North America Diagnostic Contract Manufacturing Market Size & Forecast:
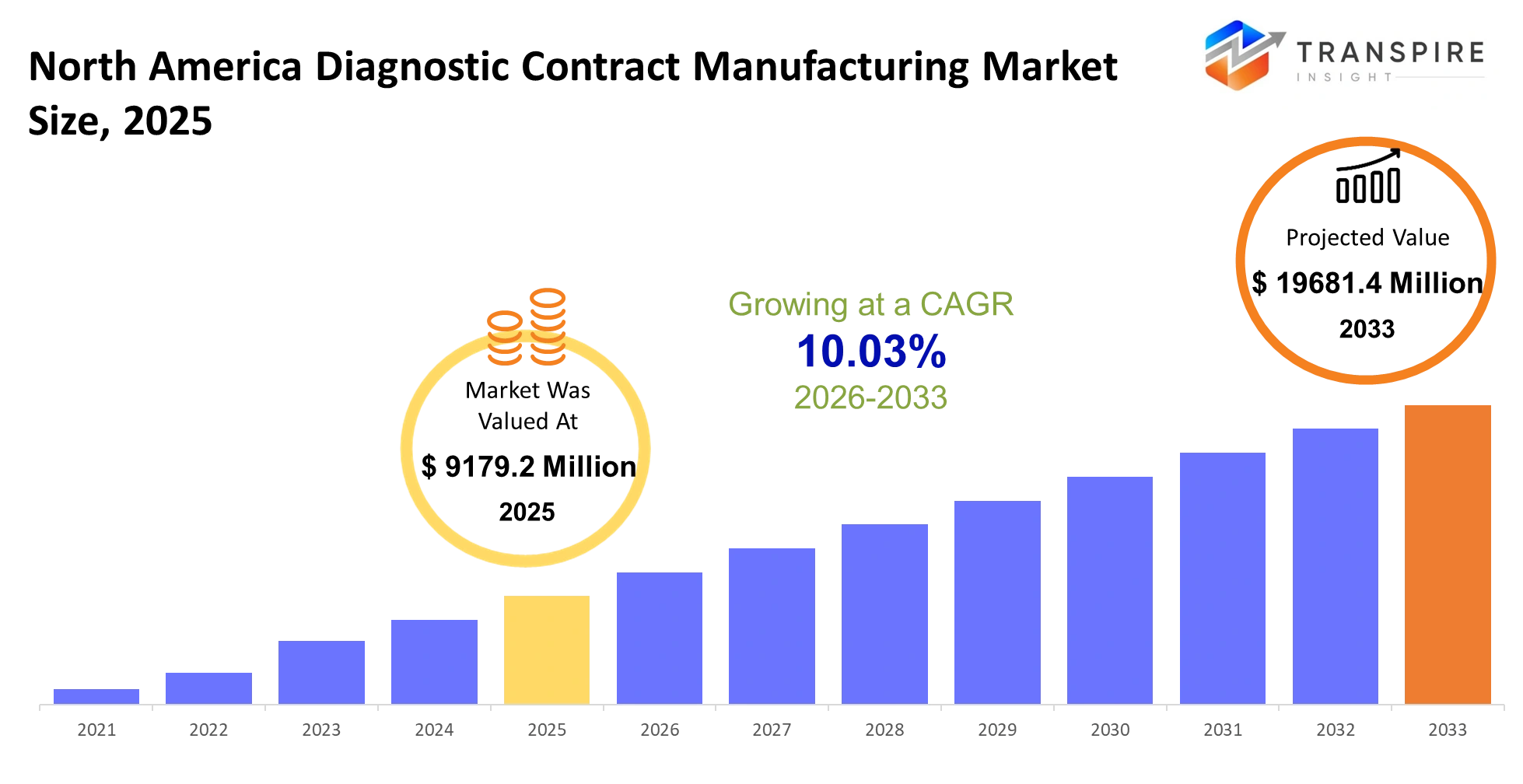
- North America Diagnostic Contract Manufacturing Market Size 2025: USD 9179.2 Million
- North America Diagnostic Contract Manufacturing Market Size 2033: USD 19681.4 Million
- North America Diagnostic Contract Manufacturing Market CAGR: 10.03%
- North America Diagnostic Contract Manufacturing Market Segments: By Type (Reagents Manufacturing, Device Manufacturing, Kit Manufacturing, Assay Development, Packaging Services, Others), By Application (IVD, Molecular Diagnostics, Clinical Diagnostics, Research, Others), By End-User (Pharma, Biotech, Diagnostics Companies, Labs, Others), By Service (OEM, ODM, Private Label, Others).
To learn more about this report, Download Free Sample Report
North America Diagnostic Contract Manufacturing Market Summary:
The North America Diagnostic Contract Manufacturing Market size is estimated at USD 9179.2 Million in 2025 and is anticipated to reach USD 19681.4 Million by 2033, growing at a CAGR of 10.03% from 2026 to 2033. The North America Diagnostic Contract Manufacturing Market operates as a hidden force within contemporary healthcare systems which helps diagnostic firms increase their production capacity for test kits and reagents and instruments without needing to establish their own production facilities. The solution enables innovators to transition from their prototype stage into commercial production because it eliminates both time delays and excessive expenses while helping them follow regulatory standards and maintain product excellence.
The market has undergone a fundamental transformation during the last three to five years because companies now establish comprehensive relationships with manufacturing partners who manage all aspects of their production processes from initial design through testing to full-scale output. The COVID-19 pandemic accelerated this transformation because it revealed weaknesses in internal manufacturing systems which forced diagnostic companies to outsource their production needs during critical testing periods.
The business landscape now requires companies to use special operational methods which enable them to maintain flexible production abilities while working with partners who have achieved full compliance with regulatory standards and these requirements lead to extended contractual agreements which result in companies delivering more valuable services throughout the entire industry.
Key Market Insights
- The United States maintains control over the North America Diagnostic Contract Manufacturing Market by holding more than 80% of the market share which will occur in 2025 because of its advanced healthcare infrastructure.
- Canada stands as the fastest expanding area until 2030 because of rising biotech investments together with its beneficial regulatory environment.
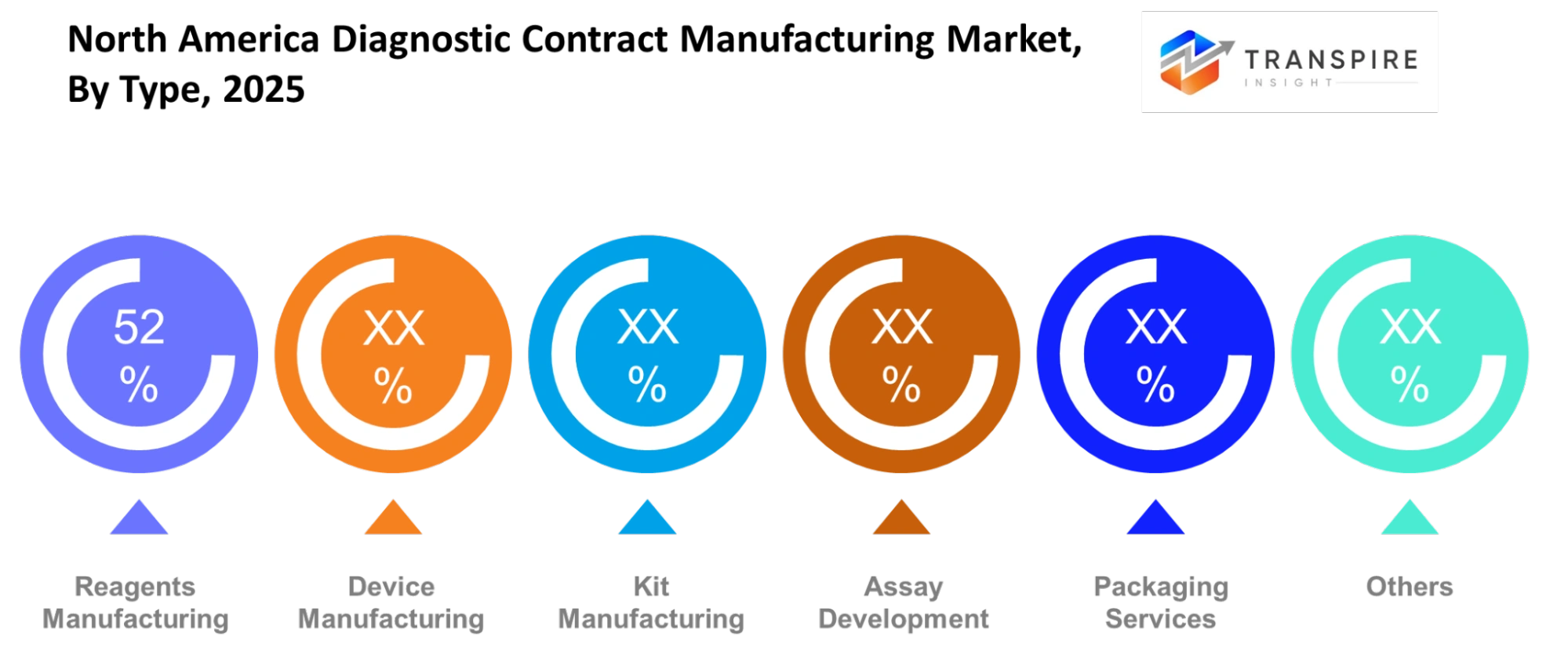
- The manufacturing of reagents and consumables will take the leading market position in 2025 with a 45% market share because of the demand from diagnostic testing which requires large testing volumes.
- The manufacturing sector for diagnostic instruments holds the second highest market share due to increased use of automated diagnostic systems.
- The fastest expanding segment includes assay development and validation services which will experience substantial growth until 2030 because of their complex testing processes.
- The North America Diagnostic Contract Manufacturing Market exists mainly through molecular diagnostics which control more than 50% of the market due to the growth of infectious disease testing and genetic testing.
- The application segment for point-of-care diagnostics shows the highest growth rate because of the increasing use of decentralized testing together with quick diagnostic testing methods.
- The share of diagnostic companies which reaches almost 60% uses contract manufacturers to obtain production capabilities that meet their needs for expansion and regulatory compliance.
- Biotech startups represent the fastest-growing end-user segment, increasing outsourcing to reduce capital expenditure and accelerate commercialization timelines.
What are the Key Drivers, Restraints, and Opportunities in the North America Diagnostic Contract Manufacturing Market?
The North America Diagnostic Contract Manufacturing Market currently experiences rapid growth because medical institutions now require advanced molecular testing abilities which utilize genetic diagnostics. The need for rapid diagnostic tests developed after COVID-19 because scientists required medical testing solutions which could be produced at high production levels. The diagnostic industry now confronts more stringent technical requirements and regulatory demands which create expensive in-house production methods that require time to develop new products. Companies now prefer to outsource their production needs to contract manufacturers who possess advanced expertise in PCR and next-generation sequencing and regulatory compliance.
The regulatory framework which exists under the U.S. Food and Drug Administration creates an essential structural barrier for organizations. Organizations must conduct extensive validation tests and produce complete documentation and perform facility assessments which will prolong their production process while increasing their operational expenses.
The requirements create obstacles for smaller contract manufacturers who cannot fulfill them which hinders their ability to enter the market and grow their operational capacity. The existing limitations create difficulties for supply chain operations because they prohibit the introduction of new diagnostic solutions which causes delays in revenue generation throughout the business operations.
The healthcare system presents a significant opportunity to develop point-of-care testing and decentralized diagnostic technologies which will enhance outpatient services and home medical treatment. The development of microfluidics and portable diagnostic systems enables the production of smaller and faster devices which still need to meet manufacturing quality standards. Contract manufacturers that operate automated production systems through their modular production lines will gain control over this market trend.
What Has the Impact of Artificial Intelligence Been on the North America Diagnostic Contract Manufacturing Market?
The operational processes of diagnostic contract manufacturers experience transformation through artificial intelligence together with advanced digital technologies which particularly impact high-volume production environments that demand strict compliance. The AI-powered automation system enables organizations to monitor their production lines in real-time through its capability to track reagent mixing processes and assay assembly operations and cleanroom facility environmental controls. The smart control systems utilize computer vision together with sensor data to identify defects which enables them to maintain consistent batch quality while reducing the need for manual inspections. The systems provide automatic compliance tracking capabilities by documenting all necessary process parameters that facilities require during regulatory audits.
Organizations use machine learning models for both predictive maintenance purposes and process optimization tasks. Manufacturers who analyze equipment performance data can use that information to predict which critical assets will fail including bioreactors and filling systems that need maintenance. Predictive analytics enhances yield forecasting accuracy while reducing material waste which results in lower unit costs and improved operational efficiency. AI-driven scheduling tools have enabled facilities to boost production uptime by 15 to 20 percent while they reduced the time needed to establish new diagnostic kits.
The high costs involved in integration together with the disjointed nature of data systems continue to limit the spread of new technologies. The majority of legacy manufacturing systems from earlier times lack standard data formats which prevents organizations from developing accurate models that can be used to deploy AI technologies throughout different production sites.
Key Market Trends
- Diagnostic firms have changed their outsourcing strategy since 2020 by establishing strategic partnerships which now cover complete manufacturing and testing processes in more than 60% of their contracts.
- North American companies adopted dual-source manufacturing techniques because post-pandemic supply disruptions forced them to create additional production capacity while decreasing dependence on single-site manufacturing operations.
- Between 2021 and 2025, businesses adopted automated fill-finish and assembly lines at nearly 25% of their operations which resulted in improved batch consistency and decreased human mistakes during regulated processes.
- Manufacturers had to enhance their documentation systems and develop digital quality management systems following 2022 emergency authorization expiration because regulatory bodies began stricter evaluation processes.
- Thermo Fisher Scientific and Danaher Corporation expanded their operational capabilities through acquisitions which enabled them to merge smaller contract manufacturers into their unified service networks.
- The period between 2020 and 2024 showed a sharp increase in molecular and genetic diagnostics demand which led to PCR and sequencing manufacturing output doubling because more clinics started using these diagnostic technologies.
- By 2025 biotech startups had increased their manufacturing outsourcing rates to over 70% which helped them avoid costly facility investments while speeding up their product development processes.
- High-throughput diagnostic production facilities achieved equipment downtime reductions of up to 20% through the implementation of digital manufacturing tools that included AI-based scheduling and predictive maintenance systems.
- Companies began nearshoring operations after 2021 to establish manufacturing sites near the United States which helped them reduce cross-border logistics expenses and decrease regulatory processing times.
North America Diagnostic Contract Manufacturing Market Segmentation
By Type
The dominant sector of reagents manufacturing exists because diagnostic processes require high volumes of their products at all their operational times. The kit manufacturing sector operates as a strong competitor because its testing systems enable medical facilities to use their products with ease. The device manufacturing sector maintains its market position because automation technologies drive its operations although they increase production costs and length the time needed to create new products.
The diagnostic field requires scientists to develop new testing methods because doctors need advanced solutions for complex medical cases which they test through molecular and genetic analysis. The company needs packaging services and other support functions because they help the company achieve compliance and prepare products for distribution. The sub-segments of business activities show growth because clients now prefer to buy complete manufacturing solutions from companies that handle all production processes. The current market trend enables contract manufacturers to expand their operations into development and validation processes which results in higher profit margins but requires them to enhance their regulatory abilities and technical knowledge.
To learn more about this report, Download Free Sample Report
By Application
Medical diagnostics through molecular methods establishes itself as the leading testing method because hospitals and laboratories now use PCR and sequencing technologies for both infectious diseases and cancer diagnostics. The medical testing field maintains its strong presence because hospitals and laboratories perform diagnostic tests on a regular basis. In vitro diagnostics serves as an extensive category that combines both diagnostic and monitoring products, which creates constant demand for manufacturing facilities.
Research applications show ongoing expansion, which originates from academic and biotech institutions that create new testing methods. Niche applications create additional demand for products, which remain below the level required for significant market effect. The production needs of each application differ from one another because molecular diagnostics demand more accurate results and thorough testing processes. The different requirements compel manufacturers to establish their specialized production spaces and implement their quality control procedures. The upcoming expansion will focus on complex applications because they create technical challenges that restrict market entry while enabling businesses to charge higher prices and secure extended service agreements.
By End-User
The market is primarily controlled by diagnostic companies which have established testing operations and depend on their manufacturing partners who can expand their production capabilities. The biotech sector is expanding at its fastest rate because companies create new products and struggle to produce them in-house. Pharmaceutical companies develop companion diagnostics which enable testing to become part of their treatment programs. Laboratories continue to require testing services although they usually choose to outsource their testing work to specialized providers who handle specific tasks.
The other end users create minimal impact on the overall market. The different organizations exhibit unique outsourcing patterns because larger diagnostic companies require complete service solutions whereas smaller biotech firms depend on quick and flexible options. The current situation pushes contract manufacturers to expand their service options and develop customized ways of working together with customers. The increase in contract agreements between end users will occur through consolidation while biotech companies require flexible manufacturing options to meet their ongoing needs.
By Service
The OEM company leads the market through its services because customers require high-volume manufacturing which meets specific standards for existing diagnostic brands. The ODM business model is growing in popularity because clients need product development partners who will handle their research and development responsibilities. Private label services target cost-sensitive customers who need standardized diagnostic kits while other services meet specific market demands. Companies now differentiate their services through technical expertise and their ability to handle regulatory requirements beyond pricing differences.
Providers now need integrated capabilities to deliver design-to-commercialization solutions because clients demand effortless transitions between these two processes. The industry now requires ODM and hybrid service models which establish early product development relationships. The future will bring success to manufacturers who merge design skills with production capabilities because this approach helps them launch products faster while staying current with market changes in diagnostic technology.
What are the Key Use Cases Driving the North America Diagnostic Contract Manufacturing Market?
The main reason organizations adopt molecular diagnostics technology is because it provides essential testing capabilities needed for both infectious diseases and cancer research. Internal facilities fail to meet the production needs of high-throughput PCR and sequencing assays which require both accurate results and extensive manufacturing capacity. Diagnostic companies depend on contract manufacturers to manage their complex needs which include developing reagents assembling tests and growing their operations according to regulatory standards. The testing case creates high demand because testing requirements change frequently which needs production capacity that matches medical demands.
The process now supports both routine clinical diagnostic procedures and companion diagnostic tests which support drug development. Hospitals and reference laboratories need standard test kits for their operations while pharmaceutical companies use contract manufacturing to produce biomarker tests that accompany targeted therapies. Biotech companies partner with contract manufacturers to speed up their initial product development process which allows them to bypass the need for constructing entire production facilities.
The diagnostics field now requires portable devices and rapid test kits to create precise manufacturing systems which enable their testing capabilities in both home and decentralized settings. The rise of digital health systems and remote patient monitoring technologies has led to an increasing requirement for affordable manufacturing solutions which can produce diagnostic products used directly by consumers.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 9179.2 Million |
|
Market size value in 2026 |
USD 10079.1 Million |
|
Revenue forecast in 2033 |
USD 19681.4 Million |
|
Growth rate |
CAGR of 10.03% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
North America (Canada, The United States, and Mexico) |
|
Key company profiled |
Thermo Fisher, Danaher, Abbott, Roche, Siemens Healthineers, BD, Bio-Rad, Agilent, Merck KGaA, Qiagen, Eurofins, WuXi AppTec, Catalent, Lonza, Sartorius. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Reagents Manufacturing, Device Manufacturing, Kit Manufacturing, Assay Development, Packaging Services, Others), By Application (IVD, Molecular Diagnostics, Clinical Diagnostics, Research, Others), By End-User (Pharma, Biotech, Diagnostics Companies, Labs, Others), By Service (OEM, ODM, Private Label, Others). |
Which Regions are Driving the North America Diagnostic Contract Manufacturing Market Growth?
The United States Diagnostic Contract Manufacturing Market in North America is dominated by the United States because of its regulatory system and its concentration of diagnostic companies. The U.S. Food and Drug Administration monitors production activities to maintain manufacturing standards, which forces businesses to work with established contract manufacturing companies. The region establishes fast product development and scale-up processes through its extensive network of biotechnology research centers and manufacturing partners. The environment enables organizations to maintain their market position because it enables them to bring products to market at a faster rate while customers need premium manufacturing solutions.
Canada maintains its position as a dependable market partner because healthcare investments maintain their course and regulatory standards align with international guidelines. The North American market develops with controlled growth, which the regioning public healthcare systems and their long-term procurement methods. The government provides strong funding support to life sciences and diagnostics programs, which enables contract manufacturers to receive consistent production volumes. The company establishes partnerships to maintain production activities, which creates a predictable income stream that enables stable revenue growth.
Mexico has become the fastest-growing area because of its nearshoring practices and its ability to offer manufacturing services at competitive prices. The country's diagnostic production capacity now receives better conditions because of recent industrial capacity improvements and new regulatory framework developments. Companies are shifting manufacturing operations to Mexico because it enables them to decrease supply chain risks and achieve faster production times.
Who are the Key Players in the North America Diagnostic Contract Manufacturing Market and How Do They Compete?
The North America Diagnostic Contract Manufacturing Market shows moderate consolidation because two major companies control all high-margin operations through their vertical integration.) Smaller specialists work in specialized markets that include assay development and packaging. Companies need to achieve both operational capacity and extensive knowledge of regulations in order to succeed in contract bidding. The primary basis of competition has shifted from cost to technology integration and service scope, as diagnostic companies prefer partners that can handle design transfer, validation, and commercial manufacturing under one system. Established companies expand their operations to maintain market share while new companies try to compete by providing adaptable services that can be delivered more quickly.
Thermo Fisher Scientific competes through full-service integration, offering everything from reagent production to finished kit assembly, which reduces coordination risk for clients. Danaher Corporation uses its diagnostic subsidiary network to deliver manufacturing services that use advanced technology in molecular diagnostics through standardized processes. Eurofins Scientific provides regulatory testing and validation expertise as a unique service that helps clients achieve product approvals while they build manufacturing capabilities.
Avantor maintains its market position through its focus on materials and consumables production because it controls supply chain operations and produces high-purity products. Biotech companies choose WuXi AppTec as their partner because the company combines extensive global capacity with complete research and manufacturing solutions.
Company List
- Thermo Fisher
- Danaher
- Abbott
- Roche
- Siemens Healthineers
- BD
- Bio-Rad
- Agilent
- Merck KGaA
- Qiagen
- Eurofins
- WuXi AppTec
- Catalent
- Lonza
- Sartorius
Recent Development News
In April 2026, Thermo Fisher Scientific Signs Agreement to Sell its Microbiology Business to Astorg: Thermo Fisher Scientific announced the sale of its microbiology diagnostics business to Astorg for approximately $1.075 billion. This divestment reflects portfolio optimization and increased private equity interest in diagnostic manufacturing assets.
Source: https://ir.thermofisher.com
In January 2026, Thermo Fisher wins contracts as pharma shifts production to US, CEO says: Thermo Fisher Scientific secured multiple new manufacturing contracts driven by reshoring of pharmaceutical and diagnostic production to the United States. This shift strengthens domestic contract manufacturing demand and long-term capacity utilization.
Source: https://www.reuters.com
What Strategic Insights Define the Future of the North America Diagnostic Contract Manufacturing Market?
The North America Diagnostic Contract Manufacturing Market is moving toward tightly integrated, technology-led manufacturing ecosystems which enable partners to manage design work and validation procedures along with their ability to create scalable production systems through one digital platform. The market now requires complete testing solutions which include both testing procedures and testing equipment because testing methods have become more complicated and testing rules have become more demanding. The next 5 to 7 years will result in value creation for manufacturers who achieve faster product launch times through their use of automated systems and data management solutions and regulatory compliance knowledge.
The market concentration risk which people cannot see develops because few major companies gain control over manufacturing processes which require high levels of expertise. The situation will restrict customers from altering prices while creating delays to their operations during demand increases or supply chain interruptions. The market now presents new possibilities in both at-home and decentralized diagnostic testing because miniaturized testing devices need. The market now requires new manufacturing methods to produce digital health solutions which use miniaturized testing devices.
Market participants should invest in modular production systems which offer flexible production capabilities to handle both high-volume and small-batch diagnostics. The systems will enable companies to maintain operational capacity while they pursue upcoming market demands.
North America Diagnostic Contract Manufacturing Market Report Segmentation
By Type
- Reagents Manufacturing
- Device Manufacturing
- Kit Manufacturing
- Assay Development
- Packaging Services
- Others
By Application
- IVD
- Molecular Diagnostics
- Clinical Diagnostics
- Research
- Others
By End-User
- Pharma
- Biotech
- Diagnostics Companies
- Labs
- Others
By Service
- OEM
- ODM
- Private Label
- Others
Frequently Asked Questions
Find quick answers to common questions.
The approximate North America Diagnostic Contract Manufacturing Market size for the market will be USD 19681.4 Million in 2033.
The key segments of the North America Diagnostic Contract Manufacturing Market are By Type (Reagents Manufacturing, Device Manufacturing, Kit Manufacturing, Assay Development, Packaging Services, Others), By Application (IVD, Molecular Diagnostics, Clinical Diagnostics, Research, Others), By End-User (Pharma, Biotech, Diagnostics Companies, Labs, Others), By Service (OEM, ODM, Private Label, Others).
Major players in the North America Diagnostic Contract Manufacturing Market are Thermo Fisher, Danaher, Abbott, Roche, Siemens Healthineers, BD, Bio-Rad, Agilent, Merck KGaA, Qiagen, Eurofins, WuXi AppTec, Catalent, Lonza, Sartorius.
The current market size of the North America Diagnostic Contract Manufacturing Market is USD 9179.2 Million in 2025.
The North America Diagnostic Contract Manufacturing Market CAGR is 10.03%.
- Thermo Fisher
- Danaher
- Abbott
- Roche
- Siemens Healthineers
- BD
- Bio-Rad
- Agilent
- Merck KGaA
- Qiagen
- Eurofins
- WuXi AppTec
- Catalent
- Lonza
- Sartorius
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)









