North America STD Diagnostics Market Size & Forecast:
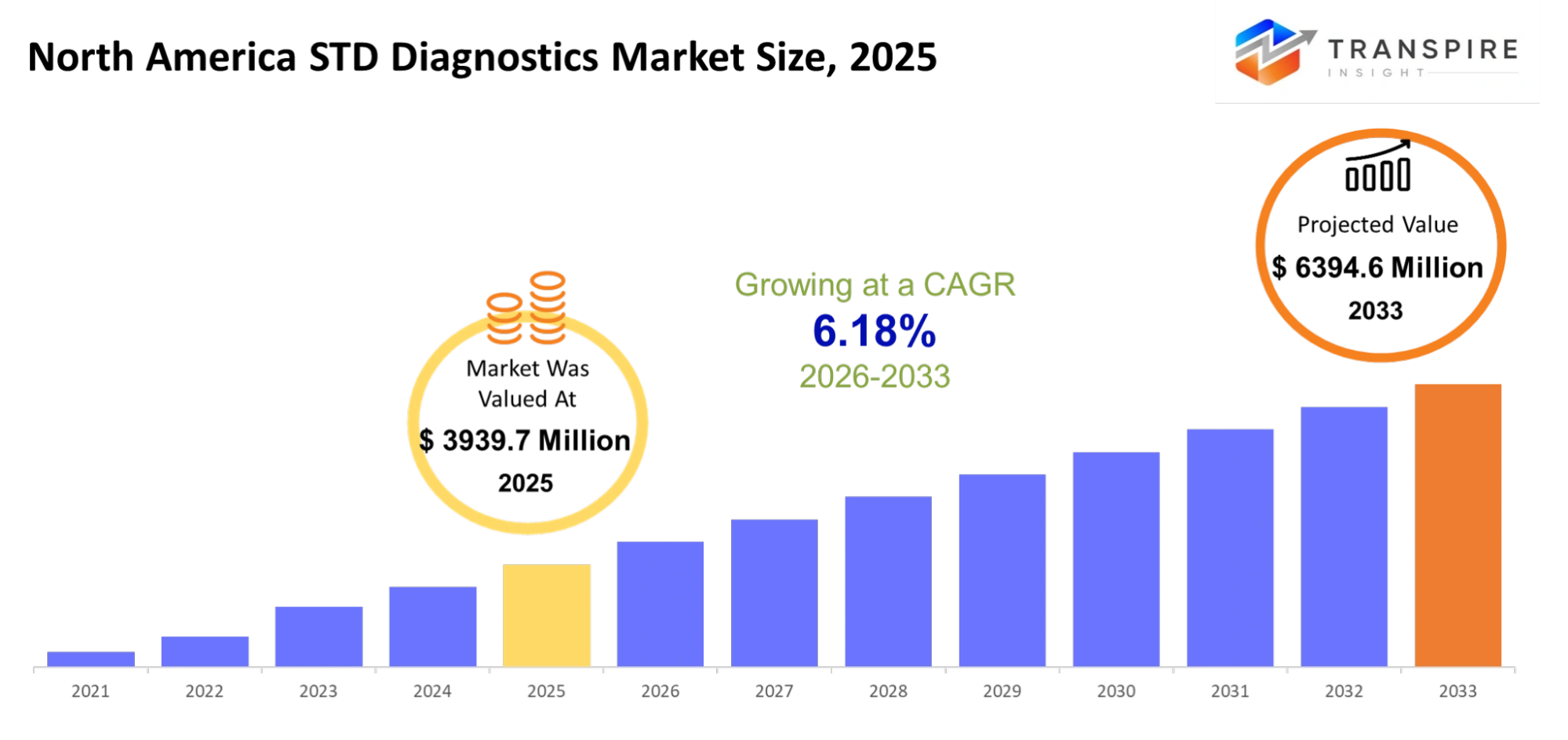
- North America STD Diagnostics Market Size 2025: USD 3939.7 Million
- North America STD Diagnostics Market Size 2033: USD 6394.6 Million
- North America STD Diagnostics Market CAGR: 6.18%
- North America STD Diagnostics Market Segments: By Test (Lab, POC), By Disease (HIV, Chlamydia), By End-User (Hospitals)
To learn more about this report, Download Free Sample Report
North America STD Diagnostics Market Summary:
The North America STD Diagnostics Market size is estimated at USD 3939.7 Million in 2025 and is anticipated to reach USD 6394.6 Million by 2033, growing at a CAGR of 6.18% from 2026 to 2033. The STD diagnostics market in North America which includes Canada and the United States and Mexico operates as a part of the larger healthcare diagnostics industry. People will demand more testing options which provide quick results and maintain their privacy and allow them to test at home. Laboratories and clinics will use molecular testing and point-of-care devices which deliver accurate results within minutes. Public health agencies will implement more strict screening requirements which will promote regular testing among all population groups. Digital health platforms will connect diagnostic testing with telemedicine consultations to enable faster medical decision-making. Testing services in the region will be affected by two factors which include insurance coverage patterns and public awareness initiatives.
Key Market Trends & Insights:
The increasing demand for testing which provides privacy and requires minimal effort will create new service delivery methods throughout North America. People will prefer to use self-collection kits and pharmacy-based testing methods because they want to avoid the social stigma connected with visiting medical facilities. The shift in these patterns will lead diagnostic companies to develop solutions which include packaging systems and delivery methods and simple user guides. The healthcare systems of the future will implement remote sample processing methods that provide accurate results while protecting patient privacy during their screening process.
Further development of technology will make the process even more precise and quicker. The health care industry will adopt these novel molecular devices due to their ability to detect diseases in their early stages. Automation in laboratories will reduce human error and improve throughput, especially in urban centers. Portable diagnostic devices will become more common in community clinics and outreach programs which will help improve screening access for underserved communities throughout Canada and the United States and Mexico.
The development of regulatory frameworks will permit faster product approvals while enforcing complete quality control procedures. The authorities will place greater importance on confirming the accuracy of at-home testing kits and digital reporting systems. Cross-border collaboration may strengthen data sharing on infection trends which helps governments to respond more effectively. The compliance requirements will compel manufacturers to provide clearer product performance information which will affect procurement choices made by hospitals and diagnostic networks across the region.
Public health priorities will determine the public's needs which will change as people learn more about health conditions. Public education efforts will lead to increased screening rates among all young people and those who are at risk of health problems. Private companies will work together with government agencies to develop new programs which will assist people who live in rural areas and semi-urban communities. People will choose to get tested more frequently because decreased stigma creates easier testing access which results in improved health outcomes.
North America STD Diagnostics Market Segmentation
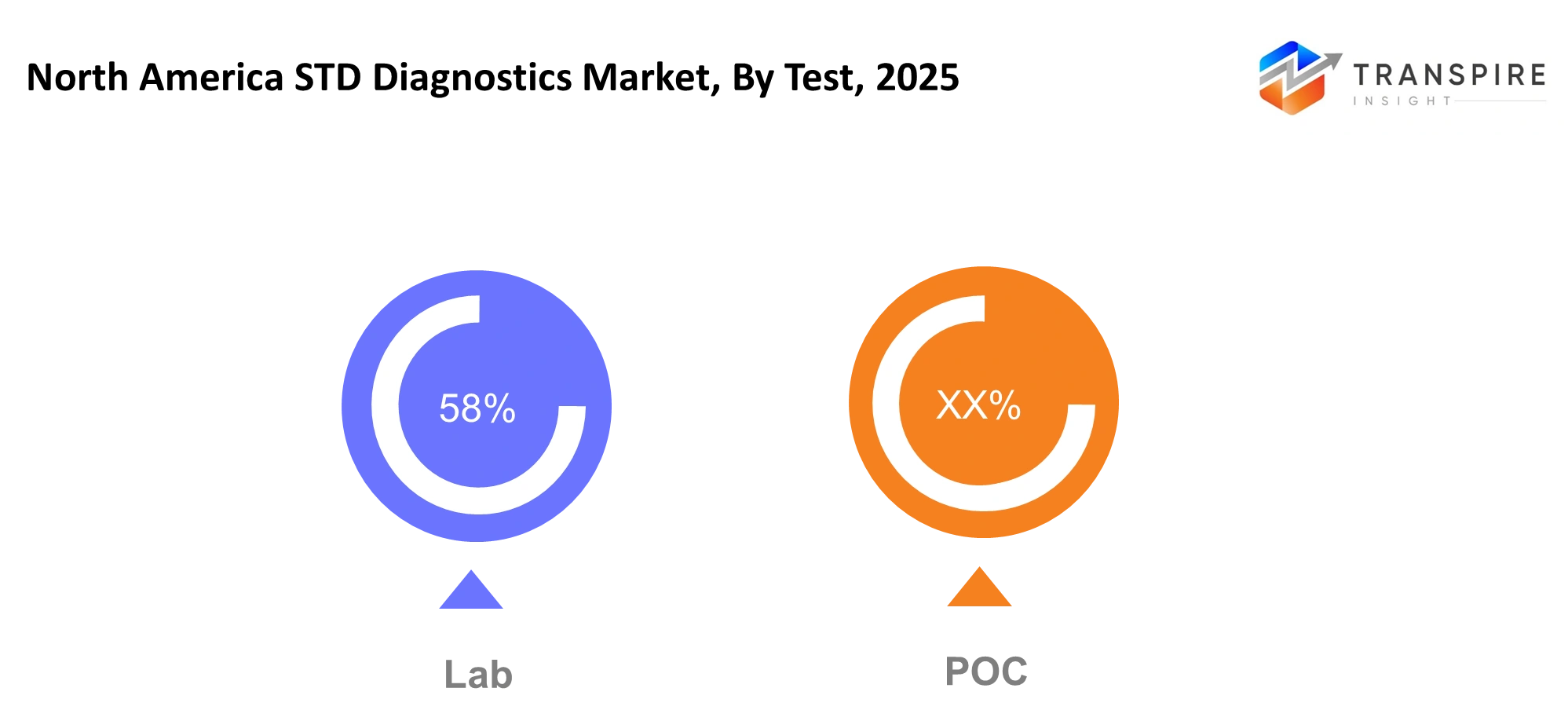
By Test
Lab- The laboratory testing process will maintain its position as the primary testing method because it produces results which are both valid and dependable. Advanced molecular diagnostics machines and automatic analyzers will be used to process samples in large numbers in order to ensure consistency in operations. Hospitals and laboratories will begin adopting standardized procedures that guarantee reduced turnaround time. The trust in laboratory services will continue because they provide complex case testing and follow-up evaluations and surveillance programs which need precise clinical validation.
POC- POC (Point-of-Care) testing will experience growth because healthcare providers want to reduce the time required for diagnosis and treatment. The solutions will be implemented in clinics and mobile units and community outreach locations to provide immediate results without the need for advanced infrastructure. Patients will receive same-visit counseling and treatment decisions which will minimize their chances of losing to follow-up. The combination of improved device sensitivity and portability will make POC testing feasible especially in regions that experience laboratory access problems or testing delays.
To learn more about this report, Download Free Sample Report
By Disease
HIV- The main focus of HIV diagnostics will remain on finding new cases of the disease and conducting tests at regular intervals. The testing methods will expand their reach to conduct tests at both community centers and residential locations which will lead to more frequent testing. The use of fourth-generation tests and rapid test kits will enable better identification of infected individuals during their initial stages of sickness. Public health programs will make sure that high-risk groups receive continuous screenings while digital reporting systems will help track disease cases. The combined efforts of these activities will enhance population control measures while decreasing disease spread throughout communities.
Chlamydia- Chlamydia testing will focus on early identification, particularly among younger and sexually active individuals. The healthcare system will implement screening programs through health facilities and reproductive health services because most people with infections do not show any symptoms. Nucleic acid amplification tests will remain widely used due to their accuracy. The awareness campaigns will drive people to undergo testing while self-collection methods for screening will improve accessibility which will help lower the risk of long-term health issues that result from untreated infections.
By End-User
Hospitals- Hospitals will function as essential facilities which provide both standard medical tests and advanced diagnostic services. The facility will manage a large number of patients while delivering comprehensive medical services that merge testing procedures with on-site medical assessment and therapeutic interventions. The advanced laboratory systems in hospitals will enable precise identification of various medical conditions. The hospitals will work together with public health officials to document medical cases and watch disease patterns which will help them manage infection outbreaks while enhancing their patient care results.
Regional Insights
North American healthcare delivery systems will vary according to the development level of their infrastructure and the number of people living in each area. The United States will lead in adopting advanced diagnostic tools, supported by private healthcare investments and insurance-driven access. Canada will implement publicly funded screening programs which will use standardized protocols to achieve equal access for all citizens. Mexico will develop its diagnostic capabilities through urban healthcare facilities, while rural communities will depend on their local clinics and outreach programs.
The region will witness an uninterrupted trend toward consumers’ adoption of tests that focus on their privacy needs and provide reliable test results with maximum convenience. The US market will witness a sharp rise in the uptake of home testing kits and telemedicine. Users in Canada will find themselves balancing between utilizing the government-run health care system and the newly developed private system, which offers fast access to services. Mexico will see greater use of awareness programs that encourage frequent testing among its young population.
Technology adoption will proceed through three countries because it will show different patterns of progress through time. The United States will implement artificial intelligence and automation technologies to improve its laboratory operations. Canada will establish healthcare system interoperability which enables hospitals to share patient information without complications. Mexico will invest in affordable diagnostic solutions which can be implemented throughout its metropolitan and suburban areas. Portable testing devices will become more common throughout all areas because they will help outreach programs and increase early detection testing success.
The development of diagnostic services in each region will depend on existing regulatory and policy frameworks. The United States will balance its innovation activities with testing solution approvals because it wants to achieve progress at a faster rate. Canada will continue developing its national guidelines which help people access preventive screening services. Mexico will improve its regulatory framework by working with private providers for better industry oversight. The United States and Canada will work together to improve their data sharing systems which will enable them to better track infectious disease patterns.
Recent Development News
In early 2026, regulatory authorities approved advanced at-home diagnostic kits capable of detecting multiple infections with high accuracy, supported by integrated telehealth services for treatment guidance. At the same time, public health infrastructure has faced setbacks, including the closure of a key federal STI research laboratory, raising concerns about long-term surveillance and diagnostic capacity.
In 2025, Canada has moved forward with regulatory approvals for high-performance rapid diagnostic tools, strengthening access to timely screening. Health authorities approved new point-of-care HIV tests with strong sensitivity and specificity, allowing broader use across clinical and outreach settings. These advancements are being aligned with publicly funded healthcare systems, where consistent screening guidelines and nationwide programs will support early detection and improve patient engagement in both urban and remote communities.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 3939.7 Million |
|
Market size value in 2026 |
USD 4201.7 Million |
|
Revenue forecast in 2033 |
USD 6394.6 Million |
|
Growth rate |
CAGR of 6.18% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
North America (Canada, The United States, and Mexico) |
|
Key company profiled |
Abbott, Roche, Siemens, BD, Thermo Fisher, Hologic, Bio-Rad, Danaher, Qiagen, Cepheid, OraSure, PerkinElmer, Trinity Biotech, Fujirebio, Labcorp |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Test (Lab, POC), By Disease (HIV, Chlamydia), By End-User (Hospitals) |
Key North America STD Diagnostics Market Company Insights
The major North American diagnostic enterprises will create their market advantages through their process of developing new products and expanding their existing product lines. The companies Abbott Roche BD Hologic and QIAGEN will develop integrated healthcare solutions through their research on laboratory systems and rapid testing technologies. The research funding of the organization will enhance its molecular testing capabilities through automated systems and digital system connections which will lead to improved diagnostic speed and accuracy. Organizations will develop easy-to-use self-testing products and expandable testing systems which will help them maintain their market position as healthcare services move to decentralized patient-focused testing systems throughout the region.
Company List
- Abbott
- Roche
- Siemens
- BD
- Thermo Fisher
- Hologic
- Bio-Rad
- Danaher
- Qiagen
- Cepheid
- OraSure
- PerkinElmer
- Trinity Biotech
- Fujirebio
- Labcorp
North America STD Diagnostics Market Report Segmentation
By Test
- Lab
- POC
By Disease
- HIV
- Chlamydia
By End-User
- Hospitals
Frequently Asked Questions
Find quick answers to common questions.
The approximate North America STD Diagnostics Market size for the market will be USD 6394.6 Million in 2033.
Key segments for the North America STD Diagnostics Market By Test (Lab, POC), By Disease (HIV, Chlamydia), By End-User (Hospitals).
Major players in the North America STD Diagnostics Market are Abbott, Roche, Siemens, BD, Thermo Fisher, Hologic, Bio-Rad, Danaher, Qiagen, Cepheid, OraSure, PerkinElmer, Trinity Biotech, Fujirebio, Labcorp.
The North America STD Diagnostics Market size is USD 3939.7 Million in 2025.
The North America STD Diagnostics Market CAGR is 6.18%.
- Abbott
- Roche
- Siemens
- BD
- Thermo Fisher
- Hologic
- Bio-Rad
- Danaher
- Qiagen
- Cepheid
- OraSure
- PerkinElmer
- Trinity Biotech
- Fujirebio
- Labcorp
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
May 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)