Japan In Vitro Diagnostics Market Size & Forecast:
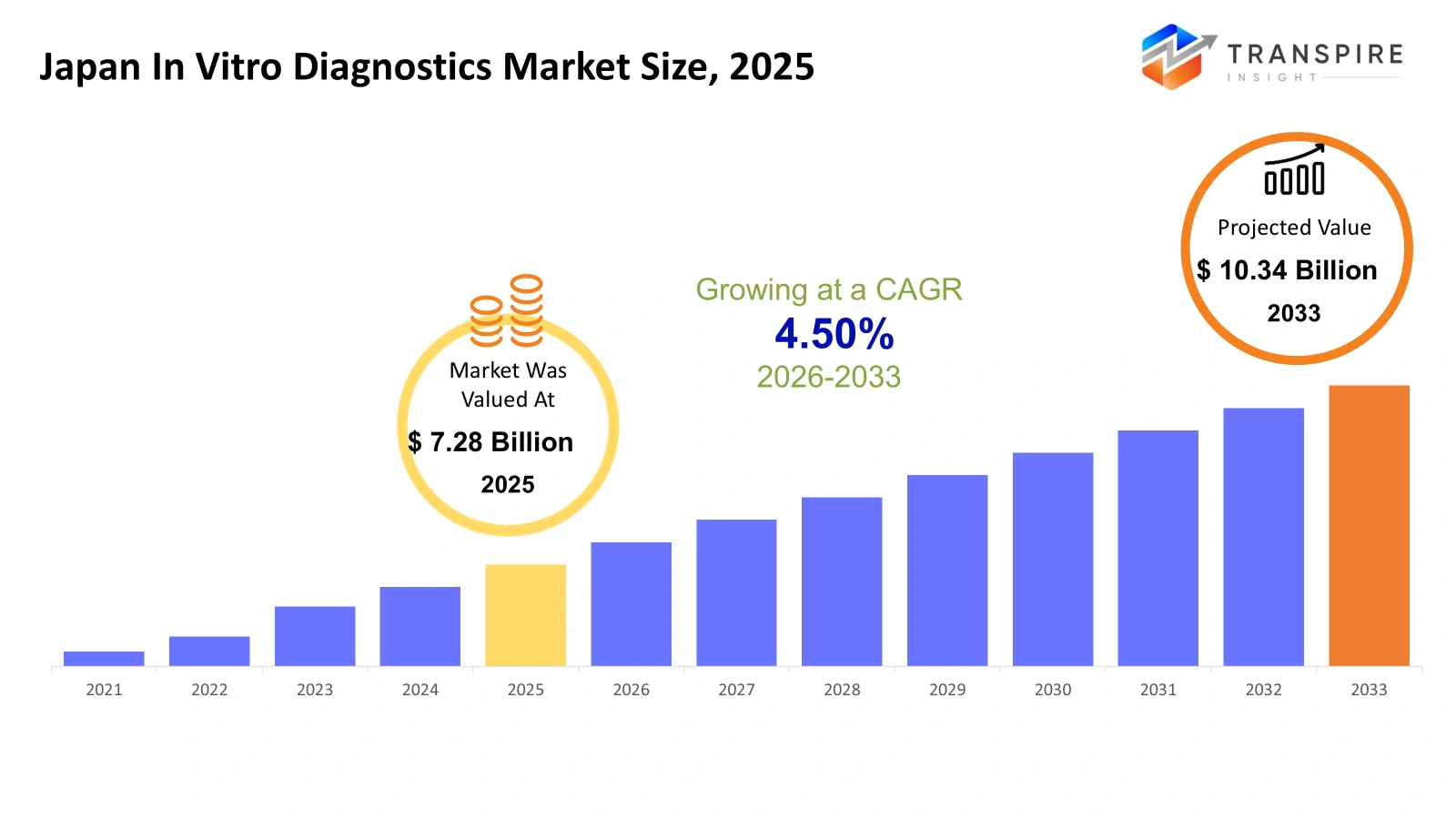
- Japan In Vitro Diagnostics Market Size 2025: USD 7.28 Billion
- Japan In Vitro Diagnostics Market Size 2033: USD 10.34 Billion
- Japan In Vitro Diagnostics Market CAGR: 4.50%
- Japan In Vitro Diagnostics Market Segments:By Product Type (Reagents & Kits, Instruments, Software & Services), By Technology (Immunoassay, Molecular Diagnostics, Clinical Chemistry), By End User (Hospitals, Diagnostic Laboratories).
To learn more about this report, Download Free Sample Report
Japan In Vitro Diagnostics Market Summary:
The Japan In Vitro Diagnostics Market size is estimated at USD 7.28 Billion in 2025 and is anticipated to reach USD 10.34 Billion by 2033, growing at a CAGR of 4.50% from 2026 to 2033. The Japan In Vitro Diagnostics (IVD) Market experiences steady growth because of three factors, which include the increasing number of elderly citizens, rising chronic disease rates and the growing need for early disease detection. Advanced technologies such as molecular diagnostics and automated testing systems are transforming laboratory practices and improving diagnostic accuracy. The market expansion receives backing through two factors, which include the strong healthcare infrastructure and the ongoing medical technology company innovations. Government initiatives which promote preventive healthcare and personalised medicine create a positive impact on the adoption of in vitro diagnostic solutions throughout hospitals and clinics and research laboratories across Japan.
Key Market Trends & Insights:
- The rising number of elderly people in Japan creates a greater demand for precise medical testing to detect diseases, including cancer, diabetes and cardiovascular disorders. Through in vitro diagnostics scientists can discover medical conditions at their initial stages, which helps to enhance treatment results.
- People prefer molecular diagnostic technologies because these systems enable them to identify medical conditions through genetic and molecular testing. Japanese hospitals and laboratories are starting to use advanced tests which deliver more accurate results within shorter time periods.
- The laboratory industry experiences transformation through automation and AI systems and high-throughput testing solutions. The healthcare sector uses these breakthroughs to generate faster and more accurate diagnostic results.
- The Japanese government maintains its commitment to advancing preventive healthcare programmes. Diagnostic testing requirements throughout the nation are increasing because of expanded health screening initiatives and public education programmes.
- Clinics and small healthcare facilities are increasingly using point-of-care diagnostic solutions. The tests produce immediate results which enable doctors to determine treatment options without needing to wait for laboratory results.
- Healthcare professionals use genetic and biomarker data analysis to develop customised treatment plans for each patient.
Japan In Vitro Diagnostics Market Segmentation
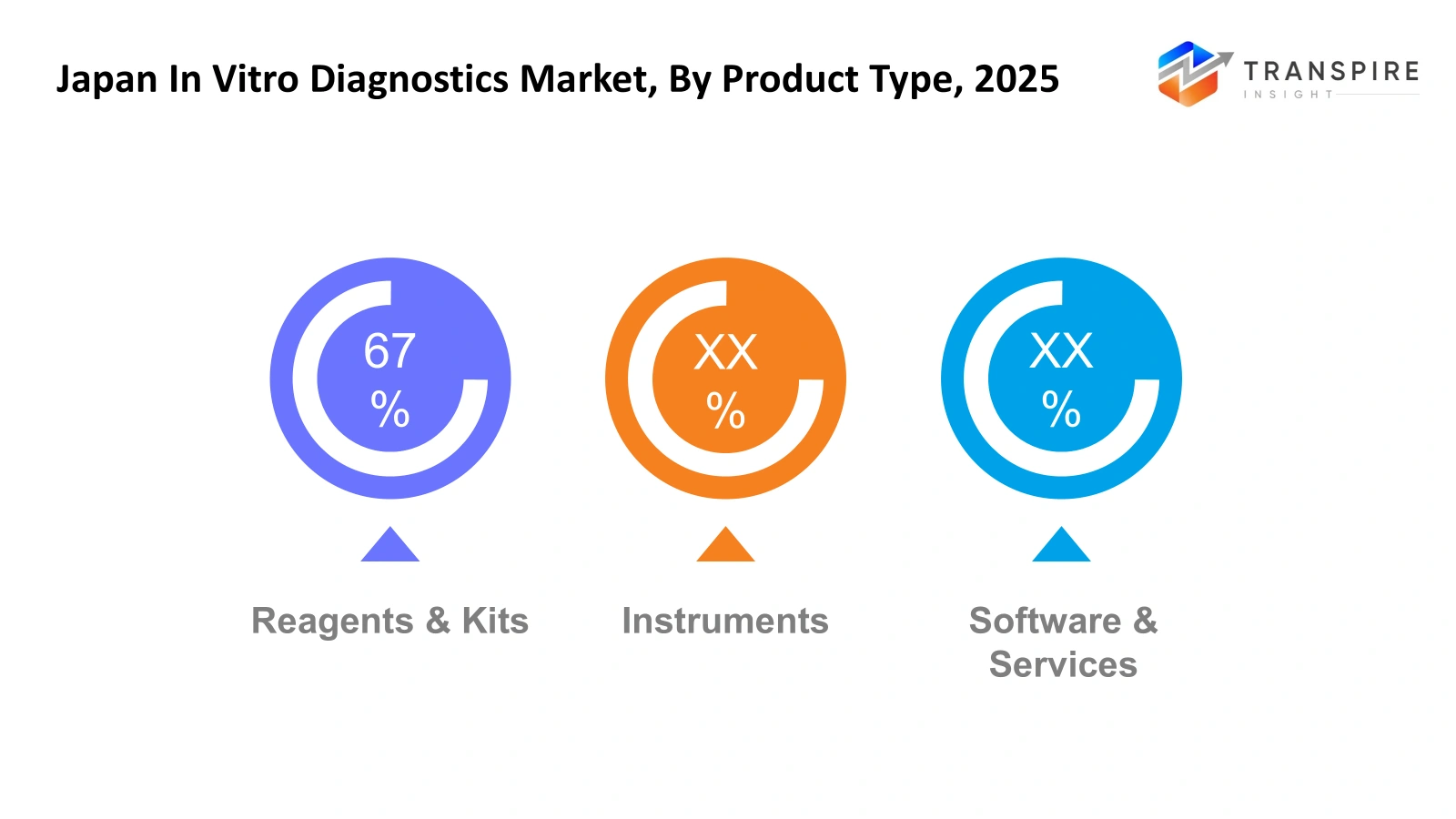
By Product Type
- Reagents & Kits: The Japan In Vitro Diagnostics market depends on reagents and kits, which serve as vital components that enable both common and advanced diagnostic procedures. Hospitals and laboratories depend on these materials, which they use every day to achieve precise testing outcomes. Healthcare organisations maintain high levels of reagent and kit usage because they need to operate continuous disease screening and monitoring programmes.
- Instruments: Laboratories and hospitals depend on diagnostic instruments to produce accurate test results with high operational effectiveness. Japan has implemented advanced analysers together with automated systems to achieve faster testing procedures while minimising human errors. Healthcare organisations continue to evolve their diagnostic systems because they want to implement new technological advancements through equipment upgrades.
- Software & Services: Diagnostic data management and workflow efficiency improvement now rely more on software together with its associated services. Digital platforms enable laboratories to store their data while analysing and sharing patient test results in a secure manner. Japan enables better data management and reporting accuracy through its software solutions, which work together with diagnostic equipment to enhance laboratory productivity.
To learn more about this report, Download Free Sample Report
By Technology
- Immunoassay: The Japanese medical field uses immunoassay technology to identify hormone levels and infectious diseases and cancer biomarkers. The system measures antigen-antibody interactions to provide accurate and precise diagnostic outcomes. The technology maintains its current status as a favoured testing method because it delivers efficient results with precise measurements during standard laboratory assessments.
- Molecular Diagnostics: Molecular diagnostics is gaining strong traction in Japan as it enables the detection of diseases at the genetic and molecular level. The system helps identify infectious diseases and genetic disorders and particular cancer types. The system delivers precise results which drive the increasing need for personalised medicine and precision medicine.
- Clinical Chemistry: Clinical chemistry tests use blood and urine and other body fluids to assess a person's health status. The medical field uses these tests to track metabolic disorders, assess how organs function, and monitor chronic health conditions. The Japanese healthcare system considers clinical chemistry tests as essential diagnostic tests because of their dependable results and common application during health assessments.
By End User
- Hospitals: Japan's in vitro diagnostics market hospitals function as primary customers because their diagnostic testing needs serve as the main reason for their existence. The facilities depend on cutting-edge diagnostic equipment which helps them make treatment choices. The continuous patient flow together with urgent diagnostic needs has led hospitals to implement IVD solutions.
- Diagnostic Laboratories: The specialised testing services of diagnostic laboratories operate as essential testing facilities which deliver both specialised testing services and extensive testing capabilities. Many healthcare providers in Japan use independent laboratories when they need to perform comprehensive diagnostic testing. Laboratories possess advanced equipment and trained staff members, which enables them to conduct multiple tests accurately while maintaining operational efficiency.
Country Insights
The Japan In Vitro Diagnostics Market demonstrates the country's robust healthcare system and its increasing dedication to preventive medical practices. Japan experiences a surge in chronic disease cases, which includes cancer, diabetes and cardiovascular diseases, because it has one of the fastest-growing elderly populations worldwide. The need for precise and swift diagnostic methods has experienced substantial growth. Medical facilities throughout the nation implement advanced medical technologies, which include molecular diagnostic tools and automated laboratory systems, to enhance their testing capacities and support their clinical decision processes.
Government initiatives which promote regular health examinations and disease prevention programmes help hospitals and laboratories expand their use of diagnostic testing procedures. The strong research capabilities of Japan, combined with its medical institutions and technology companies working together, create an environment that fosters ongoing development of diagnostic equipment. The country's commitment to delivering exceptional healthcare and specialised medical services drives the progress of dependable and sophisticated in vitro diagnostic technologies. The increasing healthcare knowledge among people of Japan will lead to sustained high demand for quick and effective diagnostic testing services throughout the nation.
Recent Development News
Japan Approves Companion Diagnostic Test for Breast Cancer Drug.
Foundation Medicine Expands Precision Diagnostics with New Approvals in Japan.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 7.28 Billion |
|
Market size value in 2026 |
USD 7.60 Billion |
|
Revenue forecast in 2033 |
USD 10.34 Billion |
|
Growth rate |
CAGR of 4.50% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Japan |
|
Key company profiled |
Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Thermo Fisher Scientific Inc., Danaher Corporation, Bio-Rad Laboratories Inc., Sysmex Corporation, Qiagen NV, Hologic Inc., Becton Dickinson & Company, Agilent Technologies Inc., Illumina Inc., PerkinElmer Inc., Ortho Clinical Diagnostics, Mindray Medical International. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Product Type (Reagents & Kits, Instruments, Software & Services), By Technology (Immunoassay, Molecular Diagnostics, Clinical Chemistry), By End User (Hospitals, Diagnostic Laboratories). |
Key Japan In Vitro Diagnostics Company Insights
The Japan In Vitro Diagnostics market includes international healthcare corporations together with established Japanese companies as its main competitors. The companies concentrate their efforts on creating new diagnostic technologies which provide more rapid and precise testing outcomes. The companies maintain their research and development spending, which enables them to develop new testing technologies that focus on molecular diagnostics and automated laboratory systems. The companies use strategic partnerships with hospitals and laboratories and research institutions to increase their product distribution. The market participants enhance their competitive position in Japan's diagnostic market by focusing on three key areas, which include product quality, operational reliability and technological progress.
Company List
- Roche Diagnostics
- Abbott Laboratories
- Siemens Healthineers
- Thermo Fisher Scientific Inc.
- Danaher Corporation
- Bio-Rad Laboratories Inc.
- Sysmex Corporation
- Qiagen NV
- Hologic Inc.
- Becton Dickinson & Company
- Agilent Technologies Inc.
- Illumina Inc.
- PerkinElmer Inc.
- Ortho Clinical Diagnostics
- Mindray Medical International.
Japan In Vitro Diagnostics Market Report Segmentation
By Product Type
- Reagents & Kits
- Instruments
- Software & Services
By Technology
- Immunoassay
- Molecular Diagnostics
- Clinical Chemistry
By End User
- Hospitals
- Diagnostic Laboratories
Frequently Asked Questions
Find quick answers to common questions.
The approximate Japan In Vitro Diagnostics market size for the market will be USD 10.34 Billion in 2033.
The key segments of the Japan In Vitro Diagnostics Market are By Product Type (Reagents & Kits, Instruments, Software & Services), By Technology (Immunoassay, Molecular Diagnostics, Clinical Chemistry), By End User (Hospitals, Diagnostic Laboratories).
Major players in the Japan In Vitro Diagnostics market are Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Thermo Fisher Scientific Inc., Danaher Corporation, Bio-Rad Laboratories Inc., Sysmex Corporation, Qiagen NV, Hologic Inc., Becton Dickinson & Company, Agilent Technologies Inc., Illumina Inc., PerkinElmer Inc., Ortho Clinical Diagnostics, Mindray Medical International.
The current market size of the Japan In Vitro Diagnostics market is USD 7.28 Billion in 2025.
The Japan In Vitro Diagnostics Market CAGR is 4.50%.
- Roche Diagnostics
- Abbott Laboratories
- Siemens Healthineers
- Thermo Fisher Scientific Inc.
- Danaher Corporation
- Bio-Rad Laboratories Inc.
- Sysmex Corporation
- Qiagen NV
- Hologic Inc.
- Becton Dickinson & Company
- Agilent Technologies Inc.
- Illumina Inc.
- PerkinElmer Inc.
- Ortho Clinical Diagnostics
- Mindray Medical International.
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)









