Japan Liver Cirrhosis Drugs Market Size & Forecast:
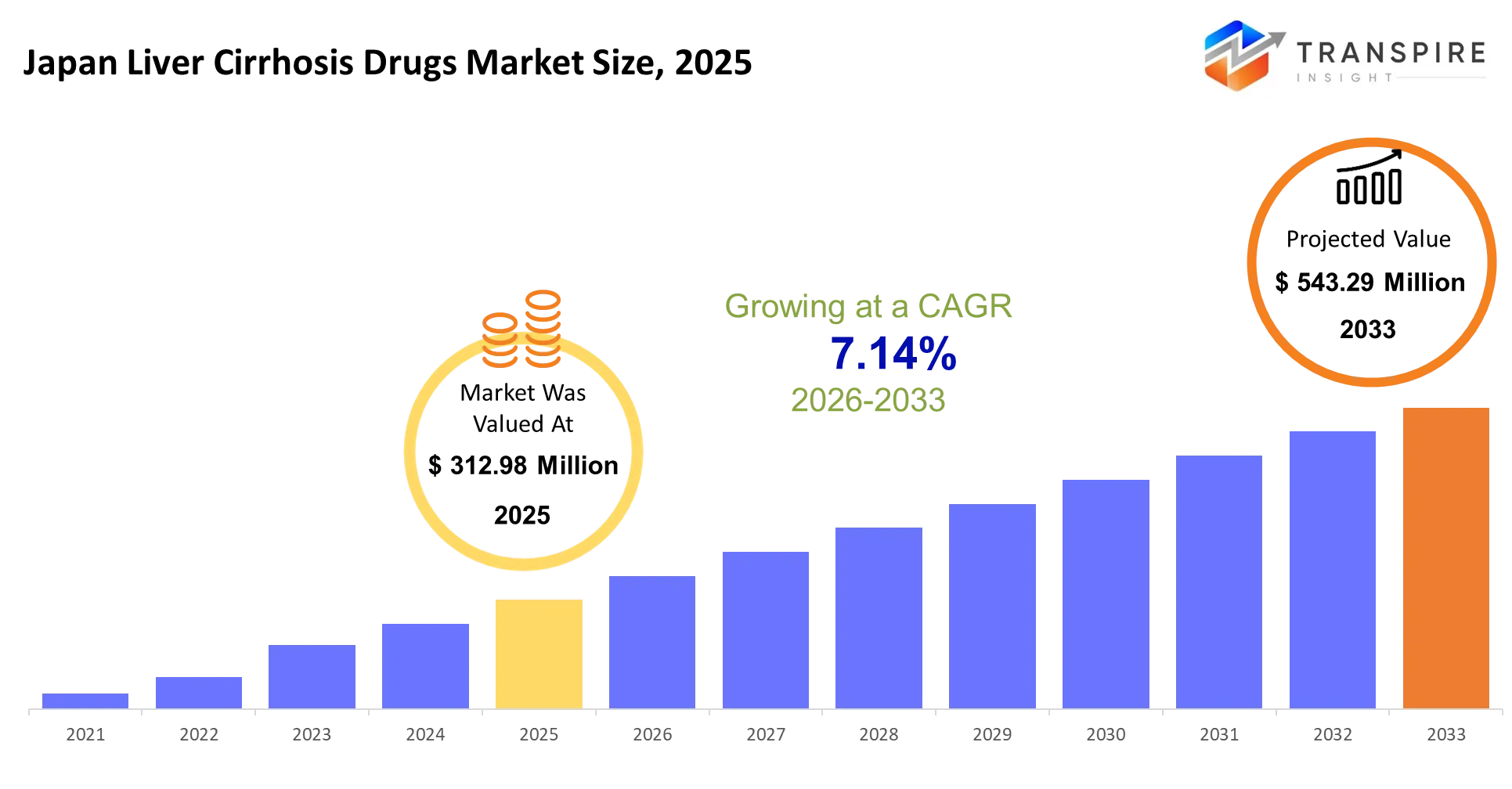
- Japan Liver Cirrhosis Drugs Market Size 2025: USD 312.98 Million
- Japan Liver Cirrhosis Drugs Market Size 2033: USD 543.29 Million
- Japan Liver Cirrhosis Drugs Market CAGR: 7.14%
- Japan Liver Cirrhosis Drugs Market Segments: By Type (Antiviral Drugs, Diuretics, Antibiotics, Immunosuppressants, Antifibrotic Drugs, Others); By Application (Alcoholic Cirrhosis, Viral Hepatitis, Non-alcoholic Fatty Liver Disease, Liver Failure Management, Others); By End-User (Hospitals, Clinics, Specialty Centers, Research Institutes, Others); By Distribution (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Others)
To learn more about this report, Download Free Sample Report
Japan Liver Cirrhosis Drugs Market Summary
The Japan Liver Cirrhosis Drugs Market was valued at USD 312.98 Million in 2025. It is forecast to reach USD 543.29 Million by 2033. That is a CAGR of 7.14% over the period.
The Japan liver cirrhosis drugs market supports clinicians managing progressive liver failure by stabilizing patients whose liver function has deteriorated due to hepatitis damage, fatty liver progression, and alcohol-related fibrosis. The organization develops therapeutic methods for treating primary health problems which include hepatic encephalopathy and portal hypertension and fluid buildup from ascites.
The treatment landscape has undergone fundamental changes during the past three to five years because the medical community has shifted its focus from hepatitis C as a cause of cirrhosis to nonalcoholic steatohepatitis and alcohol-related liver disease. Japan's national hepatitis elimination program together with direct-acting antiviral treatment has successfully decreased new HCV cases while the country's aging population has led to increased metabolic liver disease occurrence.
The COVID-19 pandemic created disruptions in screening procedures which resulted in later-stage disease detection and increased use of therapies for managing symptoms and long-term care. The company achieves revenue growth through its chronic care programs and advanced-stage disease treatment drugs which have replaced its previous revenue sources from antiviral medicines that provide permanent cures.
Key Market Insights
- Japan fulfills its entire national requirement because the Kanto region supplies approximately 38% of the needs due to its numerous hospitals and special liver treatment facilities.
- The Kansai region is the fastest-growing sub-market through 2026–2030, driven by expanding hepatology screening programs.
- Supportive drug therapies dominate the Japan Liver Cirrhosis Drugs Market with over 55–60% share, led by diuretics and lactulose-based treatments.
- The second-largest market segment consists of antiviral-related liver complication medications because post-hepatitis patients need extended medical treatment.
- Rifaximin-based therapies represent the fastest-growing segment, which will experience continuous growth because of increasing hepatic encephalopathy cases.
- The primary use of hepatic encephalopathy research accounts for 40 percent of total applications because Japanese hospitals report high rates of patient readmission.
- The application of ascites management develops into the most rapidly expanding field because medical professionals now identify more cases of advanced cirrhosis after the COVID pandemic.
- The hospital pharmacy sector controls approximately 70 percent of end-user market share because healthcare providers need direct supervision when they treat patients with chronic liver diseases.
- The decentralized hepatology care models create new specialty clinics which become the fastest expanding end-user market segment.
- Gilead and AbbVie focus on post-antiviral complication ecosystems strengthening their Japan Liver Cirrhosis Drugs Market positioning.
- The companies Roche and Merck develop new medical solutions through their creation of advanced hepatic biomarker systems which enable treatment tracking.
What are the Key Drivers, Restraints, and Opportunities in the Japan Liver Cirrhosis Drugs Market?
Rising metabolic dysfunction-associated steatohepatitis and alcohol-related liver disease together with Japan's growing elderly population and decreasing physical activity rates serve as the primary cause for this situation. Healthcare systems currently handle the increased requirement for treatment of advanced metabolic liver damage which needs ongoing medication because hepatitis C-related cirrhosis has become less common through effective antiviral therapies. The new treatment approach has increased diuretics and lactulose and rifaximin prescriptions which now generate ongoing revenue for pharmaceutical companies instead of producing single payments for antiviral therapies.
The main obstacle exists because advanced cirrhosis develops permanent disease reversibility limitations. Medical professionals can only provide palliative care to patients after they develop advanced fibrosis or decompensation because there are no effective treatment methods. The situation restricts treatment results which forces patients to remain in expensive medical programs that do not enhance their survival chances and this pattern restricts market growth while delaying the use of advanced disease-modifying treatments.
Early-stage fibrosis detection which uses biomarker-based diagnostic methods presents a new business opportunity that hospital networks in Tokyo and Osaka should develop. The pilot programs which utilize advanced elastography together with AI-assisted liver imaging technology achieve better results for early detection of liver disease which allows doctors to start preventive treatment through drugs. The Japan Liver Cirrhosis Drugs Market will evolve from its current state of treating established diseases into a new phase of growth that focuses on precise liver disease treatment if this program reaches full implementation.
What Has the Impact of Artificial Intelligence Been on the Japan Liver Cirrhosis Drugs Market?
Japan's liver disease treatment system uses artificial intelligence technology to change both its hepatology treatment delivery and its pharmaceutical medicine treatment methods. AI-based clinical decision support systems in hospitals use electronic health record data and medical imaging results and laboratory biomarker data to perform automated patient classification which medical professionals use to determine cirrhosis treatment options. The system enables tertiary care centers to work more effectively while it decreases the time needed for diagnosis in Japanese hospitals that handle large patient volumes.
Healthcare facilities use predictive analytics models to forecast disease progression events which include hepatic encephalopathy episodes and fluid accumulation and hospital readmission risks. Machine learning tools trained on longitudinal patient datasets help physicians identify high-risk patients earlier, enabling timely adjustment of diuretics, lactulose, or rifaximin-based regimens. The predictive capabilities of the system support better hospitalization planning while they help with resource management, which results in fewer emergency admissions and reduced overall treatment expenses.
AI-based imaging interpretation systems use artificial intelligence to improve fibrosis staging accuracy through elastography and CT analysis while increasing early treatment detection rates. The system forces hospitals to adopt its system because their clinical data networks lack proper connection methods between different hospitals. The high privacy protection requirements and small disease-specific datasets from Japan create challenges for model training, while the expensive integration process hampers implementation in smaller healthcare facilities.
Key Market Trends
- The hepatitis C cure programs have achieved a 70% reduction in antiviral cirrhosis cases since their 2018 implementation which now directs medical resources toward treating chronic liver disease complications.
- The COVID pandemic which began in 2020 caused delays in liver screening programs that resulted in more advanced cirrhosis cases being diagnosed while drug treatments in hospitals throughout Japan increased.
- The success of direct-acting antivirals changed prescribing patterns in the Japan Liver Cirrhosis Drugs Market by decreasing the need for curative treatments and increasing the demand for extended maintenance therapies.
- The adoption of rifaximin increased after 2022 because the incidence of hepatic encephalopathy recurrence in older Japanese patients began to rise.
- The hospital system has achieved better real-time liver function monitoring since 2021 through digitalization which allows doctors to make quicker changes to cirrhosis drug treatments.
- AbbVie and Gilead changed their business approach from antiviral market leadership to treatment of post-cure liver complications, which they considered essential for maintaining their market presence.
- Tokyo hospitals adopted artificial intelligence technology for fibrosis detection starting in 2023, which improved their ability to identify patients early and led to modifications in their drug treatment protocols.
- Since 2019 the demand for cirrhosis treatment has changed because people now seek treatment for lifestyle-related cirrhosis more than they seek treatment for viral cirrhosis and alcohol-related liver disease cases have increased.
- The specialty clinics experienced rapid growth after the 2022 policy changes which allowed them to provide treatment outside of tertiary hospitals while making medications more available to outpatient services.
Japan Liver Cirrhosis Drugs Market Segmentation
By Type :
The antiviral medications treat liver cirrhosis which results from viral infections by decreasing viral replication while they protect against further liver deterioration. The medical community employs these medications to treat cases associated with hepatitis. Diuretics help patientswho experience fluid retention because they provide better comfort to their medical condition. The medical team uses antibiotics to treat infection threats, while antifibrotic medications work to decrease liver tissue fibrosis progression. The medical team applies immunosuppressant therapy to select patients who show immune system disorders.
Medical professionals assign each medication to its appropriate treatment function according to the patient's health status and disease progression. The combination of antiviral and antifibrotic treatments enables patients to achieve extended treatment results, whereas diuretics and antibiotics help manage their immediate medical symptoms and protect against possible health issues. Specific medical situations require doctors to use immunosuppressant medications. The combination of these two drug types enhances patient health outcomes in various medical facilities throughout Japan.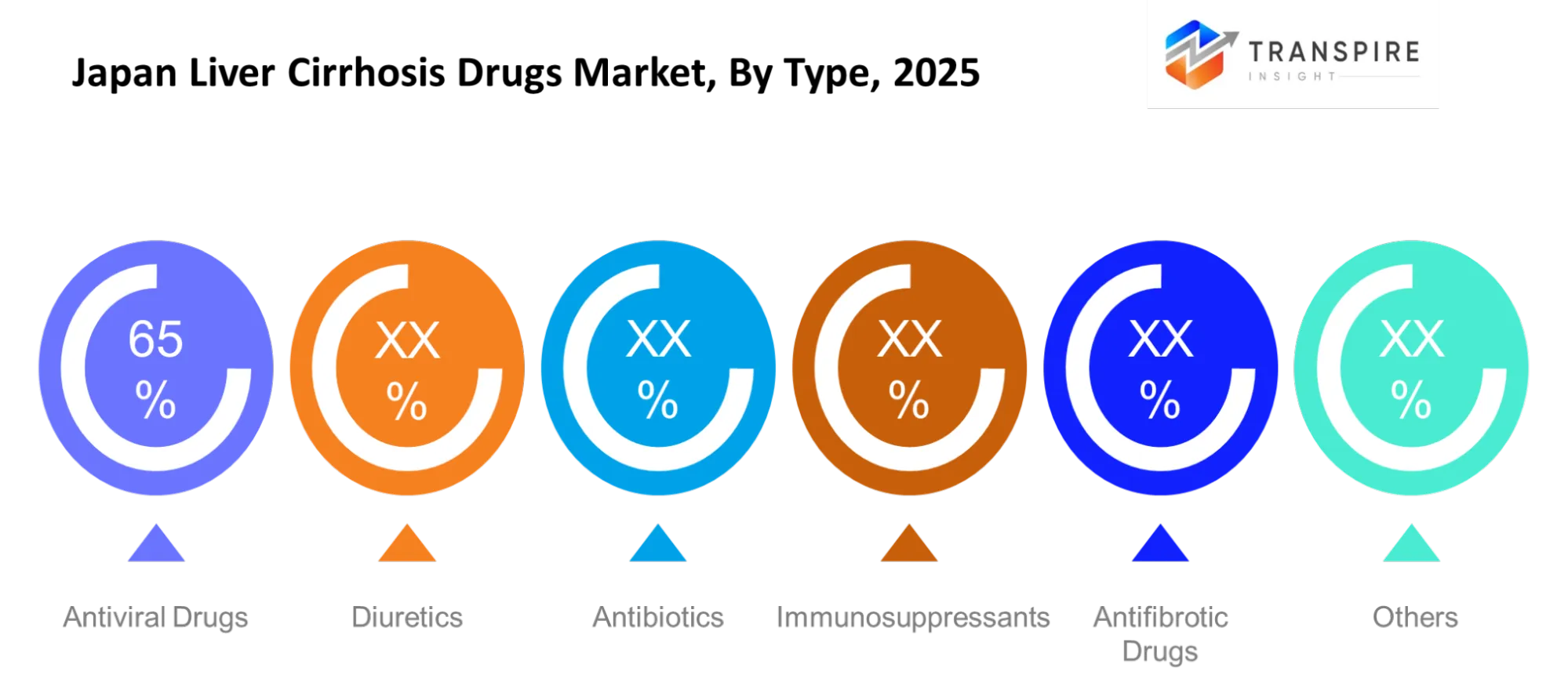
To learn more about this report, Download Free Sample Report
By Application :
The medical team uses alcohol treatment programs for alcoholic cirrhosis to minimize liver destruction which results from extended alcohol consumption. Doctors in the medical field use antiviral medications to treat patients who have viral hepatitis. Patients with non-alcoholic fatty liver disease require assistance to achieve their metabolic control goals. Healthcare providers use intensive medication treatment to manage acute liver failure cases until patients achieve stability.
Different treatment requirements exist for each application area because of variations in disease cause and severity. The development of cirrhosis through viral hepatitis remains the primary reason for patients who require medication treatment. Alcohol-related cases require long-term management approaches. The rising incidence of non-alcoholic fatty liver disease results from modern changes in human dietary practices and their physical activity patterns. Emergency medical treatment with pharmaceutical drugs becomes necessary for patients who experience liver failure.
By End-User :
The main responsibility of hospitals lies in providing treatments for patients with liver cirrhosis through their extensive medical facilities which include advanced treatment methods and dangerous condition medication delivery. Clinics provide treatment for patients who have not yet reached advanced stages while their health status gets continuous supervision. Specialty centers develop treatment approaches which specifically target liver diseases. Research institutes develop new medications and clinical research initiatives. Other healthcare facilities provide assistance to patients who need follow-up treatment and rehabilitation services.
Each end-user segment contributes to different stages of patient care. Emergency treatment and advanced medical procedures go through hospitals while clinics handle regular patient treatment and diagnostic procedures. Specialty centers deliver specialized liver treatment through their team of skilled professionals. Research institutes strengthen future treatment development through studies. The combination of all institutional efforts results in improved disease management practices.
By Distribution :
The primary distribution method for liver cirrhosis medication occurs through hospital pharmacies which serve both inpatient treatment needs and emergency room requirements. Patients can obtain prescribed medications through retail pharmacies which provide direct access to outpatient services. Patients who need ongoing medication treatment can use online pharmacies to easily refill their prescriptions and access their required medications from their doctor.
Each distribution channel supports different patient needs. Hospital pharmacies provide essential medications that doctors need for immediate patient care.Retail pharmacies enable communities to obtain their prescription medications. Online pharmacies provide patients with a simple solution to manage their medications throughout their extended treatment period. The combination of these channels enhances medication distribution while maintaining uninterrupted treatment for liver cirrhosis patients throughout Japan.
What are the Key Use Cases Driving the Japan Liver Cirrhosis Drugs Market?
The primary function of the Japan Liver Cirrhosis Drugs Market involves using its products to provide medical facilities with solutions that treat patients suffering from decompensated cirrhosis who need ongoing treatment for their ascites and hepatic encephalopathy and portal hypertension deficiencies. The clinical requirements of advanced-stage patients create the highest need for medical treatment because they need ongoing drug therapy which does not provide them with a complete cure.
The existing use cases now extend to outpatient treatment of chronic liver diseases through specialized clinics and medical services which provide care to patients who have been discharged from hospitals. The medical facilities within urban networks of Tokyo and Osaka now use diuretics and lactulose and rifaximin to treat patients who need stabilization before they return home except for the growing demand for clinic-based hepatology services.
Organizations began to explore new usage scenarios which focus on creating preventive measures for managing early fibrosis through health screenings and AI-based imaging technology used in corporate health assessment programs. The medical field now sees transplant-preparatory pharmacological optimization at tertiary hospitals because patients use drugs to achieve stability before they undergo liver transplantation surgeries.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 312.98 Million |
|
Market size value in 2026 |
USD 335.34 Million |
|
Revenue forecast in 2033 |
USD 543.29 Million |
|
Growth rate |
CAGR of7.14% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Japan |
|
Key company profiled |
Roche, Gilead Sciences, Pfizer, Novartis, AbbVie, Merck, Bristol Myers Squibb, AstraZeneca, Sanofi, Takeda, Eli Lilly, Johnson & Johnson, Bayer, Teva, Amgen |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Antiviral Drugs, Diuretics, Antibiotics, Immunosuppressants, Antifibrotic Drugs, Others); By Application (Alcoholic Cirrhosis, Viral Hepatitis, Non-alcoholic Fatty Liver Disease, Liver Failure Management, Others); By End-User (Hospitals, Clinics, Specialty Centers, Research Institutes, Others); By Distribution (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Others) |
Which Regions are Driving the Japan Liver Cirrhosis Drugs Market Growth?
The Japan Liver Cirrhosis Drugs Market industry in the Kanto region operates as the leading market because its hospitals and medical centers and specialized liver treatment facilities all operate at advanced levels. Tokyo's medical system functions optimally because the government provides substantial financial resources for both chronic illness treatment and preventive health examination initiatives. The region maintains ongoing requirements for cirrhosis drugs because patients arrive from nearby areas which create constant need for these treatments. The establishment of major pharmaceutical distributors together with complete hospital pharmacy systems creates a reliable system which enables patients to access medications at all times.
The Kansai region functions as a dependable secondary center which exhibits healthcare system diversity throughout its three cities Osaka and Kyoto and Hyogo. The healthcare system in the Kansai region develops through both public and private sector investments which fund the establishment of new medical facilities and specialty clinics that serve the whole region. The local pharmacy system operates through established hospital networks which implement standardized treatment procedures for chronic liver diseases. The local economy maintains stable economic conditions which enable patients to access necessary treatments throughout all periods of the year.
The Kyushu region has become the most rapidly developing area because it received new hospital infrastructure improvements and the government began its regional healthcare equity initiative in 2023. The Fukuoka health system expanded its liver disease treatment facilities while rural areas increased their health screening capacity which resulted in higher rates of early liver disease detection. The new program increased use of cirrhosis drugs in areas beyond standard urban locations. The healthcare market in Kyushu presents investors and new businesses with major growth opportunities which will last until 2026-2033 because the region's healthcare services become more accessible throughout different areas.
Who are the Key Players in the Japan Liver Cirrhosis Drugs Market and How Do They Compete?
The Japan Liver Cirrhosis Drugs Market exhibits moderate competition between international pharmaceutical companies, while Japanese markets for supportive-care generics show complete market division.The field of leadership has transitioned from antiviral treatments that cure diseases to therapies that manage chronic cirrhosis. The companies in this market compete based on their clinical evidence capabilities and their ability to access hospital formularies and their actual patient results in advanced liver disease treatment. The decline in hepatitis C cases has compelled companies to redirect their current products towards developing treatments for metabolic disorders and alcohol-related liver diseases.
Gilead Sciences functions through its antiviral products and its Japanese market post-cure liver complication systems to create enduring hospital prescribing partnerships. AbbVie expands fibrosis-focused collaborations with Japanese universities, leveraging prior hepatitis C portfolio strength. Bristol Myers Squibb develops immunology-based research programs that target inflammatory pathways responsible for liver disease progression. Merck & Co. strengthens its market position through clinical trials that use biomarkers and its advanced liver disease treatment partnerships.
Roche achieves improved fibrosis staging results through its proprietary biomarker diagnostic solutions and imaging technologies, which enable Japanese hospitals to begin treatment at an earlier point.Eisai uses its extensive hospital network in Japan to develop liver disease treatment services that reach clinics in regional areas. The two organizations focus on implementing precision medicine solutions to enhance their adoption rate in Japan's developing hepatology treatment system.
Company List
- Roche
- Gilead Sciences
- Pfizer
- Novartis
- AbbVie
- Merck
- Bristol Myers Squibb
- AstraZeneca
- Sanofi
- Takeda
- Eli Lilly
- Johnson & Johnson
- Bayer
- Teva
- Amgen
Recent Development News
In February 2026, Verrica Pharmaceuticals and its Japanese partner Torii Pharmaceutical commercially launched YCANTH in Japan for molluscum contagiosum following regulatory approval from Japan’s Ministry of Health, Labour and Welfare. The new product launch expands Torii's specialized pharmaceutical market presence in Japan while the company develops its complete portfolio of liver-related infectious diseases and antiviral treatments through its partnership with Shionogi, which specializes in hepatic disease and virology-based treatments.
Source https://www.investing.com/
In September 2025, Ipsen received approval in Japan for Bylvay (odevixibat), a first-in-class bile acid transport inhibitor for treating pruritus associated with progressive familial intrahepatic cholestasis (PFIC). PFIC is a rare but severe pediatric liver disorder that often progresses to liver cirrhosis, making this approval a key expansion in Japan’s rare liver disease treatment landscape and strengthening the orphan drug segment of hepatology therapeutics.
Source https://live.euronext.com/
What Strategic Insights Define the Future of the Japan Liver Cirrhosis Drugs Market?
The Japan Liver Cirrhosis Drugs Market is heading towards treatment methods which provide long-term solutions for patients who experience complications because the need for antiviral cures has decreased and metabolic liver disease has become the main medical challenge. The period of 5–7 years will show increased development through the development of treatment plans which use drugs as part of hospital and specialty clinic chronic care systems. The transition occurs because Japan's elderly citizens and increasing non-viral cirrhosis cases will create ongoing medical needs which require regular prescription use instead of temporary treatment sessions.
The emergence of fibrosis-reversal biologics and regenerative medicine treatments which substitute existing medical therapies creates a less noticeable danger because these new treatments will decrease dependency on standard supportive medications. The clinical development of these therapies will reduce the need for long-term pharmaceutical treatment during advanced patient care.
The development of AI-based early fibrosis detection systems which national health screening programs will implement in urban areas of Tokyo and Osaka represents a new business opportunity. This path allows medical professionals to begin drug treatments earlier while patients can receive care for longer time periods. The market needs to focus on forming alliances with diagnostic technology companies and hospital systems because this will enable them to establish their role in the early intervention market which will become more important as prevention methods start to dominate treatment approaches.
Japan Liver Cirrhosis Drugs Market Report Segmentation
By Type
- Antiviral Drugs
- Diuretics
- Antibiotics
- Immunosuppressants
- Antifibrotic Drugs
By Application
- Alcoholic Cirrhosis
- Viral Hepatitis
- Non-alcoholic Fatty Liver Disease
- Liver Failure Management
By End-User
- Hospitals
- Clinics
- Specialty Centers
- Research Institutes
By Distribution
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
Frequently Asked Questions
Find quick answers to common questions.
The Japan Liver Cirrhosis Drugs Market size is USD 543.29 Million in 2033.
Key Segments for the Japan Liver Cirrhosis Drugs Market are By Type (Antiviral Drugs, Diuretics, Antibiotics, Immunosuppressants, Antifibrotic Drugs, Others); By Application (Alcoholic Cirrhosis, Viral Hepatitis, Non-alcoholic Fatty Liver Disease, Liver Failure Management, Others); By End-User (Hospitals, Clinics, Specialty Centers, Research Institutes, Others); By Distribution (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Others).
Major Japan Liver Cirrhosis Drugs Market Players are Roche, Gilead Sciences, Pfizer, Novartis, AbbVie, Merck, Bristol Myers Squibb, AstraZeneca, Sanofi, Takeda, Eli Lilly, Johnson & Johnson, Bayer, Teva, Amgen.
The Current Japan Liver Cirrhosis Drugs Market size is USD 312.98 Million in 2025.
The Japan Liver Cirrhosis Drugs Market CAGR is 7.14% from 2026 to 2033.
- Roche
- Gilead Sciences
- Pfizer
- Novartis
- AbbVie
- Merck
- Bristol Myers Squibb
- AstraZeneca
- Sanofi
- Takeda
- Eli Lilly
- Johnson & Johnson
- Bayer
- Teva
- Amgen
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


