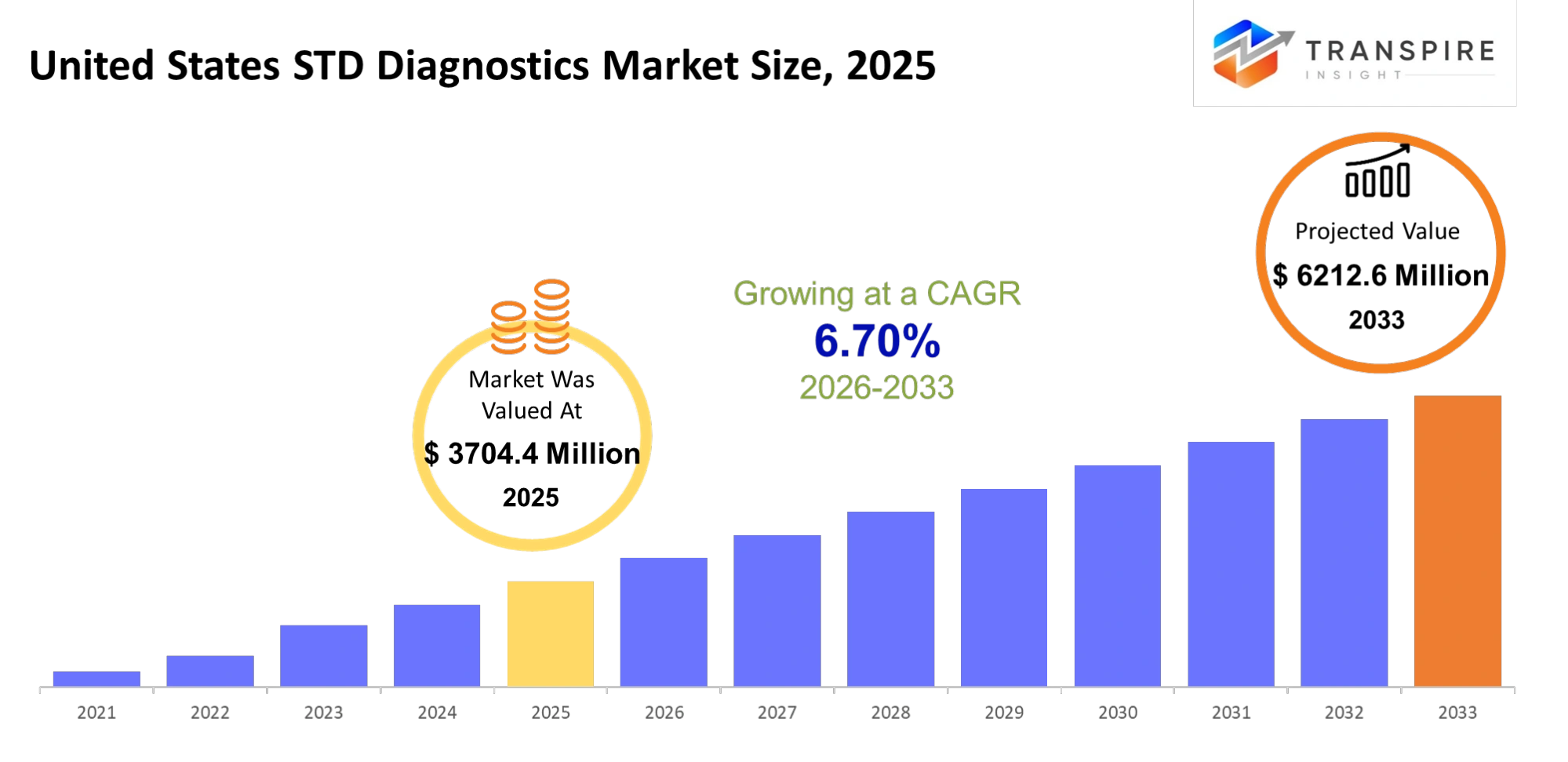
United States STD Diagnostics Market Size & Forecast:
- United States STD Diagnostics Market Size 2025: USD 3704.4 Million
- United States STD Diagnostics Market Size 2033: USD 6212.6 Million
- United States STD Diagnostics Market CAGR: 6.70%
- United States STD Diagnostics Market Segments:By Test Type (Immunoassays, Molecular Diagnostics, Rapid Diagnostic Tests), By Disease Type (Chlamydia, Gonorrhea, Syphilis, HPV, HIV), By End User (Hospitals, Diagnostic Laboratories, Clinics).
To learn more about this report, Download Free Sample Report
United States STD Diagnostics Market Summary:
The United States STD Diagnostics Market size is estimated at USD 3704.4 million in 2025 and is anticipated to reach USD 6212.6 million by 2033, growing at a CAGR of 6.70% from 2026 to 2033. The United States STD diagnostics Market is undergoing rapid changes because of increasing public awareness and better screening programmes and new testing technologies. People now prefer home testing solutions that provide fast results because more people understand the importance of early disease detection and preventive medical methods. Government programmes, together with public health campaigns, work to establish routine testing as a standard practice, which helps decrease the social stigma associated with sexually transmitted diseases. The introduction of new molecular diagnostic technologies and point-of-care testing systems has improved STD diagnostic processes by making them easier to access and more dependable for patients in various medical environments.
Key Market Trends & Insights:
- The growing public understanding of sexual health issues leads people to choose regular health checkups. The practice of testing becomes customary because early diagnosis prevents complications while decreasing transmission rates, which affect all age demographics.
- Consumers show a growing preference for testing methods that provide both privacy and ease of use. People choose at-home STD test kits because they want to maintain their private life while testing becomes simple, and they can receive test results without going to medical facilities.
- The current tests use PCR-based technology, which enhances both testing speed and testing precision. The new technologies enable healthcare professionals to identify diseases at earlier stages while achieving higher accuracy rates, which result in better patient care.
- The demand for quick diagnostic tools which provide results within minutes is experiencing growth. The tools assist with making quick clinical choices, which proves essential in both emergency medical facilities and public health centres.
- The combination of increased funding and nationwide awareness campaigns leads to higher public knowledge about STDs and their testing procedures. The programmes work to decrease social stigma, which leads more people to get immediate health assessments.
- Medical diagnostic testing services now extend through telehealth platforms and mobile applications. The digital system enables patients to interact with doctors and request tests and receive their results throughout the whole testing procedure.
- The program now implements testing systems which specifically assist at-risk populations. The method provides improved methods for disease treatment and helps prevent disease outbreaks within designated neighbourhoods.
United States STD Diagnostics Market Segmentation
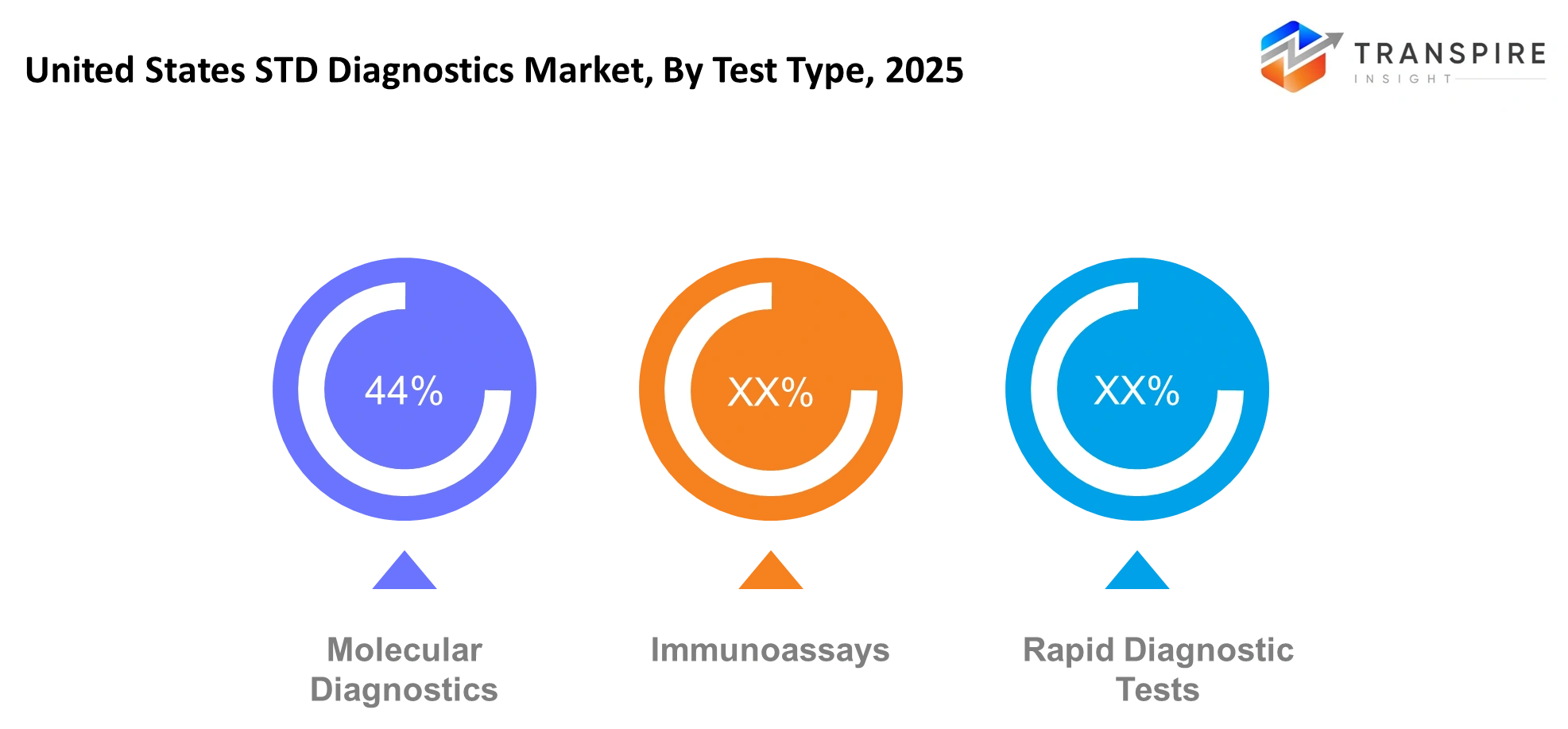
By Test Type
- Immunoassays: Immunoassays remain widely used because they provide affordable solutions that accurately detect both antibodies and antigens. The testing method functions as a standard tool which health facilities implement to evaluate patients in their daily operations. Healthcare laboratories across the United States select this testing method because it offers simple operation and automated system compatibility.
- Molecular Diagnostics: Molecular diagnostics enable STD detection through their capability to identify infections at extremely low pathogen levels. The PCR method enables healthcare professionals to detect infections during their early stages. Advanced laboratories increasingly adopt these tests because they deliver rapid results which help doctors make accurate treatment decisions in contemporary medical environments.
- Rapid Diagnostic Tests: Rapid diagnostic tests have become more popular because they provide quick results and require minimal effort to use. The tests deliver results within minutes, which makes them suitable for usage in point-of-care environments. The tests enable rapid diagnosis and emergency patient treatment, which benefits both first responders and medical professionals working in distant locations.
To learn more about this report, Download Free Sample Report
By Disease Type
- Chlamydia: The Market for chlamydia testing includes a substantial section because chlamydia exists as an extremely common disease which frequently shows no symptoms. The medical community recommends regular screening as essential for all individuals but especially for young adults. The need for accurate and easily available diagnostic tests exists because early diagnosis helps to stop future medical problems.
- Gonorrhea: Gonorrhea diagnostics are experiencing ongoing growth because the number of reported cases continues to increase. Health professionals need to perform accurate and timely tests because these results help fight the problem of developing antibiotic resistance. Medical facilities now use advanced testing technologies which provide accurate results to doctors so they can treat patients and stop the infection from spreading.
- Syphilis: Syphilis testing needs to remain available because untreated syphilis can lead to severe health problems. The combination of increased awareness and prenatal screening programmes, which now operate as standard practice, has resulted in higher testing rates. The modern diagnostic methods enable medical professionals to identify diseases at multiple stages, which results in timely treatment and improved patient results.
- HPV (Human Papillomavirus): HPV testing capabilities are expanding through funding from cervical cancer screening initiatives. The process of regular testing, which women should especially do, will help them detect health issues early and stop their progression. The system now helps people learn about their health through direct health assessments, which lead them to take better care of themselves.
- HIV (Human Immunodeficiency Virus): HIV testing needs to remain a primary goal for STD testing procedures. Public testing centres and educational campaigns about testing create an environment which encourages people to undergo regular health examinations. People now have better access to their health status because of improvements in rapid testing and self-testing methods, which also help them obtain medical assistance.
By End User
- Hospitals: Hospitals function as essential institutions for STD diagnosis because they provide complete testing services and treatment options. The facility contains modern diagnostic equipment which trained staff members operate. Patients prefer hospitals because they provide precise medical assessments which doctors need for diagnosing difficult medical situations.
- Diagnostic Laboratories: Diagnostic laboratories function as the primary facility for large-scale STD testing because they conduct tests with exceptional accuracy. The laboratory uses modern technology together with automated processes to achieve dependable testing outcomes. The organisation provides essential support to healthcare professionals by delivering precise information which enables them to diagnose patients and create treatment strategies.
- Clinics: Clinics offer STD testing services which people can access and use anytime because they need regular tests. Patients who want fast medical treatment typically go to these facilities as their initial contact point. Clinics expand their testing abilities because more people understand their services, which enables them to deliver efficient, non-invasive testing options to patients.
Country Insights
The current healthcare system in the United States shows active changes because of rising STD rates, effective public health campaigns, and ongoing development of new testing methods. The country benefits from a well-established healthcare infrastructure which provides people in both cities and rural areas access to advanced diagnostic services. The implementation of awareness campaigns together with government-funded screening initiatives has successfully encouraged people to undergo testing, which resulted in decreased stigma and improved identification of conditions at an early stage.
The increasing use of at-home testing kits together with digital health platforms is changing the way people manage their sexual well-being by providing them with better options for private and easy-to-use testing methods. The Market gains strength from its combination of top diagnostic companies and active research initiatives, which create advancements in developing faster and more precise testing methods. The United States remains committed to its focus on preventive medical care together with early disease identification, which makes STD diagnostic testing essential for its public health initiatives.
Recent Development News
At-Home STD Testing Expands Across the U.S.: At-home testing solutions are gaining momentum as healthcare providers push for wider screening access. New FDA-approved kits are making STD diagnostics more private and convenient for patients.
Rise of App-Connected STD Diagnostic Tools: New STD tests are integrating mobile apps to deliver results and guidance. This digital shift is enhancing patient engagement and follow-up care.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 3704.4 Million |
|
Market size value in 2026 |
USD 3945.3 Million |
|
Revenue forecast in 2033 |
USD 6212.6 Million |
|
Growth rate |
CAGR of 6.70% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
United States |
|
Key company profiled |
Abbott Laboratories, Roche Diagnostics, Becton Dickinson and Company, Hologic Inc., Thermo Fisher Scientific Inc., Siemens Healthineers AG, Danaher Corporation, Bio-Rad Laboratories Inc., Qiagen NV, Cepheid Inc., Trinity Biotech PLC, OraSure Technologies Inc., Sekisui Diagnostics LLC, Fujirebio Diagnostics Inc., Meridian Bioscience Inc. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Test Type (Immunoassays, Molecular Diagnostics, Rapid Diagnostic Tests), By Disease Type (Chlamydia, Gonorrhea, Syphilis, HPV, HIV), By End User (Hospitals, Diagnostic Laboratories, Clinics). |
Key United States STD Diagnostics Company Insights
The United States STD diagnostics Market exists because established healthcare companies and diagnostic companies operate their businesses in the United States while developing new testing techniques. The companies develop their advanced testing solutions because they need to fulfil the rising Market demand for their products, which include molecular diagnostics and rapid test kits. The companies pursue Market expansion through research investments, product launches and strategic partnerships. Market players are developing simple testing solutions that people can use at home to meet the growing demand for private medical tests.
Company List
- Abbott Laboratories
- Roche Diagnostics
- Becton Dickinson and Company
- Hologic Inc.
- Thermo Fisher Scientific Inc.
- Siemens Healthineers AG
- Danaher Corporation
- Bio-Rad Laboratories Inc.
- Qiagen NV
- Cepheid Inc.
- Trinity Biotech PLC
- OraSure Technologies Inc.
- Sekisui Diagnostics LLC
- Fujirebio Diagnostics Inc.
- Meridian Bioscience Inc.
United States STD Diagnostics Market Report Segmentation
By Test Type
- Immunoassays
- Molecular Diagnostics
- Rapid Diagnostic Tests
By Disease Type
- Chlamydia
- Gonorrhea
- Syphilis
- HPV
- HIV
By End User
- Hospitals
- Diagnostic Laboratories
- Clinics
Frequently Asked Questions
Find quick answers to common questions.
The Approximate United States STD Diagnostics Market size for the Market will be USD 6212.6 Million in 2033.
The key Segments of the United States STD Diagnostics Market are By Test Type (Immunoassays, Molecular Diagnostics, Rapid Diagnostic Tests), By Disease Type (Chlamydia, Gonorrhea, Syphilis, HPV, HIV), By End User (Hospitals, Diagnostic Laboratories, Clinics).
Major Players in the United States STD Diagnostics Market are Abbott Laboratories, Roche Diagnostics, Becton Dickinson and Company, Hologic Inc., Thermo Fisher Scientific Inc., Siemens Healthineers AG, Danaher Corporation, Bio-Rad Laboratories Inc., Qiagen NV, Cepheid Inc., Trinity Biotech PLC, OraSure Technologies Inc., Sekisui Diagnostics LLC, Fujirebio Diagnostics Inc., Meridian Bioscience Inc.
The Current Market size of the United States STD Diagnostics Market is USD 3704.4 Million in 2025.
The United States STD Diagnostics Market CAGR is 6.70%.
- Abbott Laboratories
- Roche Diagnostics
- Becton Dickinson and Company
- Hologic Inc.
- Thermo Fisher Scientific Inc.
- Siemens Healthineers AG
- Danaher Corporation
- Bio-Rad Laboratories Inc.
- Qiagen NV
- Cepheid Inc.
- Trinity Biotech PLC
- OraSure Technologies Inc.
- Sekisui Diagnostics LLC
- Fujirebio Diagnostics Inc.
- Meridian Bioscience Inc.
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


