France Independent Diagnostic Testing Facility Market Size & Forecast:
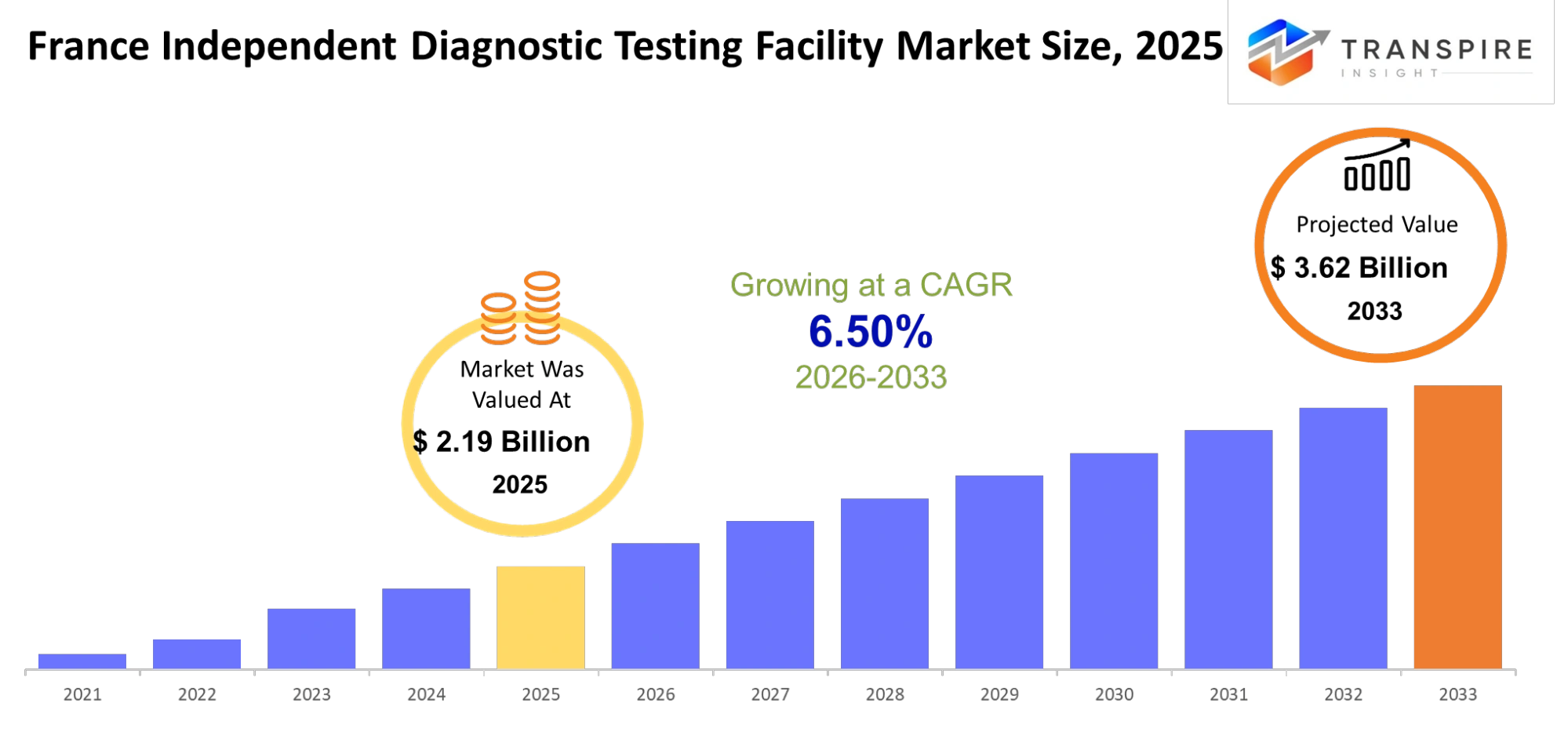
- France Independent Diagnostic Testing Facility Market Size 2025: USD 2.19 Billion
- France Independent Diagnostic Testing Facility Market Size 2033: USD 3.62 Billion
- France Independent Diagnostic Testing Facility Market CAGR: 6.50%
- France Independent Diagnostic Testing Facility Market Segments: By Test Type (Clinical Chemistry, Immunoassay, Molecular Diagnostics, Hematology),By End User (Hospitals, Diagnostic Laboratories).
To learn more about this report, Download Free Sample Report
France Independent Diagnostic Testing Facility Market Summary:
The France Independent Diagnostic Testing Facility Market size is estimated at USD 2.19 Billion in 2025 and is anticipated to reach USD 3.62 Billion by 2033, growing at a CAGR of 6.50% from 2026 to 2033. The independent diagnostic testing market of France shows consistent growth because people need preventive healthcare services and early disease detection and decentralized diagnostic testing services. The increasing prevalence of chronic diseases which include diabetes and cardiovascular diseases and cancer will result in higher testing demand at independent testing facilities. The market will gain traction in urban and semi-urban areas because laboratory networks are expanding and people prefer affordable and quicker diagnostic methods.
The french independent diagnostic testing facility market uses technological progress to develop its business operations through its molecular diagnostic systems and artificial intelligence analysis and digital reporting tools. Facilities are investing in automation and smart workflows to achieve better accuracy while decreasing turnaround times and handling increased testing capacity. The combination of telehealth and home sample collection services will make it easier for patients to access care while providing them with better treatment options.
The france independent diagnostic testing facility market demonstrates strong growth prospects but encounters obstacles from three main areas which include regulatory compliance and pricing pressure and competition that exists with hospital-based laboratories. Strategic partnerships together with testing service innovation and entering new markets will create new business growth opportunities. The market will develop in the long term because organizations concentrate on maintaining quality and operating efficiency and implementing new technologies.
What Has the Impact of Artificial Intelligence Been on the France Independent Diagnostic Testing Facility Market?
Artificial intelligence is implementing its power to transform the France independent diagnostic testing facility market through its ability to enhance operational performance and clinical testing accuracy. The France independent diagnostic testing facility market employs artificial intelligence for data analysis, which helps to speedily process extensive diagnostic data including medical imaging results, pathology slides and laboratory reports.
The France independent diagnostic testing facility market uses artificial intelligence to improve demand forecasting and workflow management. Smart automation tools will streamline laboratory processes, which will decrease manual work and enhance processing speed. AI-driven systems help facilities manage testing volumes more effectively which leads to better resource allocation and cost control. Supply chain optimization operates as a major advantage because predictive models will assist in controlling inventory levels while decreasing shortages and operational expenses throughout diagnostic networks.
AI makes it possible to develop new testing methods which create customized diagnostic solutions for the France independent diagnostic testing facility market. Advanced algorithms support tailored testing recommendations based on patient history and genetic data, improving treatment outcomes.
Key Market Trends & Insights:
- The Île-de-France region controls more than 30% of the France independent diagnostic testing facility market because of its extensive healthcare facilities which support its market reach.
- The Southern regions, which include Provence-Alpes-Côte d'Azur show the fastest growth at 7% annual growth rate because of their growing older population and rising demand for home testing solutions.
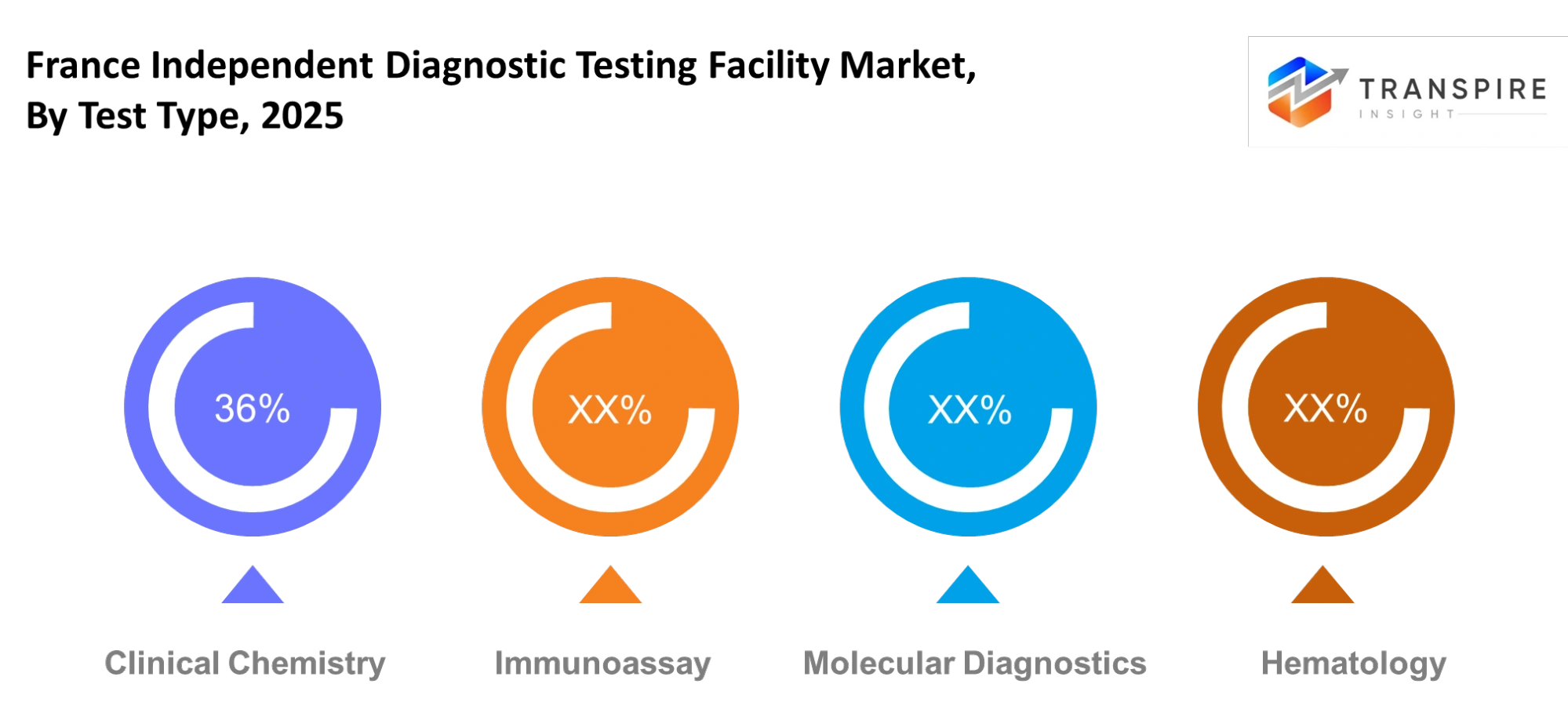
- Clinical chemistry leads the market in 2025 with 34.9% market share, driven by hospitals conducting high-volume routine diagnostics testing at their facilities. Immunoassay functions as the second largest market segment which businesses drive through their need for infectious disease testing and hormone testing solutions.
- The molecular diagnostics market segment will grow at 8% annual growth rate because of two major trends precision medicine and early disease detection. Preventive healthcare screening operates as the primary application, operating with more than 40% market share, because people increasingly understand the importance of early diagnosis together with regular health assessments.
- Home diagnostic testing for patients has emerged as a new market segment which experiences rapid growth at 9% annual growth rate because of its convenience and its combination with digital health solutions.
- Hospitals control approximately 45% of the market share which makes them the largest segment because they depend on independent testing facilities for their additional medical tests and special diagnostic services.
- The end user market segment for standalone diagnostic laboratories shows the strongest growth because customers increasingly require specialized testing which offers them affordable testing solutions.
- Competitive Landscape & Strategy: The main players Eurofins Scientific and Roche Diagnostics and Siemens Healthineers and Abbott Laboratories and Thermo Fisher Scientific, maintain their market dominance through their capacity to develop new products and their business expansion.
- The companies use their resources to develop AI-based diagnostic technologies, establish strategic partnerships, and expand their operations into new markets to achieve better competitive positioning and grow their business.
France Independent Diagnostic Testing Facility Market Segmentation
By Test Type
- The France independent diagnostic testing facility market involves multiple testing categories which enable precise disease identification and tracking throughout healthcare systems. The clinical chemistry testing field maintains its strong market position because healthcare professionals use it regularly for blood tests and metabolic panel assessments, and organ function tests. Medical facilities need these specific diagnostic assessments to conduct patient evaluations and implement preventive health programs.
- The rising need for advanced testing methods is making immunoassay and molecular diagnostics more valuable in the current testing market. Hormone testing and infectious disease identification, and cancer screening all benefit from immunoassay testing while molecular diagnostics will detect complex diseases through their analysis of DNA and RNA. The medical facilities use hematology testing to diagnose and monitor blood disorders which creates continuous demand for this diagnostic method.
To learn more about this report, Download Free Sample Report
By End User
- The independent diagnostic testing facility market across France serves multiple customers, with hospitals acting as the primary demand center for diagnostic testing services. Hospitals will depend on independent facilities to handle their additional testing needs while they work to achieve quicker result processing times. The partnership enables better patient treatment through two methods, which include reduced waiting times and expanded access to advanced diagnostic testing.
- The diagnostic laboratory sector is an important market segment due to the rising need for independent testing centres that offer both standard and advanced diagnostic testing. Independent laboratories will develop new services while implementing automated systems to boost operational efficiency. The combination of rising preventive health awareness and increasing health check-up frequency will create ongoing demand from both urban and semi-urban areas, which will drive this market segment forward.
What are the Main Challenges for the France Independent Diagnostic Testing Facility Market Growth?
The **france independent diagnostic testing facility market** faces technical and operational obstacles which reduce operational efficiency and limit system expansion capabilities. Testing accuracy remains challenging to maintain at constant levels during high-volume testing because advanced diagnostic methods which include molecular and genetic testing have seen increased demand. New technology integration interrupts laboratory workflows because supply chain issues with reagents and equipment create testing capacity and service delivery delays.
The france independent diagnostic testing facility market faces obstacles from manufacturing and commercialization constraints. Diagnostic solutions face longer approval times and higher compliance expenses because of strict regulatory requirements and quality standards which compel organizations to meet multiple standards.
The france independent diagnostic testing facility market faces ongoing restrictions because of infrastructure and adoption challenges. Service access becomes limited because laboratory professionals are scarce and diagnostic centers are not evenly distributed throughout rural regions. The digital infrastructure gaps which exist together with the slow adoption of integrated health systems create operational inefficiencies because they prevent smooth data sharing and patient management processes.
The france independent diagnostic testing facility market becomes more complex because of market competition which combines with increasing risks. In-house diagnostic services provided by large hospital networks together with integrated healthcare providers create additional competitive challenges. Organizations must make constant investments because rapid technological advancements require ongoing development.
Country Insights
The French independent diagnostic testing facility market displays substantial regional differences because Île-de-France operates as the most developed area which has both a high population and an efficient medical system. The combination of major hospitals and research centers and private laboratories will create a continuous need for diagnostic services. The urban population displays high knowledge about preventive healthcare which creates increased testing needs for routine health screenings.
The Achievements of Auvergne-Rhône-Alpes provide a continuous contribution to the France independent diagnostic testing facility market through its expanding network of diagnostic centers and laboratories. The combined growth of industrial activities and healthcare spending will lead to higher requirements for occupational health tests and clinical diagnostic assessments. The development of better transportation links together with improved public health facilities will create constant growth which enables better distribution of services to nearby communities.
The southern part of France, including Provence-Alpes-Côte d'Azur experiences increasing healthcare needs because the population ages and people prefer medical care at home. Combining telehealth services with mobile sample collection will improve access to healthcare in semi-urban areas. The French independent diagnostic testing facility market will continue to grow regionally through the expansion of private diagnostic companies and digital service platforms.
Recent Development News
In April 2026, Eurofins Scientific launched an advanced molecular diagnostics platform in France. The initiative aims to expand high-precision testing capabilities and improve turnaround time across independent diagnostic facilities.https://www.eurofins.com
In March 2026, Roche Diagnostics announced the expansion of its digital pathology solutions across French laboratory networks. The move supports AI-enabled diagnostics and strengthens collaboration with independent testing facilities to enhance clinical decision-making.https://www.roche.com
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 2.19 Billion |
|
Market size value in 2026 |
USD 2.33 Billion |
|
Revenue forecast in 2033 |
USD 3.62 Billion |
|
Growth rate |
CAGR of 6.50% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
France |
|
Key company profiled |
Quest Diagnostics, Laboratory Corporation of America, Eurofins Scientific, Synlab, Unilabs, Cerba HealthCare, BioReference Laboratories, Sonic Healthcare, ARUP Laboratories, Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, Danaher, Agilent Technologies, Thermo Fisher Scientific. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Test Type (Clinical Chemistry, Immunoassay, Molecular Diagnostics, Hematology),By End User (Hospitals, Diagnostic Laboratories). |
How Can New Companies Establish a Strong Foothold in the France Independent Diagnostic Testing Facility Market?
New entrants in the france independent diagnostic testing facility market can build a strong foothold by focusing on niche segments such as preventive screening, genetic testing, or rapid diagnostics for urban populations. New companies will create their distinct identity through their specialized services which they will deliver to areas that existing companies do not serve and which require better service delivery and faster service completion.
The France independent diagnostic testing facility market will depend on technology-driven innovation for its growth. Startups can utilize AI-based imaging and cloud-enabled reporting and home sample collection systems to enhance their operational efficiency and improve the patient experience. New diagnostic services that receive support from Doctolib in France now combine digital booking systems with laboratory network access which enables patients to schedule their medical tests from home using testing kits.
Market entry will gain additional strength through strategic partnership development. The organization will establish partnerships with hospitals and clinics and corporate entities to develop a continuous patient stream and testing requirements. New companies in the france independent diagnostic testing facility market can also partner with health-tech firms to integrate telemedicine and diagnostics, creating a more connected care ecosystem.
The company needs to achieve two main objectives which include maintaining competitive pricing and delivering quality products according to established standards. The implementation of automated systems together with standardized operational procedures will lead to decreased business expenses and enhanced precision. New entrants in the france independent diagnostic testing facility market can establish successful market positions through their specialized market approach and their development of essential partnerships and their use of modern technological advancements.
Key France Independent Diagnostic Testing Facility Market Company Insights
The French independent diagnostic testing facility market experiences steady growth because of intense market competition and rising demand for diagnostic services and ongoing technological advancements. The market players will work on developing testing methods that provide faster results and more precise outcomes and easier testing access to meet the increasing healthcare demand. The facilities will achieve efficient testing operations through their investment in automation and digital systems which will enhance their ability to manage workflows while reducing expenses.
The French independent diagnostic testing facility market will experience intensified competitive dynamics because global and regional companies are developing laboratory networks and broadening their service range. Companies will use pricing solutions together with quality control measures and speedy processing times to defend their market positions.
The French independent diagnostic testing facility market will gain advantages from strategic mergers and acquisitions and partnerships which will enhance their ability to operate in multiple markets and develop advanced technological skills. The development of personalized diagnostics together with AI-powered analysis and remote testing methods will enhance service effectiveness. The digital appointment booking system together with the digital report access system will provide customers with better convenience and engagement options.
Company List
- Quest Diagnostics
- Laboratory Corporation of America
- Eurofins Scientific
- Synlab
- Unilabs
- Cerba HealthCare
- BioReference Laboratories
- Sonic Healthcare
- ARUP Laboratories
- Abbott Laboratories
- Roche Diagnostics
- Siemens Healthineers
- Danaher
- Agilent Technologies
- Thermo Fisher Scientific.
What are the Key Use-Cases Driving the Growth of the France Independent Diagnostic Testing Facility Market?
The france independent diagnostic testing facility market is expanding rapidly because more people need decentralized and accessible healthcare services. One of the primary use-cases driving growth is preventive health screening and early disease detection. Independent diagnostic centers provide patients with affordable testing services that deliver results faster than hospitals for cancer diabetes and cardiovascular disease testing which helps hospitals reduce their patient load while delivering better results for patients. The france independent diagnostic testing facility market experiences growth because people are starting to adopt preventive healthcare services.
Chronic disease management depends on regular diagnostic monitoring as its main diagnostic application. Facilities deliver routine blood testing services and imaging services and pathology services to patients who need ongoing medical care in non-hospital environments. The france independent diagnostic testing facility market continues to grow because more people start using advanced diagnostic technologies which include molecular diagnostics and AI-powered imaging systems
The market expansion benefits from industrial and occupational health testing. Manufacturing and corporate companies use independent facilities to perform employee health screenings and drug testing and to ensure workplace safety compliance. The france independent diagnostic testing facility market will experience growth through telehealth home sample collection and digital reporting platform integration which will enable operational expansion.
France Independent Diagnostic Testing Facility Market Report Segmentation
By Test Type
- Clinical Chemistry
- Immunoassay
- Molecular Diagnostics
- Hematology
By End User
- Hospitals
- Diagnostic Laboratories
Frequently Asked Questions
Find quick answers to common questions.
The approximate France Independent Diagnostic Testing Facility Market size for the market will be USD 3.62 Billion in 2033.
Key segments for the France Independent Diagnostic Testing Facility Market are TBy Test Type (Clinical Chemistry, Immunoassay, Molecular Diagnostics, Hematology),By End User (Hospitals, Diagnostic Laboratories).
Major France Independent Diagnostic Testing Facility Market players are Quest Diagnostics, Laboratory Corporation of America, Eurofins Scientific, Synlab, Unilabs, Cerba HealthCare, BioReference Laboratories, Sonic Healthcare, ARUP Laboratories, Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, Danaher, Agilent Technologies, Thermo Fisher Scientific.
The France Independent Diagnostic Testing Facility Market size is USD 2.19 Billion in 2025.
The France Independent Diagnostic Testing Facility Market CAGR is 6.50%.
- Quest Diagnostics
- Laboratory Corporation of America
- Eurofins Scientific
- Synlab
- Unilabs
- Cerba HealthCare
- BioReference Laboratories
- Sonic Healthcare
- ARUP Laboratories
- Abbott Laboratories
- Roche Diagnostics
- Siemens Healthineers
- Danaher
- Agilent Technologies
- Thermo Fisher Scientific.
Recently Published Reports
-
Dec 2024
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Feb 2025
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Feb 2025
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Dec 2025
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)