization technology uses inventory predictions and logistics enhancements to eliminate delays while decreasing expenses. The technological advancements help core businesses maintain their competitive edge while enabling them to develop personalized diagnostic solutions which drive continuous growth in the north american enteric disease testing market.
Key Market Trends & Insights:
- The United States leads the North American market for enteric disease testing because it operates advanced healthcare systems which enable it to maintain an 80% market share in 2024.
- Canada represents the fastest-growing region because diagnostic investments will drive its market expansion which will exceed 7% annual growth until 2030.
- The molecular diagnostics field will achieve dominance in 2024 with a 45% market share because its tests deliver accurate results while detecting pathogens at high speeds.
- Immunoassays hold the second-largest share because they combine cost-effective testing methods with their use in various clinical applications by healthcare professionals.
- The multiplex PCR testing field will experience rapid growth because it enables efficient detection of multiple pathogens which will occur between 2025 and 2030.
- Bacterial infection testing leads the market because it provides over 50% of testing services which occur during foodborne illness outbreaks and contamination incidents.
- Viral infection testing has become the most rapidly progressing field because people increasingly recognize its value for identifying infections at early stages.
- Hospitals and clinical laboratories control approximately 60% of the market because their facilities receive many patients who require advanced diagnostic tests.
- Point-of-care settings represent the fastest-growing market segment because testing demand requires quick testing solutions which can be performed outside traditional laboratory settings.
North America Enteric Disease Testing Market Segmentation
By Type
Bacterial testing identifies pathogens that cause foodborne infections and digestive illness through its testing of Salmonella and E. coli bacteria. People use viral testing to identify norovirus and rotavirus infections that spread rapidly through community and healthcare environments.
Parasitic testing helps scientists find all microorganisms that cause extended stomach problems in regions where people lack proper sanitation. The different testing methods provide support for quick identification of medical conditions which enables doctors to choose appropriate treatment methods. Molecular diagnostics has gained strong attention because its tests can detect multiple pathogens with high precision through rapid testing.
Rapid tests provide emergency and point-of-care facilities with their ability to deliver quick screening results which medical staff can use for immediate patient evaluation. The other methods of testing include traditional laboratory techniques which medical professionals use to confirm results and perform in-depth testing. The combination of these testing methods will enhance diagnostic performance while improving processes for controlling disease outbreaks.

To learn more about this report, Download Free Sample Report
By Application
Gastroenteritis remains a major application area, driven by frequent cases linked to contaminated food and water sources. Cholera testing plays a critical role in outbreak control especially for regions that experience both inadequate sanitation and rapid disease transmission. People use Salmonella and E. coli testing because these two pathogens cause most foodborne illnesses and public health risks continue to rise. Each application area demonstrates that timely detection provides essential benefits which help to decrease medical problems while improving results for patients.
Foodborne diseases account for a significant share due to rising consumption of processed and packaged food products. Other applications include less common infections that still require accurate diagnosis for proper treatment. The need for dependable testing methods will increase because people become more aware and healthcare organizations prioritize early disease identification. Advanced diagnostic tools will experience consistent market demand from these application areas throughout various operational environments.
By End-User
Hospitals serve as essential end-user segments because they treat numerous patients while operating sophisticated diagnostic systems. Labs operate as essential facilities which handle extensive sample processing through their precise testing capabilities. Clinics offer testing services which enable patients to receive early diagnosis and conduct their regular medical examinations. All end-user entities work together to enhance disease detection efforts and disease management capabilities.
Research institutes focus on developing new testing methods and improving existing technologies through continuous study and innovation. Public health organizations serve as additional end-users which provide support for surveillance activities and outbreak monitoring efforts. All user groups will increase their adoption of testing services because they require both rapid results and precise outcomes. The development of healthcare infrastructure will create pathways for testing capacity expansion.
By Technology
PCR technology serves as a common diagnostic tool because it detects pathogens with extreme precision and rapid results. The ELISA test provides a dependable method for detecting specific antigens and antibodies which hospitals use for their patient testing needs. Culture methods remain essential for confirming infection cases while researchers use them to study bacterial growth patterns. The various technologies each contribute to delivering correct medical assessments together with appropriate treatment strategies.
The use of sequencing techniques is increasing because they provide researchers with better methods to analyze and identify difficult pathogens. Rapid kits deliver instant test results which medical professionals require during emergency situations when they need to make fast decisions. The other technologies enable researchers to conduct specific tests and scientific studies. The ongoing technology improvements will lead to better testing facilities which will detect diseases with greater speed and precise outcomes and better operational effectiveness.
What are the Main Challenges for the North America Enteric Disease Testing Market Growth?
The north america enteric disease testing market encounters multiple technical and operational obstacles which hinder its ability to deliver reliable results and expand its operations. Testing effectiveness decreases because sample quality varies and certain diagnostic techniques have sensitivity constraints and laboratories require intricate testing procedures. Operational expenses increase because supply chain interruptions for diagnostic components and reagents create delivery delays.
The north america enteric disease testing market requires testing methods which achieve standardization and provide dependable results. The north america enteric disease testing market experiences growth restrictions because of manufacturing and commercialization obstacles. The process of bringing new products to market requires additional time and expenses because companies must follow strict regulatory requirements which include quality standards such as GMP.
High production costs for advanced molecular diagnostics and the need for continuous validation further add pressure on manufacturers. The market restrictions create obstacles which slow down the development of new products while they limit the speed at which companies can introduce their latest products.
The north america enteric disease testing market encounters major challenges because most facilities lack essential resources and trained personnel needed for testing adoption. Most healthcare facilities do not have access to advanced diagnostic tools which require trained personnel to operate complex systems. Innovative testing methods face adoption challenges because funding gaps and budget constraints restrict their implementation. The growth barriers decrease the overall number of people who can access contemporary diagnostic technologies.
Region Insights
The United States maintains its superior status among major economies because of its developed healthcare systems and widespread knowledge of medical tests. The combination of public health initiatives and better testing access leads to ongoing Canadian progress. The market will grow if companies maintain their funding and improve their existing facilities.
The people in different regions show different levels of demand because of their population size and their understanding of food safety regulations and infectious disease transmission. Cities experience faster technology adoption because residents can easily access healthcare services while rural communities experience slower adoption rates because of their limited access to testing facilities. The ongoing development of regional healthcare systems will create equal market growth and increased access to diagnostic services.
Recent Development News
In April 2026, Quest Diagnostics raises 2026 forecast, CEO highlights broad-based growth: A major shift in the U.S. beverage landscape as 7UP expands into energy drinks through a strategic collaboration. This move reflects increasing crossover between soda and energy drink segments to capture younger consumers.
Source: https://www.reuters.com
In April 2026, Abbott trims 2026 profit forecast on Exact deal hit, shares fall: Abbott’s update followed its Exact Sciences acquisition, which adds the Cologuard stool-based colorectal screening franchise to its U.S. diagnostics portfolio. That makes this one especially relevant to stool-based screening and GI testing workflows.
Source: https://www.reuters.com
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 1092.9 Million |
|
Market size value in 2026 |
USD 1114.8 Million |
|
Revenue forecast in 2033 |
USD 1306.9 Million |
|
Growth rate |
CAGR of 2.30% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
North America (Canada, The United States, and Mexico) |
|
Key company profiled |
Thermo Fisher, Abbott, Roche, Bio-Rad, Qiagen, Danaher, Agilent, BD, Siemens Healthineers, BioMerieux, Hologic, Cepheid, PerkinElmer, Illumina, GenScript. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Bacterial Testing, Viral Testing, Parasitic Testing, Molecular Diagnostics, Rapid Tests, Others), By Application (Gastroenteritis, Cholera, Salmonella, E. coli, Foodborne Diseases, Others), By End-User (Hospitals, Labs, Clinics, Research Institutes, Others), By Technology (PCR, ELISA, Culture Methods, Sequencing, Rapid Kits, Others). |
How Can New Companies Establish a Strong Foothold in the North America Enteric Disease Testing Market?
New market entrants in North America enteric disease testing can establish competitive advantages by developing specialized tests which address diagnostic requirements that current medical techniques do not fulfill. Companies that establish their presence in the market through effective identification of particular detection challenges which require faster results and better accuracy and wider access will gain competitive advantages. Market growth drivers and industry trends will create better brand visibility for companies that establish their business model according to these factors.
The North America enteric disease testing market will achieve success through innovation which depends on advanced technologies including AI-based diagnostic systems and portable testing equipment. Companies that create unique products which solve practical problems such as slow diagnosis and restricted laboratory capacity will achieve market success. Companies need to invest in research activities and develop technologies which can expand their business operations to maintain their market presence and achieve operational effectiveness.
The North America enteric disease testing market will experience accelerated growth through partnerships which connect hospitals with diagnostic laboratories and public health entities. Organizations will partner with each other to enhance their distribution capabilities while building trust and reaching more patients. Companies that create intelligent diagnostic solutions or cloud-based data systems demonstrate how innovative approaches can help them gain market entry while providing solutions to healthcare system challenges.
Key North America Enteric Disease Testing Market Company Insights
The market experiences intense rivalry because existing diagnostic firms concentrate their efforts on developing new products and enhancing their testing capabilities to stay in competitive positions. The company will achieve product differentiation through its ongoing research and development expenditures on molecular diagnostics and automated systems. Healthcare providers will base their purchasing choices on two factors, which include strategic pricing and brand trust.
Market participants use partnerships and acquisitions and regional expansion to boost their market presence and expand their market share. Established companies with extensive distribution systems and regulatory knowledge will create challenges for new market entrants. The company will establish its future competitive advantage through three essential objectives, which include cost efficiency and product development and trustworthy product operation.
Company List
- Thermo Fisher
- Abbott
- Roche
- Bio-Rad
- Qiagen
- Danaher
- Agilent
- BD
- Siemens Healthineers
- BioMerieux
- Hologic
- Cepheid
- PerkinElmer
- Illumina
- GenScript
What are the Key Use-Cases Driving the Growth of the North America Enteric Disease Testing Market?
The north american enteric disease testing market experiences its primary market growth because clinical diagnostics have developed several use cases which enable doctors to identify gastrointestinal infections through their initial symptoms thus enabling them to deliver treatment to patients while decreasing their need for hospital services. Hospitals and diagnostic laboratories depend on sophisticated testing methods to detect dangerous pathogens which include Salmonella and E. coli with exceptional precision.
The need for trustworthy quick diagnostic methods keeps increasing because patient numbers are rising while people become more aware of foodborne diseases. Public health surveillance represents another critical use-case fueling the north america enteric disease testing market. Testing data enables government agencies together with health organizations to monitor outbreaks while tracking infection patterns and executing necessary control measures.
This approach enhances response efficiency while decreasing the spread of diseases which occur during major outbreaks that affect urban areas. The implementation of data analytics allows organizations to enhance their capabilities for predicting and managing outbreaks. The north american enteric disease testing market expands because food safety companies and manufacturing sectors establish protocols to prevent contamination throughout their production and supply processes.
Companies utilize regular testing procedures for both their raw materials and finished products to achieve compliance with regulatory requirements while preventing expensive product recalls. Food processing industries now implement quality assurance measures because they want to ensure consumer safety which has resulted in increased product adoption.
North America Enteric Disease Testing Market Report Segmentation
By Type
- Bacterial Testing
- Viral Testing
- Parasitic Testing
- Molecular Diagnostics
- Rapid Tests
- Others
By Application
- Gastroenteritis
- Cholera
- Salmonella
- E. coli
- Foodborne Diseases
- Others
By End-User
- Hospitals
- Labs
- Clinics
- Research Institutes
- Others
By Technology
- PCR
- ELISA
- Culture Methods
- Sequencing
- Rapid Kits
- Others
Frequently Asked Questions
Find quick answers to common questions.
The approximate North America Enteric Disease Testing Market size for the market will be USD 1306.9 Million in 2033.
The key segments of the North America Enteric Disease Testing Market are By Type (Bacterial Testing, Viral Testing, Parasitic Testing, Molecular Diagnostics, Rapid Tests, Others), By Application (Gastroenteritis, Cholera, Salmonella, E. coli, Foodborne Diseases, Others), By End-User (Hospitals, Labs, Clinics, Research Institutes, Others), By Technology (PCR, ELISA, Culture Methods, Sequencing, Rapid Kits, Others).
Major players in the North America Enteric Disease Testing Market are Thermo Fisher, Abbott, Roche, Bio-Rad, Qiagen, Danaher, Agilent, BD, Siemens Healthineers, BioMerieux, Hologic, Cepheid, PerkinElmer, Illumina, GenScript.
The current market size of the North America Enteric Disease Testing Market is USD 1092.9 Million in 2025.
The North America Enteric Disease Testing Market CAGR is 2.30%.
- Thermo Fisher
- Abbott
- Roche
- Bio-Rad
- Qiagen
- Danaher
- Agilent
- BD
- Siemens Healthineers
- BioMerieux
- Hologic
- Cepheid
- PerkinElmer
- Illumina
- GenScript
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)









