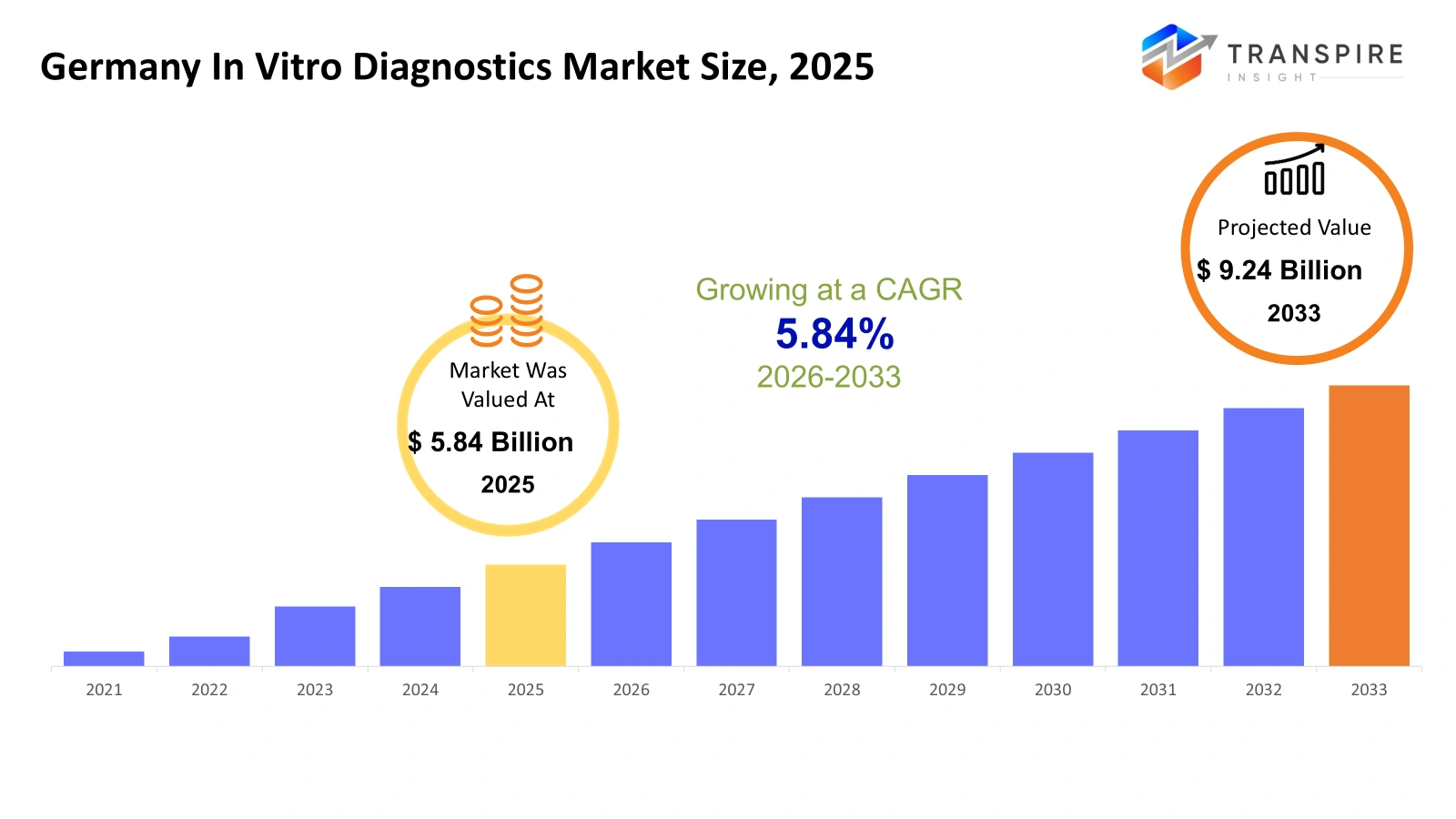
Germany In Vitro Diagnostics Market Size & Forecast:
- Germany In Vitro Diagnostics Market Size 2025: USD 5.84 Billion
- Germany In Vitro Diagnostics Market Size 2033: USD 9.24 Billion
- Germany In Vitro Diagnostics Market CAGR: 5.84%
- Germany In Vitro Diagnostics Market Segments: By Product Type (Reagents & Kits, Instruments, Software & Services), By Technology (Immunoassay, Molecular Diagnostics, Clinical Chemistry), By End User (Hospitals, Diagnostic Laboratories, Research Institutes)
To learn more about this report, Download Free Sample Report
Germany In Vitro Diagnostics Market Summary:
The Germany In Vitro Diagnostics Market size was estimated at USD 5.84 B billion in 2025 and is anticipated to reach USD 9.24 Billion by 2033, growing at a CAGR of 5.84 % from 2026 to 2033. The healthcare technology sector in Germany will develop its in vitro diagnostics market to enhance diagnostic procedures used by hospitals and laboratories and outpatient medical facilities. The need for quicker and more accurate test outcomes will shape both product development efforts and laboratory spending decisions. Healthcare providers will begin using automated analyzers and digital data systems and molecular testing platforms to enhance their capabilities for early disease identification and ongoing patient observation. The increased public understanding of preventive health assessments will determine how patients choose to undergo testing. The German regulatory authorities will continue to improve quality standards and data transparency regulations which will require manufacturers to change their product design and documentation and validation methods to meet compliance standards and deliver trustworthy diagnostic results.
Key Market Trends & Insights:
- The Germany in vitro diagnostics Market shows two main trends because its laboratory facilities and healthcare systems enable comprehensive diagnostic testing throughout Germany. The demand for early disease detection and infection monitoring along with routine screening procedures leads to ongoing development of diagnostic technologies. The Germany in vitro diagnostics Market experiences growth because hospitals and laboratories increasingly use automated analyzers and molecular testing platforms.
- The Germany in vitro diagnostics Market shows two key trends because preventive healthcare practices lead to increased diagnostic testing at medical facilities. Diagnostic kits and laboratory reagents face constant demand because screening programs for chronic conditions and infectious diseases need them. The Germany in vitro diagnostics Market expands because testing accuracy improvements and faster result delivery methods continue to advance.
- The Germany in vitro diagnostics Market shows two key trends because molecular diagnostics and genetic testing technologies expand their use in clinical laboratories to identify advanced disease cases. Diagnostic laboratories use polymerase chain reaction testing and genomic analysis to identify infections and hereditary conditions with high precision. The Germany in vitro diagnostics Market experiences research growth and commercial development because technological advancements in molecular testing lead to improved research methods.
- The testing volume at hospitals and private laboratories increases because both the elderly population and the increasing number of chronic diseases require diagnostic testing. The medical field requires more advanced blood testing systems, immunoassay technologies and disease monitoring systems, which leads to increased funding for contemporary diagnostic equipment. The German in vitro diagnostics market remains active because testing requirements continue to increase.
- The research collaboration between biotechnology companies, diagnostic manufacturers and medical research institutions drives laboratory testing technology development. The implementation of new biomarker testing systems together with advanced screening techniques results in better diagnostic results. The German in vitro diagnostics market progresses through continuous research which drives technology development.
Germany In Vitro Diagnostics Market Segmentation
By Product Type
Reagents and kits: The testing materials used in laboratories require continuous delivery of chemical substances and testing resources and ready-to-use diagnostic materials which makes reagents and kits essential for the German in vitro diagnostic market. Daily operations in hospitals and diagnostic labs depend on reagents and kits which they use to identify diseases and assess blood samples and test for infections. The ongoing need for testing will maintain constant product usage throughout this period.
Instruments: The primary operational component of the Germany in vitro diagnostics market depends on diagnostic systems which execute sample analysis and measurement tasks and automatic testing functions. The combination of laboratory analyzers and molecular testing machines and biochemical analysis devices produces precise diagnostic results. Medical facilities purchase modern equipment to boost testing efficiency while upholding testing accuracy and managing high testing demand.
Software and devices: The Germany in vitro diagnostics market operates through software and services which assist laboratories with their data management operations and workflow processes and diagnostic reporting system administration. Diagnostic centers use laboratory information management systems with digital reporting tools and maintenance services to achieve their operational goals. The expansion of digital healthcare infrastructure will drive up requirements for both diagnostic software integration and technical service assistance.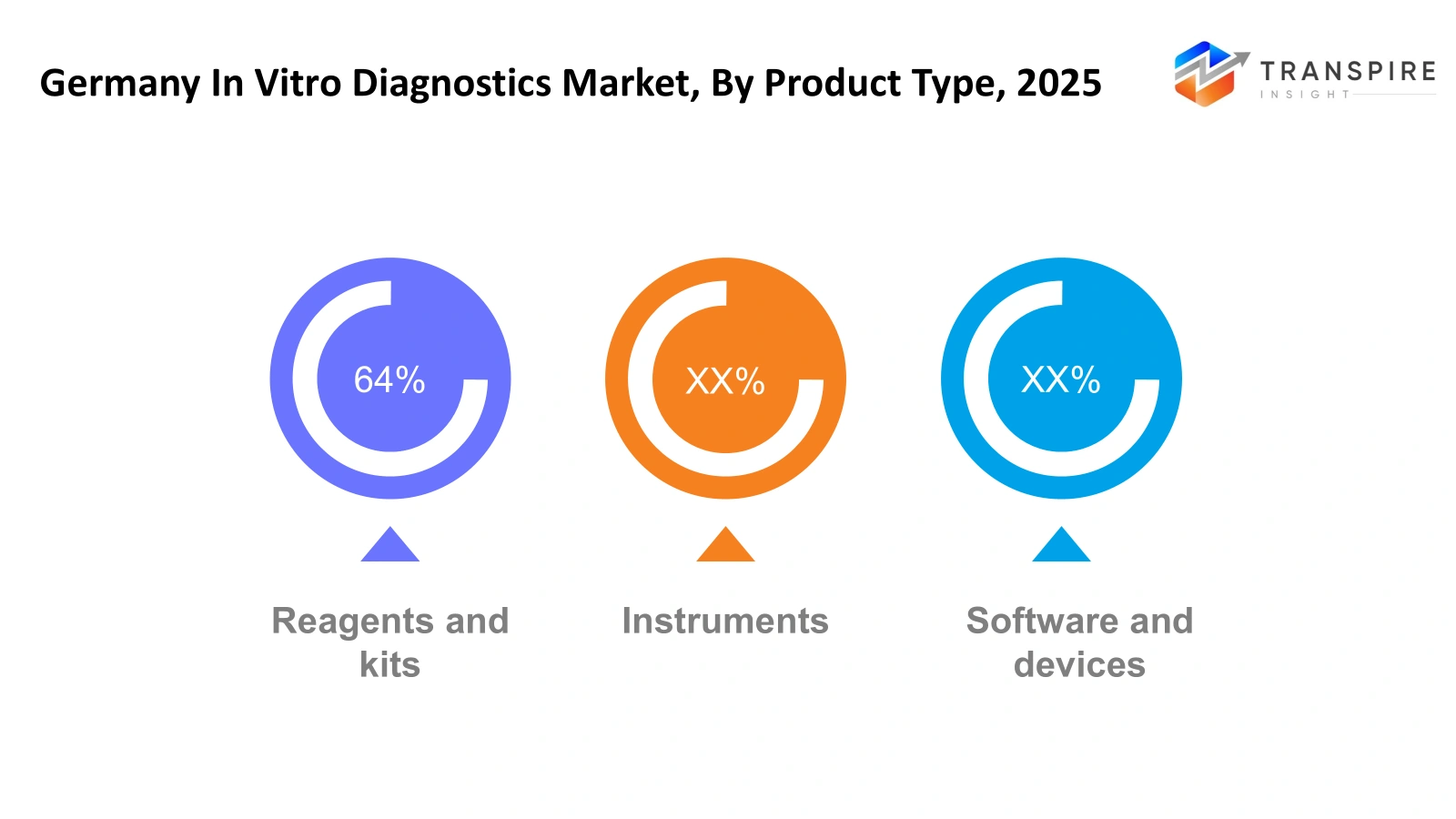
To learn more about this report, Download Free Sample Report
By Technology
Immunoassay: The German in vitro diagnostics market depends on immunoassay technology because it enables testing which detects hormonal levels and infectious agents and specific cancer indicators. Diagnostic laboratories use antigen and antibody reactions to find disease indicators present in blood samples. The medical field requires immunoassay testing solutions because they deliver dependable outcomes which can be used in various clinical settings.
Molecular Diagnostics: Molecular diagnostics technology supports disease detection through analysis of genetic material such as DNA and RNA in Germany in vitro diagnostics Market. Molecular testing enables early identification of infections, inherited disorders, and certain cancers. The medical field uses molecular diagnostic systems because they provide accurate genetic identification which enhances treatment planning and disease management.
Clinical Chemistry: Clinical chemistry technology supports routine laboratory analysis in Germany in vitro diagnostics Market by measuring chemical components in blood, urine, and other biological samples. Clinical chemistry analyzers help detect conditions such as diabetes, kidney disorders, and liver disease. The high testing demand from hospitals and diagnostic laboratories creates continuous market needs for clinical chemistry systems.
By End User
Hospitals: The in vitro diagnostics market in Germany considers hospitals as its main service provider because hospital laboratories conduct diagnostic tests for their patients who stay overnight and for their emergency and outpatient cases. The hospital diagnostic units need dependable testing equipment together with their necessary reagents and testing systems to provide medical professionals with accurate results. The number of patients requiring tests will keep increasing which will create higher demand for diagnostic services at hospitals.
Diagnostic laboratories: The in vitro diagnostics market in Germany depends on diagnostic laboratories because these facilities provide both basic and complex testing services through their specialized equipment. Independent diagnostic centers support physicians, clinics, and hospitals by delivering accurate laboratory reports. The demand for laboratory testing services will continue to grow because people increasingly recognize the importance of preventive healthcare.
Research Institutes: Research institutes in Germany develop new diagnostic methods through their medical research and laboratory innovation work which supports their role in the in vitro diagnostics market. Academic laboratories and medical research organizations study disease patterns, evaluate new diagnostic tools, and support healthcare advancements. The current research activities will drive development of new diagnostic technologies that will benefit the entire healthcare system in the country.
Country Insights
The demand for diagnostic testing across Germany benefits from Germany's strong healthcare system and advanced laboratory network. The clinical testing technologies used by hospitals and diagnostic laboratories and research institutes remain operational for both disease detection and patient monitoring purposes. The Germany in vitro diagnostics Market will achieve further development through the expansion of molecular testing and immunoassay systems and automated laboratory equipment.
The field of diagnostic innovation in Germany is experiencing strong growth through partnerships between medical research institutions and biotechnology companies. The combination of government health programs and aging population trends and increasing chronic disease cases drives up demand for diagnostic testing in hospitals and private laboratories. The ongoing process of laboratory modernization together with the implementation of advanced diagnostic platforms will drive development within the Germany in vitro diagnostics Market.
Recent Development News
Strong activity in diagnostic technology continues across Germany with increasing focus on faster laboratory testing and advanced molecular diagnostics. Investment in automated testing equipment, digital laboratory systems, and biomarker research will support clinical laboratories and hospitals. Technology progress continues to strengthen innovation across the Germany in vitro diagnostics Market.
Expansion of diagnostic manufacturing and research activity continues across the European healthcare industry. The European Commission highlights regulatory implementation of the In Vitro Diagnostic Regulation (IVDR), which strengthens safety and quality requirements for diagnostic devices across member states including Germany.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 5.84 Billion |
|
Market size value in 2026 |
USD 6.21 Billion |
|
Revenue forecast in 2033 |
USD 9.24 Billion |
|
Growth rate |
CAGR of 5.84% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Germany |
|
Key company profiled |
Roche Diagnostics, Siemens Healthineers, Abbott Laboratories, Thermo Fisher Scientific, Danaher Corporation, Becton Dickinson, Bio-Rad Laboratories, Qiagen NV, Sysmex Corporation, Hologic Inc., Agilent Technologies, Ortho Clinical Diagnostics, PerkinElmer Inc., Illumina Inc., Mindray Medical. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Product Type (Reagents & Kits, Instruments, Software & Services), By Technology (Immunoassay, Molecular Diagnostics, Clinical Chemistry), By End User (Hospitals, Diagnostic Laboratories, Research Institutes). |
Key Germany In Vitro Diagnostics Market Company Insights
The data available to you includes information that extends until the month of October in the year 2023. The German healthcare laboratories achieve continuous technological progress because both international and local diagnostic manufacturers actively participate in the market. The companies develop molecular diagnostics systems, automated testing platforms, and laboratory information systems to achieve higher testing accuracy and faster test results. The product development activities together with research partnerships create new growth opportunities for the Germany in vitro diagnostics Market.
The diagnostic companies operating in Germany expand their operations through research partnerships and laboratory modernization programs, and product launches, which improve disease detection and clinical testing efficiency. The German in vitro diagnostics Market will experience continuous commercial growth and industry innovation through the industrial focus on biomarker identification and rapid test kits, and advanced analyzers.
Company List
- Roche Diagnostics
- Siemens Healthineers
- Abbott Laboratories
- Thermo Fisher Scientific
- Danaher Corporation
- Becton Dickinson
- Bio-Rad Laboratories
- Qiagen NV
- Sysmex Corporation
- Hologic Inc.
- Agilent Technologies
- Ortho Clinical Diagnostics
- PerkinElmer Inc.
- Illumina Inc.
- Mindray Medical
Germany In Vitro Diagnostics Market Report Segmentation
By Product Type
- Reagents & Kits
- Instruments
- Software & Services
By Technology
- Immunoassay
- Molecular Diagnostics
- Clinical Chemistry
By End User
- Hospitals
- Diagnostic Laboratories
- Research Institutes
Frequently Asked Questions
Find quick answers to common questions.
The Approximate Germany In Vitro Diagnostics Market size will be USD 9.24 Billion in 2033.
The key Segments of the Germany In Vitro Diagnostics Market are By Product Type (Reagents & Kits, Instruments, Software & Services), By Technology (Immunoassay, Molecular Diagnostics, Clinical Chemistry), By End User (Hospitals, Diagnostic Laboratories, Research Institutes.
Major Players in the Germany In Vitro Diagnostics Market are Roche Diagnostics, Siemens Healthineers, Abbott Laboratories, Thermo Fisher Scientific, Danaher Corporation, Becton Dickinson, Bio-Rad Laboratories, Qiagen NV, Sysmex Corporation, Hologic Inc., Agilent Technologies, Ortho Clinical Diagnostics, PerkinElmer Inc., Illumina Inc., Mindray Medical.
The Current Market size of the Germany In Vitro Diagnostics Market is USD 5.84 Billion by 2025.
The Germany In Vitro Diagnostics Market CAGR is 5.84%.
- Roche Diagnostics
- Siemens Healthineers
- Abbott Laboratories
- Thermo Fisher Scientific
- Danaher Corporation
- Becton Dickinson
- Bio-Rad Laboratories
- Qiagen NV
- Sysmex Corporation
- Hologic Inc.
- Agilent Technologies
- Ortho Clinical Diagnostics
- PerkinElmer Inc.
- Illumina Inc.
- Mindray Medical
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


