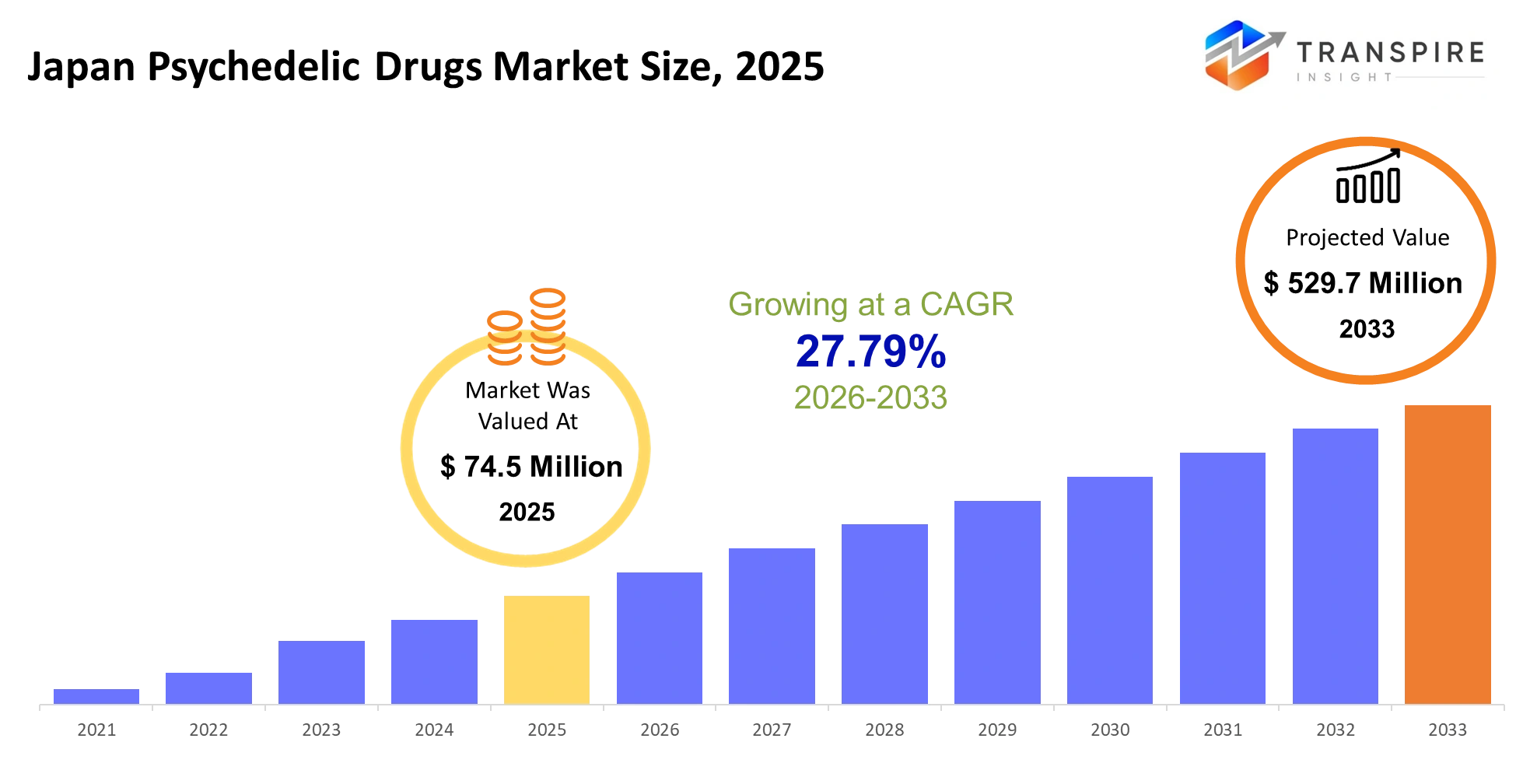
Japan Psychedelic Drugs Market Size & Forecast:
- Japan Psychedelic Drugs Market Size 2025: USD 74.5 Million
- Japan Psychedelic Drugs Market Size 2033: USD 529.7 Million
- Japan Psychedelic Drugs Market CAGR: 27.79%
- Japan Psychedelic Drugs Market Segments: By Type (Psilocybin, LSD, MDMA, Ketamine, Others); By Application (Depression Treatment, PTSD, Anxiety Disorders, Addiction Treatment, Neurological Disorders, Others); By End-User (Hospitals, Mental Health Clinics, Research Institutes, Rehabilitation Centers, Specialty Clinics, Others); By Distribution (Hospitals, Clinics, Online Therapy Platforms, Others).
To learn more about this report, Download Free Sample Report
Japan Psychedelic Drugs Market Summary
The Japan Psychedelic Drugs Market was valued at USD 74.5 Million in 2025. It is forecast to reach USD 529.7 Million by 2033. That is a CAGR of 27.79% over the period.
Japanese researchers investigate the effects of psychedelic-assisted therapies which they conduct in controlled clinical experiments to treat patients who experience treatment-resistant depression and post-traumatic stress disorder and end-of-life anxiety who do not respond to standard antidepressant medications(0). The market has developed from academic research activities during the previous two years into clinical trial procedures which hospitals use to conduct ketamine and esketamine treatments which demonstrates that institutions now embrace neuropsychiatric technological advancements.
The psychiatric treatment system experienced a major transformation because psychiatric practices became unified with precision medicine methods which received backing from enhanced clinical management and established therapy guidelines. The COVID-19 pandemic created actual mental health problems, while Japanese drug regulators and pharmaceutical companies advanced their research efforts because successful late-stage trials of psilocybin and MDMA treatments achieved positive results in international studies. The research and development partnerships and specialized clinics attract investment which leads to a cautious approach that enables steady business development.
Key Market Insights
- The Japan Psychedelic Drugs Market has progressed from academic research to the formation of hospital-based psychedelic-assisted mental health treatment facilities throughout the nation.
- The Japan Psychedelic Drugs Market ecosystem experiences increasing demand because of the rising cases of treatment-resistant depression and the substantial impact of PTSD.
- The pharmaceutical industry drives neuropsychiatric therapeutic innovations which transform the research and development approach used in the Japan Psychedelic Drugs Market.
- The Kanto region leads the Japan Psychedelic Drugs Market because it contains advanced hospitals and research universities and a high concentration of psychiatric treatment facilities.
- The period from 2024 to 2030 will see the Kansai region become the most rapidly developing area because of its growing clinical trial networks and psychiatric care investments.
- The Japan Psychedelic Drugs Market shows its main market share through ketamine-based treatments which produce quick clinical outcomes.
- Esketamine nasal formulations hold the second market position because regulatory bodies granted approval and psychiatric departments of major Japanese hospitals started using the product.
- Psilocybin-assisted therapy establishes itself as the fastest growing market segment from 2025 to 2030 through global trial success and research partnerships in Japan.
- The application area which experiences the most rapid growth includes PTSD and end-of-life anxiety management because of rising public understanding of mental health issues and increasing clinical research studies.
What are the Key Drivers, Restraints, and Opportunities in the Japan Psychedelic Drugs Market?
The Japan Psychedelic Drugs Market expands primarily because research studies which receive official approval investigate psychiatric treatment methods that do not respond to standard medical procedures. The increase in depression and PTSD cases which SSRIs failed to treat resulted in hospitals and research universities establishing supervised treatment programs which used ketamine and esketamine as therapeutic methods. Japan's Ministry of Health established clinical trial frameworks to align with international neuropsychiatric research standards which resulted in specialist hospitals increasing their usage of these frameworks to generate additional procedural revenue and develop their psychiatric medication research capabilities.
The Japanese narcotics classification system which establishes strict control over psychedelic substances through its scheduling regulations represents the main obstacle that hinders progress in Japan. Research compounds that receive approval for use face operational challenges which emerge from the requirement to obtain multiple ethical approvals together with extensive authorization processes. The medical product development process requires companies to spend considerable time on development work which prevents them from making money in their early stages while patients must wait for treatment despite strong demand for medical services. Smaller biotech companies face discouragement from entering the market because regulatory requirements create obstacles which reduce the rate of innovation in the Japan psychedelic drugs industry.
The research work of pharmaceutical companies from multiple nations will explore synthetic psilocybin and MDMA-assisted therapy development through their international collaborations. COMPASS Pathways and Japanese research hospitals are forming partnerships to create late-stage clinical validation centers for their international research projects. Japanese psychiatric institutions are constructing research facilities in Tokyo and Osaka to establish advanced neurotherapy testing sites which will make Japan a leading center for controlled psychedelic medicine development in the region.
What Has the Impact of Artificial Intelligence Been on the Japan Psychedelic Drugs Market?
The Japan Psychedelic Drugs Market experiences transformation through artificial intelligence and digital technology advancements which create new methods for developing and testing psychedelic-assisted therapies in clinical settings. AI-based platforms in drug development use neuroimaging and genomic data analysis to find patient profiles who will most probably benefit from ketamine and esketamine and new psilocybin-based treatments which helps decrease trial time and enhance trial accuracy. The healthcare sector uses machine learning models to create effective dosing solutions. The system uses digital mental health tracking to monitor patient reactions which helps doctors provide safe and precise treatment.
Predictive analytics has emerged as a vital enabler because algorithms predict patient response rates and relapse probabilities which enables clinicians to modify treatment methods before completing full therapy cycles. The Japan Psychedelic Drugs Market benefits from shorter trial periods which lead to better resource management in psychiatric research projects that improve operational efficiency while decreasing total development expenses.
The absence of extensive high-quality psychedelic therapy datasets prevents organizations from using AI technology because regulations limit both data acquisition and data distribution. The process of training algorithms becomes less efficient because of this issue whereas the precision of models decreases. The ongoing secure clinical data platform investments will gradually enhance integration capabilities which will enable organizations to develop digital psychiatry systems that can expand.
Key Market Trends
- The Japan Psychedelic Drugs Market evolved from its foundational research phase before 2022 to its current state which hospitals operate as clinical trial centers with backing from national psychiatric institutions.
- The 2023 regulations on psychedelic drugs became stricter through new classification requirements while the authorities approved additional frameworks that permit controlled use of ketamine for psychiatric treatments.
- The 2024 psilocybin trial localization process at university hospitals advanced through increased partnerships between COMPASS Pathways and other global companies which expanded their operations in Japan.
- The demand for medical treatments transformed after the 2021 pandemic period when treatment-resistant depression cases increased because people preferred rapid-acting therapies instead of conventional antidepressants.
- The period between 2023 and 2025 saw digital psychiatry tools develop new capabilities which permitted AI-enabled patient monitoring during psychedelic therapy trials at major urban hospitals.
- The clinical infrastructure development in Tokyo and Osaka experienced a major boost after 2024 which established these locations as leading centers for advanced neuropsychiatric research.
- Pharmaceutical companies redirected their research and development activities from traditional SSRIs towards developing neuroplasticity-based therapy solutions which target patients suffering from trauma and end-of-life anxiety disorders.
- The period between 2022 and 2023 brought stricter data-sharing rules which delayed the algorithm development process for creating psychedelic response prediction models used in Japan's clinical research environment.
- The adoption of institutional psychedelic therapies advanced from their initial pilot programs to multiple center research studies which showed that psychiatric systems had begun to implement these treatments.
Japan Psychedelic Drugs Market Segmentation
By Type :
The Japanese psychedelic drugs market consists of three main segments which include Psilocybin LSD and MDMA and Ketamine and other substances. The research studies Psilocybin for depression treatment and LSD for cognitive research and MDMA for PTSD treatment and Ketamine for fast-acting antidepressant results. The compounds enable researchers to conduct scientific studies in controlled medical research facilities.
The other compounds enable researchers to conduct experimental medical procedures which include clinical studies under complete medical monitoring and government approval. The developing research programs for controlled studies and psychiatric treatment methods will help these medications achieve recognition as advanced treatment options.
By Application :
The mental health crisis in Japan has created a need for depression treatment programs to become the primary focus of mental health services. The implementation of psychedelic-assisted therapy methods in clinical settings occurs because both PTSD and anxiety disorders require treatment. Patient response assessment together with emotional stabilization improvement forms the basis of structured treatment models.
Research now investigates both addiction treatment methods and neurological disorder treatment approaches. The other uses of this system involve supervised controlled experimental therapy conducted in institutional environments and organized clinical treatment programs. The expanding requirements of mental health services will drive growth in various application fields.
To learn more about this report, Download Free Sample Report
By End-User :
The hospitals and mental health clinics serve as the main customers because their needs demand controlled treatment spaces and availability of psychiatric treatment staff. The clinical supervision process establishes structured control for treatment delivery while maintaining patient safety during psychedelic-assisted therapy sessions.
The research institutes together with rehabilitation centers and specialty clinics and additional medical facilities conduct clinical trials and develop therapies and study psychedelic medicine through their controlled research programs. The medical institutions will enhance their evidence-based treatment implementation through increased collaboration efforts.
By Distribution :
The distribution of healthcare services at hospitals and clinics exists because regulations control prescription processes and require tracking of psychedelic treatment administration. The presence of strong clinical oversight maintains controlled access to patients who need medical treatment while ensuring compliance with established medical standards.
Online therapy platforms provide expanded access to consultation and screening services together with their guided therapy programs which operate under changing regulatory requirements and the growing use of digital mental health solutions. Digital platforms will enable assessment of initial stages and implementation of organized treatment methods for all patient populations.
What are the Key Use Cases Driving the Japan Psychedelic Drugs Market?
The primary application of the Japan Psychedelic Drugs Market revolves around using ketamine and esketamine therapies to treat major depressive disorder and treatment-resistant depression. Hospitals drive most of this demand because these conditions require supervised administration and structured psychiatric monitoring which makes controlled clinical environments essential to maintain patient safety and achieve reliable treatment results.
PTSD management and end-of-life anxiety care expanded into specialty psychiatric clinics and palliative care units. Urban healthcare centers in Tokyo and Osaka now implement psychedelic-assisted protocols as additional treatments when standard antidepressant medications do not deliver sufficient relief.
The emerging use cases of alcohol and substance dependence treatment and veteran trauma recovery and disaster-affected population trauma recovery operate as their own distinct use cases. Research hospitals in Japan investigate psilocybin-based treatments for cognitive rigidity disorders through their active clinical trials and the changing regulatory framework.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 74.5 Million |
|
Market size value in 2026 |
USD 95.2 Million |
|
Revenue forecast in 2033 |
USD 529.7 Million |
|
Growth rate |
CAGR of 27.79% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Japan |
|
Key company profiled |
Compass Pathways, MindMed, ATAI Life Sciences, Johnson & Johnson, Pfizer, Novartis, Roche, AstraZeneca, GSK, Takeda, AbbVie, Eli Lilly, Merck, Biogen, Otsuka |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Psilocybin, LSD, MDMA, Ketamine, Others); By Application (Depression Treatment, PTSD, Anxiety Disorders, Addiction Treatment, Neurological Disorders, Others); By End-User (Hospitals, Mental Health Clinics, Research Institutes, Rehabilitation Centers, Specialty Clinics, Others); By Distribution (Hospitals, Clinics, Online Therapy Platforms, Others) |
Which Regions are Driving the Japan Psychedelic Drugs Market Growth?
The Japan Psychedelic Drugs Market operates from Kanto, which displays extensive facilities for psychiatric research, tertiary hospitals, and government-related clinical research centers. Tokyo-based universities and medical networks have established early-stage psychedelic therapy programs, supported by strong regulatory oversight and funding for neuropsychiatric innovation. The region operates through a developed healthcare system, which enables cooperation between psychiatric specialists, academic institutions, and pharmaceutical firms who work together. The process of controlled ketamine and esketamine therapy adoption reaches its fastest speed because Kanto serves as the main center for both therapy implementation and research activities.
The Kansai region serves as a stable secondary contributor, driven by its strong private healthcare sector and long-standing pharmaceutical manufacturing base in Osaka and Kyoto. The research-heavy focus of Kanto creates an operational difference from Kansai, which emphasizes its steady clinical use and gradual incorporation of modern psychiatric solutions at existing medical facilities. The hospitals in this area maintain their treatment methods through established protocols, which they use to implement psychedelic-assisted therapies for outpatient psychiatric patients. The institutional behavior of this system maintains consistent demand patterns, which remain stable despite less research activity compared to the capital region.
The Kyushu region experiences rapid growth because of current investments in hospital modernization projects and expanded mental health services which began after the 2023 healthcare reform. The new psychiatric units established in Fukuoka and Kagoshima now conduct multi-center clinical trials which improve their access to experimental treatment options. The improved patient referral systems together with increased mental health awareness have led to faster adoption of mental health solutions. The market shift indicates to investors and new market entrants that clinical demand will expand across various regions between 2026 and 2033 while decreasing dependence on Tokyo-based healthcare services.
Who are the Key Players in the Japan Psychedelic Drugs Market and How Do They Compete?
The Japan Psychedelic Drugs Market maintains a medium level of market fragmentation because international pharmaceutical companies and biotechnology companies and university hospital systems all strive to achieve initial clinical success. The market lacks a dominant player because commercial activity remains focused on regulated trials instead of actual product launches. The main factors that drive market competition include organizations' ability to access clinical data and obtain regulatory approvals for controlled substances and establish partnerships with hospitals that support their psychiatric research studies. Companies that establish effective trial systems together with building their neuropsychiatric research recognition will secure a competitive advantage during the initial phase of their business operations.
Johnson & Johnson maintains a strong position through its esketamine therapy which is sold under the name Spravato because the treatment has received regulatory approval and hospitals distribute it. COMPASS Pathways specializes in developing psilocybin-assisted therapy by creating large clinical trials which use standardized psychotherapy methods. Atai Life Sciences and MindMed operate multiple psychedelic drug development programs while they research neuroplasticity compounds through their partnerships with academic institutions worldwide and their licensing agreements with Asian companies at the start of their development process.
Japanese companies Otsuka Pharmaceutical and Takeda expand their market presence by establishing psychiatric drug development expertise and building hospital network connections. Otsuka uses its existing central nervous system product range while Takeda develops its neuroscience research partnerships and international research studies. The companies achieve growth through their combined research initiatives with local universities and their strategic research agreements with international biotechnology companies which will enable them to enter the regulated psychedelic treatment market.
Company List
- Compass Pathways
- MindMed
- ATAI Life Sciences
- Johnson & Johnson
- Pfizer
- Novartis
- Roche
- AstraZeneca
- GSK
- Takeda
- AbbVie
- Eli Lilly
- Merck
- Biogen
- Otsuka
Recent Development News
In March 2026, Otsuka Pharmaceutical acquired Transcend Therapeutics for approximately $1.23 billion. The acquisition included Transcend’s psychedelic-based neuroplastogen program (TSND-201), strengthening Otsuka’s position in psychedelic-derived psychiatric therapeutics and expanding its global neuroscience pipeline, including Japan-linked commercialization rights.
Source: https://psychedelichealth.co.uk/
In April 2026, XTL Biopharmaceuticals announced the acquisition of Psyga Bio. The deal established a psychedelic biotechnology platform focused on psilocybin-based therapies, including clinical-stage candidates and GMP manufacturing capabilities, positioning the company for accelerated psychedelic drug development and commercialization across global markets including Asia.
Source: https://www.globenewswire.com/
What Strategic Insights Define the Future of the Japan Psychedelic Drugs Market?
The Japan Psychedelic Drugs Market is moving toward tightly regulated, hospital-centered psychiatric innovation rather than broad commercial diffusion, driven by rising treatment-resistant mental health burden and gradual acceptance of neuroplasticity-based therapies. The next 5 to 7 years will see growth come from clinical institutions successfully implementing standardized psychedelic-assisted protocols into their mainstream psychiatric practices instead of product development.
The danger exists because clinical datasets remain disorganized and scheduling systems create limits that prevent large-scale evidence development while making payers postpone their trust in reimbursement methods.
An emerging opportunity is the decentralization of clinical trials into secondary regions such as Kyushu, supported by AI-enabled patient stratification tools that improve trial efficiency. The Japanese psychedelic therapeutics market will benefit from partnership between hospital consortia and the development of real-world evidence platforms because these strategies will help market participants secure their future market position.
Japan Psychedelic Drugs Market Report Segmentation
By Type
- Psilocybin
- LSD
- MDMA
- Ketamine
By Application
- Depression Treatment
- PTSD
- Anxiety Disorders
- Addiction Treatment
- Neurological Disorders
By End-User
- Hospitals
- Mental Health Clinics
- Research Institutes
- Rehabilitation Centers
- Specialty Clinics
By Distribution
- Hospitals
- Clinics
- Online Therapy Platforms
Frequently Asked Questions
Find quick answers to common questions.
The Japan Psychedelic Drugs Market size is USD 529.7 Million in 2033.
Key segments for the Japan Psychedelic Drugs Market are By Type (Psilocybin, LSD, MDMA, Ketamine, Others); By Application (Depression Treatment, PTSD, Anxiety Disorders, Addiction Treatment, Neurological Disorders, Others); By End-User (Hospitals, Mental Health Clinics, Research Institutes, Rehabilitation Centers, Specialty Clinics, Others); By Distribution (Hospitals, Clinics, Online Therapy Platforms, Others).
Major Japan Psychedelic Drugs Market players are Compass Pathways, MindMed, ATAI Life Sciences, Johnson & Johnson, Pfizer, Novartis, Roche, AstraZeneca, GSK, Takeda, AbbVie, Eli Lilly, Merck, Biogen, Otsuka.
The Japan Psychedelic Drugs Market size is USD 74.5 Million in 2025.
The Japan Psychedelic Drugs Market CAGR is 27.79% from 2026 to 2033.
- Compass Pathways
- MindMed
- ATAI Life Sciences
- Johnson & Johnson
- Pfizer
- Novartis
- Roche
- AstraZeneca
- GSK
- Takeda
- AbbVie
- Eli Lilly
- Merck
- Biogen
- Otsuka
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


