Europe Medical, Legal, and Regulatory (MLR) Review Software Market Size & Forecast:
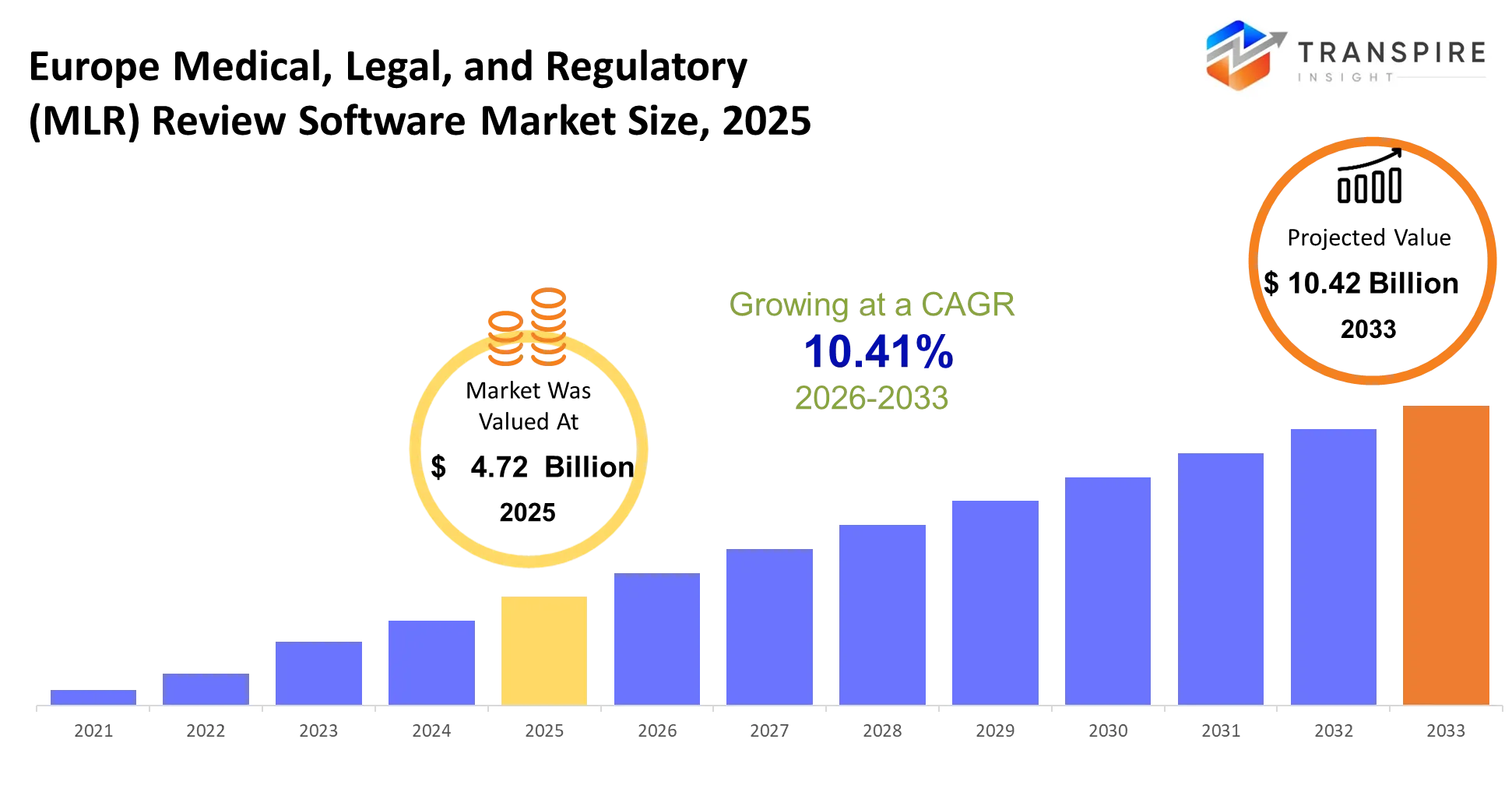
- Europe Medical, Legal, and Regulatory (MLR) Review Software Market Size 2025: USD 4.72 Billion
- Europe Medical, Legal, and Regulatory (MLR) Review Software Market Size 2033: USD 10.42 Billion
- Europe Medical, Legal, and Regulatory (MLR) Review Software Market CAGR: 10.41%
- Europe Medical, Legal, and Regulatory (MLR) Review Software Market Segments: By Type (Cloud-based, On-premise, Hybrid, Compliance Software, Others); By Application (Regulatory Compliance, Content Review, Risk Management, Medical Affairs, Legal Review, Others); By End-User (Pharma Companies, Biotech Firms, CROs, Regulatory Agencies, Healthcare Organizations, Others); By Deployment (SaaS, Private Cloud, Public Cloud, Hybrid Cloud, Others)
To learn more about this report, Download Free Sample Report
Europe Medical, Legal, and Regulatory (MLR) Review Software Market Summary:
The Europe Medical, Legal, and Regulatory (MLR) Review Software Market size is estimated at USD 4.72 Billion in 2025 and is anticipated to reach USD 10.42 Billion by 2033, growing at a CAGR of 10.41% from 2026 to 2033. The European medical legal and regulatory MLR review software market for healthcare and life sciences software is influenced by changing consumer preferences and European regulatory changes. Organizations will implement platforms that enable efficient review processes while enhancing teamwork between different departments and achieving required compliance standards. Automation technologies and secure cloud platforms and data validation solutions will transform organizational operations while European Union regulatory changes will determine how companies handle document review and approval and distribution processes to meet the new standards of document accuracy and speed and transparency that healthcare professionals require in Europe.
What Has the Impact of Artificial Intelligence Been on the Europe Medical, Legal, and Regulatory (MLR) Review Software Market?
Artificial intelligence is changing the European medical legal and regulatory MLR review software market for life sciences compliance because it enables faster document assessment and structured validation, and intelligent content checks. artificial intelligence in Europe medical legal, and regulatory (MLR) review software market supports teams in reducing manual review workloads while improving consistency across regulatory submissions. organizations use smart automation tools together with machine learning models to interpret complex guidelines which results in better approval cycle accuracy inside the Europe medical legal and regulatory MLR review software market.
The Europe medical legal and regulatory MLR review software market will use predictive analytics to identify compliance risks at an earlier stage while improving content development processes. ai-driven market trends analysis and intelligent automation improve operational efficiency while reducing delays in approvals. organizations can use machine learning models for better resource allocation and cost optimization because they need to respond quickly to changing regulatory requirements and documentation standards.
The Europe medical legal and regulatory MLR review software market will implement ai-driven systems that provide deeper customization of global compliance systems together with better compliance control in the future. this shift will strengthen innovation, improve accuracy, and enhance speed in regulated content management, positioning organizations to remain competitive in a rapidly changing regulatory environment.
Key Market Trends & Insights:
- The Western European market for medical legal and regulatory review software in Europe will reach a 42% market share by 2025 because of the strong pharmaceutical and biotechnological compliance systems.
- The medical legal and regulatory review software market in Europe expands its presence through increased adoption in Germany and the United Kingdom which benefits from the European Union's standardized regulatory framework.
- The medical legal and regulatory review software market in Eastern Europe will experience its fastest growth between 2025 and 2030 with a 11% annual growth rate because of the ongoing digital transformation within healthcare systems.
- The software platforms segment leads the market with a 55% market share because businesses choose central compliance management systems for their European medical and legal review operations.
- The second largest market segment for cloud-based deployment solutions is expanding because customers value its ability to grow and maintain secure partnership relationships.
- The medical legal and regulatory review software market in Europe experiences its highest growth through AI-powered regulatory automation tools which will continue to expand until 2030.
- The medical legal and regulatory review software market in Europe shows operational accuracy improvements through content approval and compliance tracking processes which lead the market with a 48% share.
- The European medical legal and regulatory review software market shows its fastest growth through predictive regulatory analytics which has become the leading application segment.
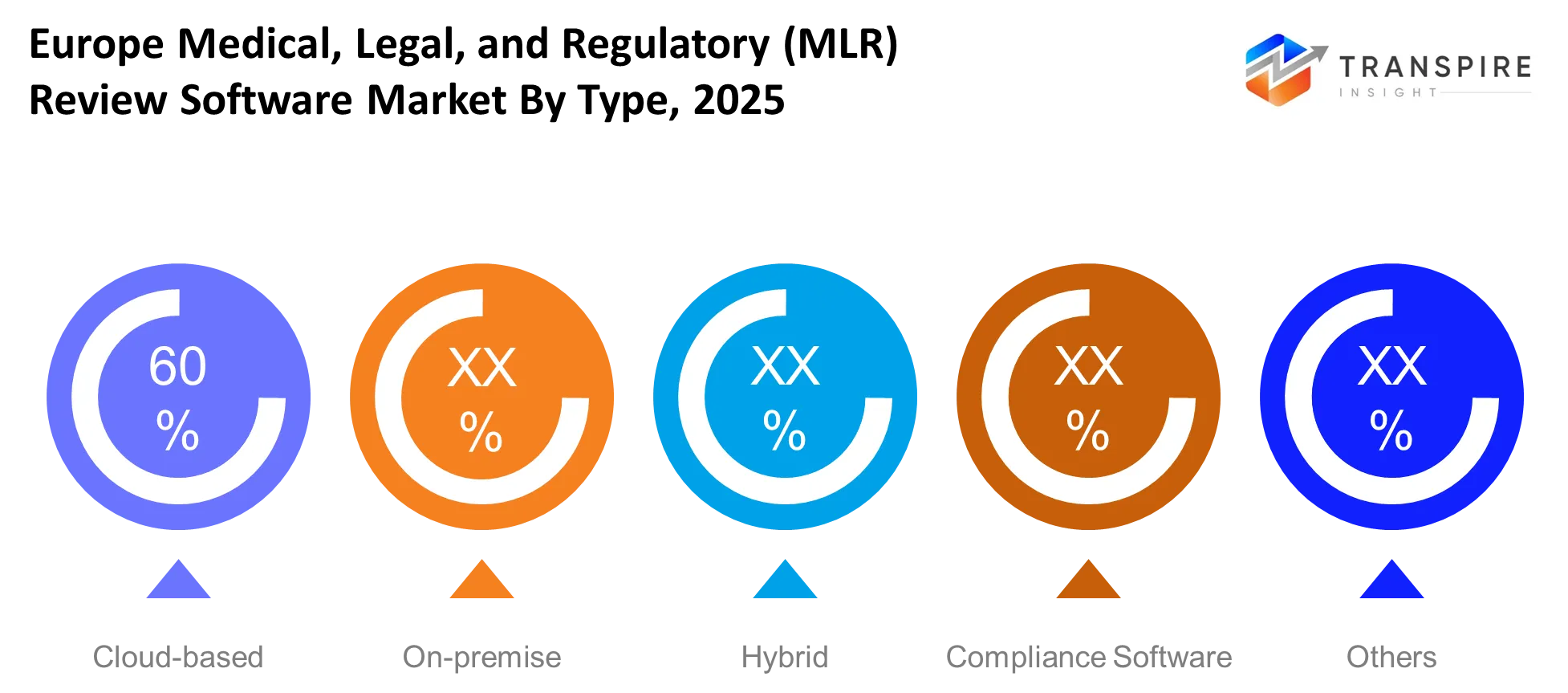
- The pharmaceutical sector constitutes the primary user base in the European medical legal and regulatory review software market because it holds a 60% market share while using structured approval systems.
- The main vendors in the market maintain their competitive edge through ongoing product development and their use of machine learning technology for automatic content verification.
Europe Medical, Legal, and Regulatory (MLR) Review Software Market Segmentation
By Type:
The Europe Medical Legal and Regulatory MLR Review Software Market provides three main software deployment options which include Cloud-based and On-premise and Hybrid deployment methods together with Compliance Software and other product types. Organizations that need to access regulatory content quickly while enabling multiple teams to work together will choose Cloud-based solutions as their primary option in the Europe Medical Legal and Regulatory MLR Review Software Market. Companies that need to maintain complete control over their internal data while using their existing systems will continue to use On-premise systems. Businesses will adopt Hybrid models which enable them to combine flexible operational methods with their security requirements during regulatory processing.
The Europe Medical Legal and Regulatory MLR Review Software Market uses compliance software together with specialized tools to deliver document validation and audit preparation capabilities. The tools will reduce the time needed for manual work during the document review process while they enhance the accuracy of the approval process. The need for different deployment methods will increase because organizations need to adapt their operations to new regulatory requirements which come with digital changes in European healthcare and life sciences.
To learn more about this report, Download Free Sample Report
By Application:
The Europe Medical Legal and Regulatory MLR Review Software Market provides six different application fields which include Regulatory Compliance and Content Review and Risk Management and Medical Affairs and Legal Review and other application areas. The Europe Medical Legal and Regulatory MLR Review Software Market will maintain its highest usage volume through regulatory compliance applications because European regulations demand precise document submission along with approval procedures. Content review systems enable faster material validation for medical and marketing purposes while they decrease faults in communication. Organizations will extend their use of risk management applications because they want to discover compliance deficiencies at earlier points within their operational processes.
The European Medical Legal and Regulatory (MLR) Review Software Market needs its medical affairs and legal review functions to boost scientific and legal team coordination. The applications will enhance document precision while they will shorten the time needed for approvals. Other supporting applications will continue to emerge as companies adopt integrated platforms to manage multiple regulatory functions inside one digital system.
By End-User:
The Europe Medical Legal and Regulatory (MLR) Review Software Market operates through Pharma Companies, Biotech Firms, CROs, Regulatory Agencies, Healthcare Organisations, and Other End-User Groups. The Europe Medical Legal and Regulatory (MLR) Review Software Market will see pharmaceutical companies lead adoption because they handle many regulatory submissions which require strict compliance. The documentation requirements for research will increase as innovation-driven research activity generates more documentation needs in the biotech industry. The CRO market will see increased adoption as European companies increasingly outsource their regulatory functions.
Digital review platforms will become operational in the Europe Medical Legal and Regulatory (MLR) Review Software Market because regulatory agencies and healthcare organizations use them to boost operational efficiency while increasing transparent processes. The consulting and support organizations will function as additional end-user groups that help with compliance documentation. Digital approval systems will become more important to all end-user groups which will lead to standardized workflow procedures across all categories.
By Deployment:
The European Medical Legal and Regulatory MLR Review Software Market will grow through SaaS deployment because it requires less infrastructural capacity and enables faster system setup. Private cloud solutions will remain important for organisations requiring high data protection and strict regulatory control. Public cloud adoption will increase as companies seek scalable and cost-effective solutions for large document volumes.
The Europe Medical Legal and Regulatory MLR Review Software Market will see increased adoption of hybrid cloud systems because organizations seek to achieve secure operations while maintaining system adaptability. Other deployment models will support niche regulatory requirements and customised workflows. Cloud-based deployment patterns will drive ongoing digital transformation in European compliance and regulatory review processes.
What are the Main Challenges for the Europe Medical, Legal, and Regulatory (MLR) Review Software Market Growth?
The European medical legal and regulatory MLR review software market faces ongoing operational and technical problems because organizations need to connect their systems with various compliance platforms. The system becomes unable to operate properly when users need to verify multiple regulatory documents at the same time while tracking their document changes. The European medical legal and regulatory MLR review software market experiences difficulties in achieving uninterrupted workflow automation because of its need to connect various enterprise systems which operate with different standards and its dependence on outdated systems which hinder its digital transformation efforts and decrease its processing power.
The European medical legal and regulatory MLR review software market faces manufacturing and commercialization obstacles which arise from its need to meet strict compliance standards and to handle high development expenses. Software solutions must align with complex regulatory frameworks, which increases time-to-market and slows product deployment. The European medical legal and regulatory MLR review software market experiences cost pressures which disrupt innovation processes while validation and approval delays make it hard to expand solutions throughout international pharmaceutical and biotech operations.
The Europe medical legal and regulatory MLR review software market faces major adoption hurdles because different regions have various levels of digital infrastructure and organizations lack enough technical specialists. The implementation of advanced compliance platforms faces difficulties because smaller organisations do not have enough qualified staff members who can operate these systems. The Europe medical legal and regulatory MLR review software market faces two major obstacles because funding shortages prevent emerging markets from acquiring modern regulatory technologies.
The Europe medical legal and regulatory MLR review software market faces increasing pressure from market competition and changing regulatory requirements because new automation solutions and AI-based compliance systems keep entering the market. The combination of pricing issues and ongoing regulatory changes creates unpredictable conditions for business operations. The combined challenges faced by the Europe medical legal and regulatory MLR review software market need ongoing changes to achieve operational stability which will support the market's capacity for future expansion.
Regional Insights
The Europe Medical Legal and Regulatory MLR Review Software Market Western Europe will maintain its status as the most advanced market because of its strong pharmaceutical industry and strict compliance requirements and early digital regulatory system implementation. Germany France and the United Kingdom will maintain their leadership position in software implementation because their healthcare systems are advanced and their professionals understand regulatory requirements. The Europe Medical Legal and Regulatory MLR Review Software Market in this region will benefit from consistent investment in compliance automation and secure data management systems that improve review accuracy and reduce approval delays.
The Northern European market for Europe Medical Legal and Regulatory MLR Review Software will experience steady growth because digital healthcare systems have developed to their current level and governments back innovative methods for regulatory process improvements. Nordic countries will focus on cloud-based platforms that improve collaboration between medical and legal teams. The European Medical Legal and Regulatory MLR Review Software Market will expand in Southern Europe because healthcare organizations will begin to modernize their regulatory systems through digital review tools which will replace their manual workflows.
The Eastern European market for Europe Medical Legal and Regulatory MLR Review Software will experience rapid growth because healthcare digitization efforts increase and demand for effective compliance solutions rises. The region will experience growth in software adoption because pharmaceutical companies will increase their outsourcing activities. The process of implementing projects will face delays because infrastructure development has not progressed equally and people cannot access state-of-the-art technologies.
Recent Development News
In March 2026, Veeva Systems reported its fiscal 2026 fourth quarter results, highlighting continued expansion in life sciences digital platforms that support medical, legal, and regulatory workflows across Europe. The company noted strong customer adoption of AI-enabled compliance tools and deeper integration of Vault applications used in regulated content review. The update reflected growing enterprise reliance on automated review systems within the Europe Medical, Legal, and Regulatory (MLR) Review Software Market as organisations aimed to reduce approval delays and improve compliance accuracy. https://ir.veeva.com
In April 2026, IQVIA expanded its collaboration ecosystem with software partners to strengthen data integration and AI-driven regulatory workflows used by pharmaceutical companies in Europe. The initiative focused on improving interoperability between analytics platforms and compliance review systems, supporting faster regulatory documentation and submission cycles. This development reinforced the growing shift toward connected digital ecosystems in the Europe Medical, Legal, and Regulatory (MLR) Review Software Market, where AI-assisted validation and cross-platform automation are becoming central to operational efficiency. https://www.iqvia.com
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 4.72 Billion |
|
Market size value in 2026 |
USD 5.21 Billion |
|
Revenue forecast in 2033 |
USD 10.42 Billion |
|
Growth rate |
CAGR of 10.41% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain, Rest of Europe) |
|
Key company profiled |
Veeva Systems, IQVIA, Oracle, Salesforce, SAP, IBM, Microsoft, MasterControl, Sparta Systems, ArisGlobal, Ennov, NAVEX Global, OpenText, Parexel, Medidata |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Cloud-based, On-premise, Hybrid, Compliance Software, Others); By Application (Regulatory Compliance, Content Review, Risk Management, Medical Affairs, Legal Review, Others); By End-User (Pharma Companies, Biotech Firms, CROs, Regulatory Agencies, Healthcare Organizations, Others); By Deployment (SaaS, Private Cloud, Public Cloud, Hybrid Cloud, Others) |
How Can New Companies Establish a Strong Foothold in the Europe Medical, Legal, and Regulatory (MLR) Review Software Market?
New companies entering the Europe medical, legal, and regulatory (MLR) review software market will need to focus on solving very specific compliance pain points instead of offering broad, generic platforms. Strong entry will come from targeting niche problems such as faster regulatory content approval, error-free document tracking, and simplified audit readiness. The Europe medical, legal, and regulatory (MLR) review software market will reward solutions that reduce manual review steps and improve accuracy in regulated industries like pharmaceuticals and biotech.
The Europe medical, legal and regulatory (MLR) review software market needs AI-based validation tools and secure cloud-based collaboration solutions for companies to establish market presence. Startups such as ReguloTech and MedProof AI, though emerging, show how intelligent document tagging and automated compliance checks can reduce approval cycles. The Europe medical, legal and regulatory (MLR) review software market needs platforms that work seamlessly with current enterprise systems while providing measurable productivity improvements for regulatory processes.
The European medical legal regulatory review software market will depend on partnership strategies to achieve its scaling goals. New market entrants will gain market credibility through their partnerships with pharmaceutical companies and CROs and regulatory consultants, which will help them develop their products according to actual user feedback. The European medical legal regulatory review software market will prefer vendors who develop technologies according to changing compliance requirements and data protection regulations.
The European medical legal regulatory review software market requires organizations to establish their market position through ongoing technological advancement and compliance with local regulations and distinct business operations built on automated systems and predictive analytics.
Key Europe Medical, Legal, and Regulatory (MLR) Review Software Market Company Insights
The European market for Medical, Legal, and Regulatory (MLR) Review Software will experience intense competition because businesses need quicker compliance processes together with systems for secure content verification. Veeva Systems, IQVIA, Oracle Health Sciences, MasterControl, and Freyr Solutions will maintain their leading market positions. The European market for Medical, Legal, and Regulatory (MLR) Review Software will experience competition because companies develop AI-powered review automation together with cloud-based solutions and real-time teamwork tools, which enable quicker approvals and decreased manual work for organizations that operate under government regulations.
New companies entering the European market for Medical, Legal, and Regulatory (MLR) Review Software will develop solutions for specific needs in regulatory content management to improve audit readiness and speed up validation processes. The competitive environment will intensify when companies introduce predictive analytics and machine learning solutions that enable more precise compliance evaluation. The European market for Medical, Legal, and Regulatory (MLR) Review Software will face competition from alternative digital compliance solutions which provide vendors with various pricing structures and modular system implementation options.
The European market for Medical, Legal, and Regulatory (MLR) Review Software will use strategic partnerships together with product development to establish its long-term market presence. Corporations will develop their cloud-native platforms through investments in AI-based decision support systems which will help them achieve market differentiation. The capacity to connect with pharmaceutical operations together with the ability to decrease regulatory hold-ups and handle multiple regional compliance requirements ensuring stronger adoption across Europe’s healthcare and life sciences sectors.
Company List
- Veeva Systems
- IQVIA
- Oracle
- Salesforce
- SAP
- IBM
- Microsoft
- MasterControl
- Sparta Systems
- ArisGlobal
- Ennov
- NAVEX Global
- OpenText
- Parexel
- Medidata
What are the Key Use-Cases Driving the Growth of the Europe Medical, Legal, and Regulatory (MLR) Review Software Market?
The market for medical legal and regulatory review software in Europe is experiencing growth because pharmaceutical and life sciences companies use structured compliance workflows to achieve faster approval of their content. The automated examination of advertising and medical documents serves as a primary application because it requires both precise results and compliance with legal requirements. The European regulatory system, which establishes standards for content publication, gets supported by the European medical, legal and regulatory review software market that helps organizations decrease manual mistakes.
The European medical, legal and regulatory review software market provides its main purpose through regulatory submission management, which helps businesses process their document approval needs across multiple European agencies. This process establishes better version control, while simultaneously decreasing duplicate work, and it enhances the ability of legal, medical, and compliance teams to work together. The European medical, legal, and regulatory review software market functions as a vital component of pharmacovigilance documentation because it enables the swift and precise processing of safety updates.
Healthcare organizations, as well as contract research organizations, utilize these platforms to manage their risks while preparing for audits. The European medical, legal, and regulatory review software market will keep growing because predictive analytics and artificial intelligence validation tools help detect compliance violations at an early stage. These abilities help businesses operate more effectively while building confidence in their European regulatory procedures.
The upcoming applications of European medical, legal and regulatory review software will depend on deeper system connections with enterprise software that provides real time teamwork capabilities and worldwide compliance solutions for healthcare systems.
Europe Medical, Legal, and Regulatory (MLR) Review Software Market Report Segmentation
By Type
- Cloud-based
- On-premise
- Hybrid
- Compliance Software
- Others
By Application
- Regulatory Compliance
- Content Review
- Risk Management
- Medical Affairs
- Legal Review
- Others
By End-User
- Pharma Companies
- Biotech Firms
- CROs
- Regulatory Agencies
- Healthcare Organizations
- Others
By Deployment
- SaaS
- Private Cloud
- Public Cloud
- Hybrid Cloud
- Others
Frequently Asked Questions
Find quick answers to common questions.
The approximate Europe Medical, Legal, and Regulatory (MLR) Review Software Market size for the market will be USD 10.42 Billion in 2033.
Key segments for the Europe Medical, Legal, and Regulatory (MLR) Review Software Market are By Type (Cloud-based, On-premise, Hybrid, Compliance Software, Others); By Application (Regulatory Compliance, Content Review, Risk Management, Medical Affairs, Legal Review, Others); By End-User (Pharma Companies, Biotech Firms, CROs, Regulatory Agencies, Healthcare Organizations, Others); By Deployment (SaaS, Private Cloud, Public Cloud, Hybrid Cloud, Others).
Major Europe Medical, Legal, and Regulatory (MLR) Review Software Market players are Veeva Systems, IQVIA, Oracle, Salesforce, SAP, IBM, Microsoft, MasterControl, Sparta Systems, ArisGlobal, Ennov, NAVEX Global, OpenText, Parexel, Medidata.
The Europe Medical, Legal, and Regulatory (MLR) Review Software Market size is USD 4.72 Billion in 2025.
The Europe Medical, Legal, and Regulatory (MLR) Review Software Market CAGR is 10.41%.
- Veeva Systems
- IQVIA
- Oracle
- Salesforce
- SAP
- IBM
- Microsoft
- MasterControl
- Sparta Systems
- ArisGlobal
- Ennov
- NAVEX Global
- OpenText
- Parexel
- Medidata
Recently Published Reports
-
Apr 2026
3D Optical Profiler Market
3D Optical Profiler Market Size, Share & Analysis Report By Type (Desktop 3D Optical Profiler, and Portable 3D Optical Profiler), By Technology (Confocal Technology, and White Light Interference), By End-Use Industry (Manufacturing, Research Institutions, Automotive, Aerospace and Defense, Medical Devices, and Other), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Depth Sensor Market
Depth Sensor Market Size, Share & Analysis Report By Type (Infrared Depth Sensors, Time-of-Flight (ToF) Sensors, Stereo Vision Sensors, Structured Light Sensors, Ultrasonic Depth Sensors), By Application (Automotive, Robotics, Gaming, Consumer Electronics, Industrial Automation, Healthcare, Security & Surveillance, Others), By End Users (Automotive Manufacturers, Consumer Electronics Companies, Healthcare Providers, Industrial Companies, Security Agencies, Gaming Companies, Robotics Companies, Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Digital Manufacturing Market
Digital Manufacturing Market Size, Share & Analysis Report By Component (Hardware, Software, and Services), By Technology (Robotics, 3D Printing, Internet of Things (IoT), and Others), By Application (Automotive and Transportation, Aerospace and Defense, Consumer Electronics, Industrial Machinery, and Others), By Process Type (Computer-Based Designing, Computer-Based Simulation, Computer 3D Visualization, Analytics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Digital Visa Services Market
Digital Visa Services Market Size, Share & Analysis Report By Type (Individual Travelers, Group Travelers), By Application (Tourism, Business Travel, Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031


