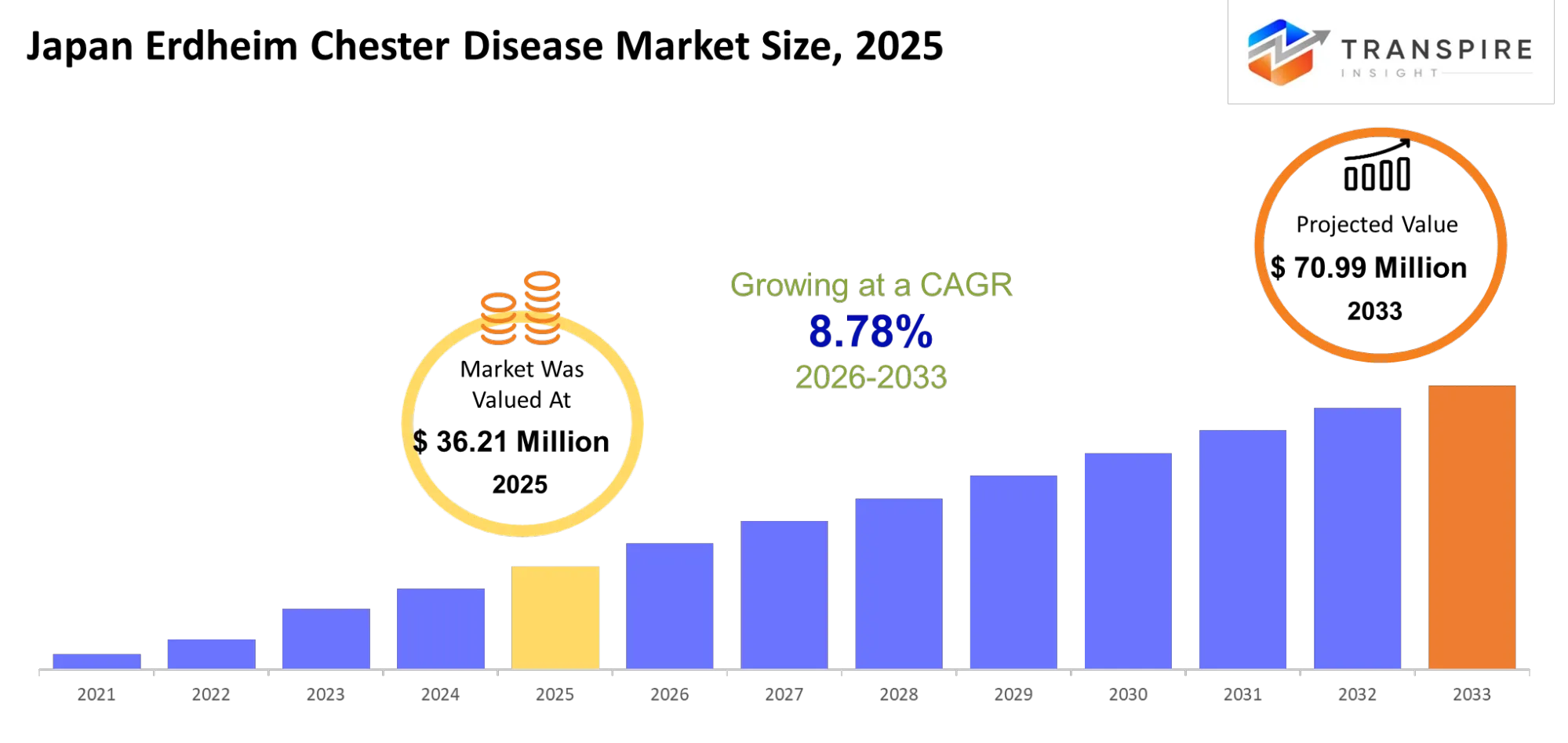
Japan Erdheim Chester Disease Market Size & Forecast:
- Japan Erdheim Chester Disease Market Size 2025: USD 36.21 Million
- Japan Erdheim Chester Disease Market Size 2033: USD 70.99 Million
- Japan Erdheim Chester Disease Market CAGR: 8.78%
- Japan Erdheim Chester Disease Market Segments: By Type (Targeted Therapy, Chemotherapy, Immunotherapy, Corticosteroids, Combination Therapy, Others); By Application (Rare Disease Treatment, Oncology Treatment, Immunological Disorders, Clinical Research, Others); By End-User (Hospitals, Clinics, Research Institutes, Specialty Centers, Academic Institutes, Others); By Distribution (Hospital Pharmacies, Specialty Pharmacies, Others)
To learn more about this report, Download Free Sample Report
Japan Erdheim Chester Disease Market Summary
The Japan Erdheim Chester Disease Market was valued at USD 36.21 Million in 2025. It is forecast to reach USD 70.99 Million by 2033. That is a CAGR of 8.78% over the period.
The Japan Erdheim Chester Disease market focuses on diagnosing and treating a rare multi-system histiocytic disorder which causes progressive organ damage and needs coordinated treatment from oncology and neurology and cardiology and nephrology physicians. The market uses advanced imaging and molecular testing methods to detect diseases at earlier stages which enables doctors to provide patients with extended targeted treatments that decrease disease progression while minimizing hospital admissions.
The market experienced structural changes during the last 3 to 5 years as it moved away from using broad immunosuppressive treatments to implement precision oncology methods which target MAPK pathways through BRAF and MEK inhibitors that genomically profile patients. Japan has experienced a diagnosis boost because of increased orphan drug incentives and next-generation sequencing adoption by its tertiary hospitals which had previously caused diagnosis delays and missed cases. The new treatment pathways have led to more patients using specialty medications which has enhanced treatment continuity and created additional revenue opportunities for pharmaceutical companies that develop medications for rare diseases.
Key Market Insights
- The Japan Erdheim Chester Disease Market is evolving from its current state which uses empirical immunosuppression to develop mutation-guided precision oncology treatments that utilize BRAF and MEK inhibitors.
- The market experiences growth because next-generation sequencing becomes more widely used which helps hospitals in Japan to detect rare diseases more effectively.
- The Kanto region controls more than 45% of the Japanese Erdheim Chester Disease market because it possesses better hospital facilities and research institutions.
- The Kansai region experiences the fastest growth because it implements new rare disease screening initiatives which will extend until 2028.
- Rural hospitals continue to experience extended diagnosis times because they lack proper access to molecular testing facilities.
- The targeted therapy segment leads the Japan Erdheim Chester Disease Market because it has the highest usage of BRAF and MEK inhibitor therapies.
- The diagnostic services segment ranks as the second-largest segment which grows because more people depend on genomic sequencing and advanced imaging technologies.
- The market for molecular diagnostics will expand until 2026 because precision medicine shows continuous growth in its adoption.
- Systemic disease management remains dominant because it accounts for almost 50% of treatment usage in the Japan Erdheim Chester Disease Market.
- The Japan Erdheim Chester Disease Market operates under hospital networks and specialized tertiary care centers as its main distribution channel.
What are the Key Drivers, Restraints, and Opportunities in the Japan Erdheim Chester Disease Market?
The Japan Erdheim Chester Disease Market is primarily driven by the rapid integration of precision diagnostics into routine rare disease pathways. National rare disease funding frameworks together with enhanced genomic testing reimbursement have resulted in next-generation sequencing expansion across major Japanese tertiary hospitals. The new process allows earlier detection of MAPK pathway mutations especially for BRAF variants which need to be identified for selecting appropriate targeted therapies. The process enables treatment start times to become more precise which leads to greater acceptance of high-value biologics that result in increased earnings for specialty pharmaceutical companies.
A key restraint remains the structural scarcity of confirmed Erdheim Chester Disease cases combined with persistent diagnostic fragmentation. Advanced healthcare systems still experience diagnostic problems because patients with hematologic and autoimmune disorders show similar symptoms to these conditions. The limitation exists because the disease itself occurs at such low frequency which creates a need for official testing methods that only research institutions have developed. Treatment access to patients remains limited which creates obstacles to market growth while decreasing expected revenue growth.
The most promising opportunity exists because Japan is increasing its funding for centralized rare disease registries and AI-supported diagnostic imaging systems. The pilot programs currently active at Tokyo and Osaka university hospitals utilize machine learning models to analyze PET-CT data and identify histiocytic abnormalities at earlier stages. The nationwide implementation of this system will enable faster diagnosis processes which will result in earlier medical treatments and create a more organized data-based treatment network for Japan's Erdheim Chester Disease Market.
What Has the Impact of Artificial Intelligence Been on the Japan Erdheim Chester Disease Market?
The Japan Erdheim Chester Disease Market experiences transformation through artificial intelligence because the technology enables better diagnostic capabilities and advanced clinical decision-making systems for rare histiocytic disorders. The Japanese tertiary hospitals use machine learning algorithms to analyze PET-CT and MRI imaging which enables them to identify hidden tissue infiltration patterns that clinicians typically overlook during initial patient assessments. The AI-based imaging systems decrease the time needed for diagnosis while they increase the number of cases that hospitals identify which enables doctors to start MAPK-targeted treatments earlier and results in better patient outcomes.
The predictive analytics system uses advanced analytics models to analyze patient records from multiple healthcare facilities and predict how diseases will progress and which organs will become affected. The systems which combine genomic interpretation tools with existing systems enable clinicians to predict patient complications and select better treatment sequences for their patients which results in more effective long-term disease management. The hospitals that implement integrated AI systems experience improved diagnostic processes which lead to fewer instances of patients needing repeat imaging tests and this results in cost savings and quicker patient treatment times.
The lack of sufficient training data creates challenges for technology adoption because Erdheim Chester Disease cases occur at extremely low rates and display diverse characteristics which makes it difficult to develop accurate models for real-world applications. The existing hospital IT systems create obstacles for organizations that need to pay for AI system integration costs throughout their entire network. The current healthcare network in Japan benefits from AI technology which enhances precision and efficiency but the system faces obstacles for complete expansion throughout the country.
Key Market Trends
- BRAF and MEK-targeted precision oncology became the new standard treatment after Japan launched empirical steroid therapy in 2022 for Erdheim Chester Disease.
- The use of next-generation sequencing technology increased in Japanese hospitals between 2023 and 2026, which resulted in better diagnostic performance for uncommon histiocytic disorders.
- The hospital network expanded its AI-based PET-CT research systems, which enabled faster diagnosis and better detection of early-stage systemic disease lesions.
- The approval process for orphan drugs received stronger backing from regulatory authorities after 2023, which resulted in quicker approval ratings for targeted medications and greater availability of specialist treatments throughout the country.
- The development of patient identification systems received a boost from university hospitals, which established centralized rare disease registries that combined genomic data with imaging and clinical information in 2024.
- Roche and Novartis turned their focus to MAPK pathway inhibition clinical trials in Japan, which they conducted for investigational therapies targeting unique cancer types.
- The period from 2022 to 2025 saw treatment centers move towards multidisciplinary care models, which integrated oncology and neurology and cardiology expertise to treat systemic diseases.
- Rural healthcare facilities need better access to molecular diagnostics, which creates a gap between urban and rural areas in their ability to identify Erdheim Chester Disease during its initial stage.
- Digital pathology systems now operate in major hospitals, which allow faster biopsy analysis and decrease the need for additional testing in intricate situations.
Japan Erdheim Chester Disease Market Segmentation
By Type :
The most effective treatment method for this medical field relies on MAPK pathway inhibitors which doctors use to treat confirmed cases of Erdheim Chester Disease. Japanese tertiary hospitals now focus more on molecularly guided treatment which has decreased their use of general immunosuppressive methods. Doctors still use corticosteroids and chemotherapy as supportive treatments, which now represent a smaller portion of medical usage because doctors have developed better methods to treat patients.
The development of targeted therapy proceeds because more patients can access genomic testing and the orphan drug approvals system has improved since 2023. Doctors increasingly use combination therapy for treatment of refractory cases because single-agent treatment has failed to produce effective results in patients who experience multiple organ problems. The healthcare system is moving away from its practice of standardized treatment toward implementing personalized oncology treatment methods.
Japan will direct its future drug development efforts toward targeted therapies and combination treatments which companies are now testing in late-stage trials. Immunotherapy will achieve gradual expansion, but its development faces limits because disease-specific efficacy data remains scarce. Companies that develop mutation-targeted drugs together with their diagnostic tools will gain the most sustainable market power.

To learn more about this report, Download Free Sample Report
By Application :
The segment receives its primary focus from rare disease treatment because Erdheim Chester Disease operates as an ultra-orphan condition which requires treatment at Japanese hospitals that provide specialized medical services. The second-most significant treatment area exists within oncology because molecular oncology systems now identify the disease as histiocytic neoplasms. Academic hospitals drive clinical research growth through their studies which create evidence for effective rare disease treatment methods.
The expansion of genomic classification systems together with implementation of precision oncology protocols into rare disease patient pathways serves as the main factor behind business expansion. The increase in university hospitals participating in multicenter registries has resulted in better case identification together with more clinical trial participants. Early-stage assessments show immunological disorder applications face diagnostic overlap issues, which limit their usage in clinical settings.
Oncology and rare disease frameworks will develop stronger connections, which will enable researchers to convert their findings into therapies at an accelerated pace. Clinical research will become a key commercialization gateway for pharmaceutical companies entering Japan’s rare disease ecosystem. The process will decrease development times while researchers will gain better access to studies which validate targeted therapy methods.
By End-User :
Advanced imaging systems and genomic testing and multidisciplinary care teams are exclusive resources which hospitals and specialty centers use to dominate this segment. Academic institutes hold a strong secondary position as primary drivers of research and clinical trial activity. Clinics operate at a limited capacity because they need advanced diagnostic tools which only dedicated medical facilities can provide.
The centralized model of rare disease treatment in Japanian tertiary hospitals drives the expansion of rare disease medical services throughout the country. Specialty centers increasingly function as referral centers which treat complex histiocytic disorders that need both oncology and neurology expertise. Research institutes are expanding their participation in both rare disease databases and international research collaboration programs.
Academic hospitals will expand their future roles as primary centers which develop new treatment methods and diagnostic tests. The development of structured referral systems will enable specialty centers to attract more patients to their facilities. Hospital networks which control early diagnosis and treatment initiation will create partnership opportunities that benefit both investors and developers.
By Distribution :
The distribution process for hospitals operates through their pharmacies which handle high-priced targeted medicines that are used in inpatient and specialty care facilities. The market share of specialty pharmacies has increased from earlier times because Japan now offers more organized outpatient therapy programs that provide continuation treatment. The primary method for accessing rare disease medications remains direct hospital procurement which enables hospitals to obtain these medications. The market expansion results from growing demand for expensive biologic treatments which need complete medical monitoring and certified medication distribution systems. Specialty pharmacies are expanding their role in programs which help patients stick to their treatment plans and in systems which provide therapy at home. The distribution of safety-critical therapies through hospitals requires regulatory monitoring which makes this method the primary distribution method.
The specialty pharmacy network will expand its operations to handle more outpatient services which will become necessary for managing rare diseases. The combination of clinical complexities and monitoring needs will keep hospital pharmacies as the dominant service providers. Companies that produce pharmaceuticals must develop two sales strategies which will enable them to sell products to hospitals while maintaining patient treatment over extended periods.
What are the Key Use Cases Driving the Japan Erdheim Chester Disease Market?
The Japan Erdheim Chester Disease Market uses targeted therapy which genomic profiling supports as its primary treatment method. Hospitals choose MAPK pathway inhibitors because these drugs provide solutions for patients who have multiple organ problems while decreasing their chances of long-term disease advancement. Tertiary care oncology and neurology departments require this clinical precision because it creates a steady need for their services.
The use of clinical research and diagnostic imaging has grown into additional applications which academic institutes and specialty centers now use. Next-generation sequencing together with PET-CT integration enables earlier disease detection while assisting with patient group classification which leads to better trial participant selection and treatment development processes.
Specialty hospitals increasingly rely on these tools to refine complex case management. The hospital imaging systems now use AI-assisted rare disease screening which operates through registry-linked predictive analytics that track disease progression. Digital health infrastructure expansion has led to increased use of these solutions by university hospitals throughout Japan. The tools will enable diagnosis methods to progress from reactive identification toward proactive case detection throughout the forecast period.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 36.21 Million |
|
Market size value in 2026 |
USD 39.39 Million |
|
Revenue forecast in 2033 |
USD 70.99 Million |
|
Growth rate |
CAGR of 8.78% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Japan |
|
Key company profiled |
Novartis, Roche, Pfizer, Bristol Myers Squibb, Merck, AstraZeneca, GSK, Sanofi, Takeda, AbbVie, Amgen, Eli Lilly, Johnson & Johnson, Bayer, Teva |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Targeted Therapy, Chemotherapy, Immunotherapy, Corticosteroids, Combination Therapy, Others); By Application (Rare Disease Treatment, Oncology Treatment, Immunological Disorders, Clinical Research, Others); By End-User (Hospitals, Clinics, Research Institutes, Specialty Centers, Academic Institutes, Others); By Distribution (Hospital Pharmacies, Specialty Pharmacies, Others) |
Which Regions are Driving the Japan Erdheim Chester Disease Market Growth?
The Japan Erdheim Chester Disease Market exists in the Tokyo area together with its Kanto region because this area contains numerous tertiary hospitals and genomic research facilities and advanced diagnostic systems. The university hospitals in Tokyo have established routine rare disease testing procedures which combine next-generation sequencing with PET-CT imaging to boost detection rates for early-stage conditions. The regional advantages of government funding which supports rare disease research and the establishment of centralized referral networks create a strong base for the region. The region offers pharmaceutical companies the ability to locate their clinical trial sites which creates better access for patients to specialized treatments and their associated medical services.
The Kansai region provides stable secondary support because Osaka and Kyoto academic hospitals and their permanent oncology centers keep investing in their operations. The growth of Kansai depends on consistent hospital upgrades and permanent partnerships between local hospitals and international pharmaceutical companies which differ from Kanto's requirement for faster policy implementation. The healthcare systems in this region focus on maintaining patient care continuity through established referral systems which enable them to handle patient needs in regular operation. The region generates consistent revenue for Kansai because it maintains stable operations even when fewer diagnostic tests occur.
The Kyushu area has experienced rapid growth because new hospitals and government programs for rare diseases started operating in 2023. The establishment of new molecular diagnostic laboratories in Fukuoka together with increased national rare disease registry participation has resulted in better detection capabilities. The enhanced connections between regional hospitals and central research institutes have led to faster case reporting and treatment start times. The Kyushu market offers expansion possibilities for new businesses and investors because diagnostic testing becomes more common while clinical trial activities increase between 2026 and 2033.
Who are the Key Players in the Japan Erdheim Chester Disease Market and How Do They Compete?
The Japan Erdheim Chester Disease Market shows moderate concentration because multiple top companies exist in the market yet operate through specialized rare disease organizations which do not permit any single treatment to establish itself as the standard for medical use. The market develops through two key factors which include the development of precision oncology solutions that target MAPK pathways and the speed at which Japan approves orphan drugs through its regulatory system. Companies establish their competitive edge by integrating diagnostic tools into hospital networks and genomic testing systems instead of using price-based methods.
Roche combines its diagnostic tools with oncology systems to create a system that connects imaging data with molecular testing results. Novartis develops MEK inhibitors while establishing partnerships with Japanese academic hospitals to improve drug accessibility. Takeda uses its domestic rare disease networks to speed up patient recruitment while enhancing evidence development for histiocytic disorders.
Japanese hospitals conduct clinical trials with Pfizer which develops targeted oncology drugs through these research partnerships. Bristol Myers Squibb establishes its market position through its immunotherapy research on histiocytic cancers which enables it to develop new research programs. Sanofi establishes its competitive edge by expanding its rare disease treatment options and implementing structured access initiatives throughout hospital networks in the Asia-Pacific region.
Company List
- Novartis
- Roche
- Pfizer
- Bristol Myers Squibb
- Merck
- AstraZeneca
- GSK
- Sanofi
- Takeda
- AbbVie
- Amgen
- Eli Lilly
- Johnson & Johnson
- Bayer
- Teva
Recent Development News
In February 2026, Chugai Pharmaceutical Co., Ltd. launched ELEVIDYS following conditional approval and National Health Insurance reimbursement listing in Japan. While primarily indicated for Duchenne muscular dystrophy, this launch reinforces Japan’s accelerating framework for rare disease gene and regenerative therapies, which also governs access pathways for histiocytic disorders such as Erdheim–Chester Disease. Source https://www.chugai-pharm.co.jp/
In March 2026, Pharming Group N.V. received approval from Japan’s Ministry of Health, Labour and Welfare for Joenja (leniolisib) via its local partner OrphanPacific Inc. Although targeting APDS, this approval is significant for the rare immune-dysregulation segment overlapping mechanistically with histiocytic diseases like Erdheim–Chester Disease, particularly in PI3K pathway-targeted therapeutic development.Source https://www.globenewswire.com/
What Strategic Insights Define the Future of the Japan Erdheim Chester Disease Market?
The Japan Erdheim Chester Disease Market is developing into a precision-based system which employs genomic diagnostics and targeted oncology as its main methods for treating rare diseases. The next 5–7 years will experience expansion when hospitals start using next-generation sequencing as a standard procedure and when rare disease registries become fully integrated with clinical decision-support systems. The new approach will decrease the time needed for diagnosis while boosting the number of patients who receive treatment at early stages which will lead to higher demand for MAPK-targeted therapies and dedicated medical treatment facilities.
The medical system of Japan faces two serious threats which include overdependence on expensive targeted drugs and. The current healthcare system of Japan depends on urban tertiary centers as its main testing facilities which creates barriers for patient access to medical services throughout the entire country.
The emerging opportunity involves using AI-based rare disease registries which connect imaging data with genomic information and clinical records to create predictive tools that diagnose pre-symptomatic conditions. The university hospitals of Tokyo and Osaka are currently testing this capability through pilot programs. Market participants should focus on establishing partnerships with hospital networks while developing integrated diagnostic-therapeutic platforms to establish their presence in this data-centric specialized field.
Japan Erdheim Chester Disease Market Report Segmentation
By Type
- Targeted Therapy
- Chemotherapy
- Immunotherapy
- Corticosteroids
- Combination Therapy
By Application
- Rare Disease Treatment
- Oncology Treatment
- Immunological Disorders
- Clinical Research
By End-User
- Hospitals
- Clinics
- Research Institutes
- Specialty Centers
- Academic Institutes
By Distribution
- Hospital Pharmacies
- Specialty Pharmacies
Frequently Asked Questions
Find quick answers to common questions.
The Japan Erdheim Chester Disease Market size is USD 70.99 Million in 2033.
Key segments for the Japan Erdheim Chester Disease Market are By Type (Targeted Therapy, Chemotherapy, Immunotherapy, Corticosteroids, Combination Therapy, Others); By Application (Rare Disease Treatment, Oncology Treatment, Immunological Disorders, Clinical Research, Others); By End-User (Hospitals, Clinics, Research Institutes, Specialty Centers, Academic Institutes, Others); By Distribution (Hospital Pharmacies, Specialty Pharmacies, Others).
Major Japan Erdheim Chester Disease Market players are Novartis, Roche, Pfizer, Bristol Myers Squibb, Merck, AstraZeneca, GSK, Sanofi, Takeda, AbbVie, Amgen, Eli Lilly, Johnson & Johnson, Bayer, Teva.
The Japan Erdheim Chester Disease Market size is USD 36.21 Million in 2025.
The Japan Erdheim Chester Disease Market CAGR is 8.78% from 2026 to 2033.
- Novartis
- Roche
- Pfizer
- Bristol Myers Squibb
- Merck
- AstraZeneca
- GSK
- Sanofi
- Takeda
- AbbVie
- Amgen
- Eli Lilly
- Johnson & Johnson
- Bayer
- Teva
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033