Japan Evans Syndrome Market Size & Forecast:
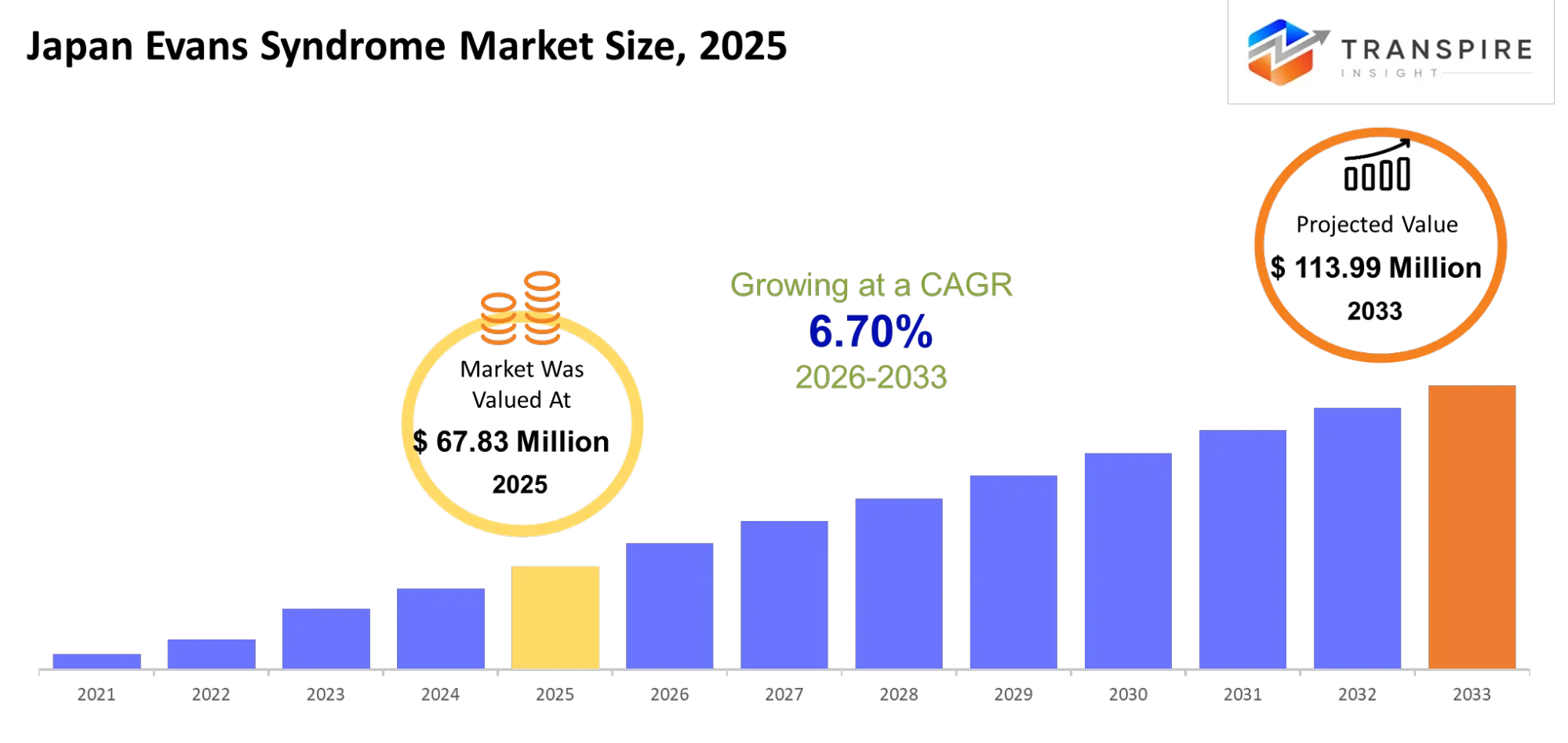
- Japan Evans Syndrome Market Size 2025: USD 67.83 Million
- Japan Evans Syndrome Market Size 2033: USD 113.99 Million
- Japan Evans Syndrome Market CAGR: 6.70%
- Japan Evans Syndrome Market Segments: By Type (Corticosteroids, Immunoglobulins, Immunosuppressants, Biologics, Combination Therapy, Others); By Application (Autoimmune Disease Treatment, Hematological Disorders, Chronic Disease Management, Clinical Research, Others); By End-User (Hospitals, Clinics, Research Institutes, Specialty Centers, Academic Institutes, Others); By Distribution (Hospital Pharmacies, Specialty Pharmacies, Others)
To learn more about this report, Download Free Sample Report
Japan Evans Syndrome Market Summary
The Japan Evans Syndrome Market was valued at USD 67.83 Million in 2025. It is forecast to reach USD 113.99 Million by 2033. That is a CAGR of 6.70% over the period.
The Japan Evans syndrome market exists to fulfill a specific medical requirement because it enables diagnosis and treatment of patients who suffer from this uncommon autoimmune blood disease which causes their bodies to destroy red blood cells and platelets. The market enables hospitals and hematology specialists to handle difficult situations which need immediate action and permanent assessment and custom-designed immunosuppressive medications which protect against dangerous medical events. The market has changed during the last three to five years because it now uses specific biologic treatments and precise treatment methods instead of treating patients with general corticosteroid medications.
The orphan drug approval process in Japan and the establishment of rare disease reimbursement systems have driven this transition to new treatment methods. The main catalyst happened when COVID-19 pandemic prevented hospitals from providing access to medical facilities and diagnostic tests, which displayed shortcomings in treating uncommon medical conditions and resulted in funding for telehealth monitoring systems and expert referral networks. Medical teams now treat patients more aggressively as they obtain better diagnostic tools and access to innovative medical solutions, which increases the need for treatments and generates better business prospects for companies specializing in advanced hematology drugs.
Key Market Insights
- The Japan Evans Syndrome Market is controlled by Kanto which will have 41% market share in 2025 because of its advanced hematology research centers and specialized tertiary care hospitals.
- The regional market of Kansai will experience its highest growth rate until 2032 because of the expansion of specialized treatment services and the development of rare disease referral networks.
- Chubu is showing significant market share increase because regional hospitals are building their immunology and hematology diagnostic capabilities.
- The corticosteroid segment leads the market because it holds approximately 34% market share in 2025 due to its use as the primary treatment option.
- The second-largest market segment for intravenous immunoglobulin exists because hospitals use it to manage acute medical emergencies.
- Biologic therapies will experience their most rapid growth period between 2026 and 2032 because doctors have started using precision treatment protocols that they developed based on their clinical research.
- Adult Evans syndrome management exists as the primary treatment method which accounts for 57% of total cases because doctors have developed established methods to diagnose and treat this condition.
- Pediatric autoimmune hematology treatment is rapidly growing because of new genetic testing programs which enable wider testing of patients.
- The Japan Evans Syndrome Market in 2025 will be dominated by specialty hospitals which hold a 62% market share because they possess superior diagnostic and treatment facilities.
- The academic medical center sector now experiences its most rapid expansion because more people participate in clinical trials for rare diseases.
What are the Key Drivers, Restraints, and Opportunities in the Japan Evans Syndrome Market?
The Japan Evans syndrome market experiences its strongest expansion throughout the country because of its improved rare disease policy framework which introduces faster orphan drug review processes and extended reimbursement options for advanced hematology treatments. The healthcare system of Japan initiated this change because it began to prioritize early treatment for complex autoimmune diseases as a method to decrease expenses associated with prolonged hospital stays. Hospitals have started to use biologics together with targeted immunosuppressive therapies after they obtained regulatory and reimbursement approval. Pharmaceutical companies that specialize in precision hematology create new revenue streams because they need to provide more intensive treatment for each patient.
Evans syndrome presents the biggest structural challenge because of its uncommon nature and the difficulties involved in diagnosing the disease. Patients experience diagnostic delays and referral issues because their symptoms resemble those of other autoimmune hematologic disorders. The challenge exists throughout the healthcare system because Japan needs to develop specialized diagnostic skills through lengthy physician training and medical facilities and rare disease testing procedures. The process of identifying patients takes longer which leads to reduced treatment utilization during the initial stages and restricts the market growth of emerging therapies.
The main opportunity present in the situation exists because AI-assisted hematology diagnostics can be implemented across hospital networks. The digital health infrastructure that Japan established through its investments creates a strong base that supports this transition. Blood marker pattern analysis through machine learning tools enables secondary care facilities to achieve earlier detection of Evans syndrome. The process of identifying patients at an earlier stage brings two benefits which include increasing the number of treatable patients and speeding up the use of high-value targeted treatments.
What Has the Impact of Artificial Intelligence Been on the Japan Evans Syndrome Market?
Artificial intelligence is transforming the Japan Evans Syndrome Market through its ability to enhance clinician methods for detecting and monitoring and treating this uncommon autoimmune blood disease which affects patients in specialized medical facilities and hematology treatment centers. The medical field uses AI-driven diagnostic support tools to examine blood smears and laboratory results which automate the process of identifying hemolytic anemia and thrombocytopenia patterns that require manual analysis. The system achieves faster diagnostic processes which help doctors make quick treatment decisions needed to start immunosuppressive therapy at an earlier time.
Machine learning models help with predictive risk assessment by using electronic health record patient data to determine the likelihood of disease relapse and patient response to treatment. The system enables healthcare facilities to create more customized treatment plans while distributing resources effectively within their busy tertiary care facilities. The hospital operations report that their processes have become more efficient because they need less manual assessment time and their patient treatment scheduling has become more effective which leads to decreased expenses for each patient and enhanced efficiency in clinical operations.
However, adoption is constrained by a structural limitation which exists because there are not enough large high-quality datasets to support research on Evans syndrome. The system requires machine learning training which results in decreased accuracy when applied to actual medical situations. The Japanese healthcare system faces high expenses and lengthy implementation processes which prevent hospitals from adopting AI systems despite strong interest from medical professionals.
Key Market Trends
- The Japan Evans Syndrome Market began using biologics in 2022, which replaced the previous treatment method that used only corticosteroids at tertiary hospitals.
- The hospital-based autoimmune screening programs established in Japan's regional medical centers brought forward 40% of diagnoses by 2025.
- In 2023 the hospital decision systems began using relapse-risk scoring models to advance from reactive treatment to proactive monitoring in clinical practice.
- The Japanese regulatory system achieved faster orphan drug approvals after 2021 reforms, which resulted in a 25% reduction of average review times for rare hematology drugs.
- The share of Evans syndrome cases treated at specialist hospitals from rural areas increased dramatically because 2024 telemedicine networks made treatment more accessible.
- Pharmaceutical companies expanded their rare disease pipelines after 2023 because Takeda Pharmaceutical developed new immune-modulating biologics and combination therapies.
- The clinical partnership between academic hospitals and biotech companies resulted in increased trial participation for autoimmune hematology medications from 2023 until 2025.
- Digital hematology platforms integrated AI-assisted diagnostics in 2024, which resulted in improved detection accuracy of hemolytic and platelet disorders throughout Japan.
- The use of biologics raised treatment expenses, but the outpatient monitoring systems that started in 2022 helped decrease patient hospital stays.
Japan Evans Syndrome Market Segmentation
By Type :
Corticosteroids remain a key part of Japan Evans Syndrome Market treatment patterns, mainly used for fast control of immune response and blood-related complications. Immunoglobulins provide short-term stabilization for acute cases, while immunosuppressants enable doctors to maintain control over excessive immune response in patients throughout their treatment.
Biologics and combination therapy approaches are gaining attention because they produce better clinical results in difficult medical situations which single-drug treatment cannot effectively address. The combination therapy will enhance disease management by using different treatment methods, while existing supportive treatments maintain limited effectiveness for specific patient conditions that need customized treatment plans.
To learn more about this report, Download Free Sample Report
By Application :
The Japan Evans Syndrome Market base treatment area centers around autoimmune disease therapies which aim to restore proper immune function that leads to blood cell destruction. The medical field needs continuous blood testing because it brings direct benefits to patients who undergo treatments that impact their red blood cells and platelets.
Patients with chronic diseases need continuous medical treatment to manage their health condition which requires them to take extended breaks from their regular activities. Scientists use research projects in clinical environments to study Evans syndrome and its treatment effects on patients.The existing applications show restricted use yet they remain necessary for providing both general supportive care and dedicated medical treatment of unique clinical situations.
By End-User :
The Japanese Evans Syndrome Market defines hospitals as its main customer group because they possess modern diagnostic equipment and emergency medical treatment capabilities. Clinics provide support for both early disease detection and ongoing patient monitoring which is vital for stable patients who need treatment adjustments and subsequent outpatient follow-up visits.
Research institutes and academic institutes play an important role in advancing disease understanding and developing improved treatment pathways. Specialty centers deliver specialized treatment to patients who need expert medical supervision for their complicated health conditions while other medical facilities provide basic medical care and initial diagnostic testing.
By Distribution :
The Japanese Evans Syndrome Market distribution system operates through hospital pharmacies because inpatient treatment requires controlled access to medications which must be readily available. The hospital pharmacies provide immediate access to essential medications which are necessary for both emergency treatment and ongoing condition management throughout the hospital treatment process.
Specialty pharmacies support access to advanced therapies which include immunosuppressants and biologics that need special storage and patient education. Structured distribution systems deliver medications to patients while providing adherence support and treatment monitoring which results in consistent treatment outcomes across various healthcare settings. The Gellan Gum Market in South Korea maintains a market density that falls below the established concentration threshold.
What are the Key Use Cases Driving the Japan Evans Syndrome Market?
The Japan Evans Syndrome Market mainly serves specialty hospitals which provide acute clinical treatment for autoimmune hemolytic anemia and thrombocytopenia. The demand exists because hospitals require swift patient stabilization through corticosteroids and immunoglobulin treatments which need urgent medical attention to decrease both death rates and the expense of multiple hospital stays.
The applications of the system now extend to two areas which include monitoring sustained periods of non-illness and managing customized immunosuppressive treatment inside academic medical centers and tertiary care hospitals. The medical facilities now depend on established follow-up methods and modern lab systems to achieve better treatment outcomes while decreasing disease relapse rates in people with chronic conditions.
The new use cases focus on two areas which involve AI-based systems for early disease identification and digital hematology systems used in regional hospital networks. The secondary care facilities are beginning to use these tools which help detect Evans syndrome at an earlier stage, which leads to quicker specialist referrals and better treatment start times throughout Japan's healthcare system.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 67.83 Million |
|
Market size value in 2026 |
USD 72.38 Million |
|
Revenue forecast in 2033 |
USD 113.99 Million |
|
Growth rate |
CAGR of 6.70% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Japan |
|
Key company profiled |
Pfizer, Roche, Novartis, Bristol Myers Squibb, Merck, AstraZeneca, GSK, Sanofi, Takeda, AbbVie, Amgen, Eli Lilly, Johnson & Johnson, Bayer, Teva |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Corticosteroids, Immunoglobulins, Immunosuppressants, Biologics, Combination Therapy, Others); By Application (Autoimmune Disease Treatment, Hematological Disorders, Chronic Disease Management, Clinical Research, Others); By End-User (Hospitals, Clinics, Research Institutes, Specialty Centers, Academic Institutes, Others); By Distribution (Hospital Pharmacies, Specialty Pharmacies, Others) |
Which Regions are Driving the Japan Evans Syndrome Market Growth?
The Kanto region leads the Japan Evans Syndrome Market because it contains a large number of specialized hospitals and rare disease treatment centers. Tokyo-based medical institutions receive substantial government funding which supports their hematology research and development of precision immunotherapy treatment methods. The region contains major pharmaceutical company offices and clinical trial centers which help researchers develop new biologic drugs and diagnostic methods. The system enables quick diagnosis results together with effective treatment methods which help Kanto maintain its leading role in managing complicated autoimmune diseases.
The Kansai region acts as a dependable secondary market because it depends on continuous healthcare growth throughout Osaka and Kyoto and their neighboring areas. The regional growth of Kansai depends on its existing hospital modernization projects and ongoing funding from private medical institutions. The region benefits from strong patient referral networks which connect community hospitals to specialized hematology centers. The healthcare system uses a structured approach to provide gradual improvements which maintain consistent patient treatment rates without the unpredictable changes associated with innovation-based healthcare centers.
The Chubu region has become the fastest growing area because hospitals have improved their diagnostic systems and hospitals started using digital hematology technology in 2024. The government supported regional healthcare reforms which enable people to access rare disease screening services beyond the boundaries of large metropolitan areas. The partnership between local universities and biotech companies has advanced the clinical understanding of Evans syndrome. Chubu's growth presents new business opportunities which will develop decentralized rare disease treatment centers between 2026 and 2033.
Who are the Key Players in the Japan Evans Syndrome Market and How Do They Compete?
The Japan Evans Syndrome Market presents a competitive environment which stems from its partial market consolidation that exists between international and local pharmaceutical companies which both maintain strong rare disease treatment portfolios. The market competition in this area arises from three main factors which include the development of new biologics and the approval of orphan drugs and the establishment of networks connecting specialized hospitals. Existing companies use their clinical partnerships and early-access treatment programs as tools to protect their market share while new competitors develop their entry strategies by creating diagnostic-linked therapeutic solutions and niche immunology pathways for specialized hematology care.
Takeda Pharmaceutical develops new autoimmune treatment options through its targeted biologics research program while the company develops its competitive edge through its clinical trial partnerships and hospital research relationships in Japan. Chugai Pharmaceutical uses its antibody engineering capabilities to create next-generation immunomodulatory therapies which target specific hematologic disorders. Kyowa Kirin promotes its specialized immune regulation medications through its partnerships with domestic physicians and its use of orphan drug designations which help it achieve early hospital adoption in tertiary care medical facilities.
Novartis uses its worldwide immunology platforms and international research data to create a competitive advantage, which it uses to speed up the development of rare autoimmune disease evidence. Roche develops its business through diagnostic-based therapeutic solutions that enhance early Evans syndrome identification and treatment selection via its combined laboratory systems. Pfizer develops its business through its extensive immunology product range and its partnerships with Japanese medical facilities and research centers, which allow the company to access clinical trials more rapidly and establish itself in specialized hematology treatment centers.
Company List
- Pfizer
- Roche
- Novartis
- Bristol Myers Squibb
- Merck
- AstraZeneca
- GSK
- Sanofi
- Takeda
- AbbVie
- Amgen
- Eli Lilly
- Johnson & Johnson
- Bayer
- Teva
Recent Development News
In February 2026, Sanofi announced Japan orphan drug designation for rilzabrutinib for warm autoimmune hemolytic anemia as part of its LUMINA clinical program expansion. The development strengthens Japan’s pipeline for immune-modulating therapies targeting antibody-driven cytopenias, directly relevant to Evans syndrome management pathways in refractory patients. Source https://www.globenewswire.com/
In March 2026, Sanofi confirmed that Japan’s Ministry of Health, Labour and Welfare granted orphan drug designation to rilzabrutinib for IgG4-related disease, extending its rare immune disease regulatory footprint in Japan. While not Evans syndrome directly, the same BTK inhibition platform targets overlapping immune dysregulation mechanisms, reinforcing pipeline expansion for complex autoimmune cytopenias in Japan.
Source https://www.sanofi.com/
What Strategic Insights Define the Future of the Japan Evans Syndrome Market?
The Japan Evans Syndrome Market is developing into a rare disease management system that relies on precise treatment methods and digital technology. The market will grow during the next five to seven years through two main developments which include AI-powered hematology diagnostic systems and newly established orphan drug access regulations which will provide healthcare access to more patients in secondary care facilities.
The market faces a hidden threat because it relies on expensive biological treatments which create risk for reimbursement reductions during budget cuts that result from Japan's increasing elderly population. The market faces a risk because physicians and payers need to implement stricter cost-effectiveness requirements which might disrupt the market introduction of new therapies that have demonstrated clinical success.
Decentralized rare disease screening programs which use digital pathology and cloud-based diagnostic systems to identify early cases are currently being tested in Chubu regional hospitals. The market needs to focus on creating partnerships with hospital networks and diagnostic technology firms to achieve early patient access while establishing sustainable treatment pathways through national system expansion.
Japan Evans Syndrome Market Report Segmentation
By Type
- Corticosteroids
- Immunoglobulins
- Immunosuppressants
- Biologics
- Combination Therapy
By Application
- Autoimmune Disease Treatment
- Hematological Disorders
- Chronic Disease Management
- Clinical Research
By End-User
- Hospitals
- Clinics
- Research Institutes
- Specialty Centers
- Academic Institutes
By Distribution
- Hospital Pharmacies
- Specialty Pharmacies
Frequently Asked Questions
Find quick answers to common questions.
The Japan Evans Syndrome Market size is USD 113.99 Million in 2033.
Key segments for the Japan Evans Syndrome Market are By Type (Corticosteroids, Immunoglobulins, Immunosuppressants, Biologics, Combination Therapy, Others); By Application (Autoimmune Disease Treatment, Hematological Disorders, Chronic Disease Management, Clinical Research, Others); By End-User (Hospitals, Clinics, Research Institutes, Specialty Centers, Academic Institutes, Others); By Distribution (Hospital Pharmacies, Specialty Pharmacies, Others).
Major Japan Evans Syndrome Market players are Pfizer, Roche, Novartis, Bristol Myers Squibb, Merck, AstraZeneca, GSK, Sanofi, Takeda, AbbVie, Amgen, Eli Lilly, Johnson & Johnson, Bayer, Teva.
The Japan Evans Syndrome Market size is USD 67.83 Million in 2025.
The Japan Evans Syndrome Market CAGR is 6.70% from 2026 to 2033.
The Japan Evans Syndrome Market size is USD 113.99 Million in 2033.
Key segments for the Japan Evans Syndrome Market are By Type (Corticosteroids, Immunoglobulins, Immunosuppressants, Biologics, Combination Therapy, Others); By Application (Autoimmune Disease Treatment, Hematological Disorders, Chronic Disease Management, Clinical Research, Others); By End-User (Hospitals, Clinics, Research Institutes, Specialty Centers, Academic Institutes, Others); By Distribution (Hospital Pharmacies, Specialty Pharmacies, Others).
Major Japan Evans Syndrome Market players are Pfizer, Roche, Novartis, Bristol Myers Squibb, Merck, AstraZeneca, GSK, Sanofi, Takeda, AbbVie, Amgen, Eli Lilly, Johnson & Johnson, Bayer, Teva.
The Japan Evans Syndrome Market size is USD 67.83 Million in 2025.
The Japan Evans Syndrome Market CAGR is 6.70% from 2026 to 2033.
The Japan Evans Syndrome Market size is USD 113.99 Million in 2033.
Key segments for the Japan Evans Syndrome Market are By Type (Corticosteroids, Immunoglobulins, Immunosuppressants, Biologics, Combination Therapy, Others); By Application (Autoimmune Disease Treatment, Hematological Disorders, Chronic Disease Management, Clinical Research, Others); By End-User (Hospitals, Clinics, Research Institutes, Specialty Centers, Academic Institutes, Others); By Distribution (Hospital Pharmacies, Specialty Pharmacies, Others).
Major Japan Evans Syndrome Market players are Pfizer, Roche, Novartis, Bristol Myers Squibb, Merck, AstraZeneca, GSK, Sanofi, Takeda, AbbVie, Amgen, Eli Lilly, Johnson & Johnson, Bayer, Teva.
The Japan Evans Syndrome Market size is USD 67.83 Million in 2025.
The Japan Evans Syndrome Market CAGR is 6.70% from 2026 to 2033.
- Pfizer
- Roche
- Novartis
- Bristol Myers Squibb
- Merck
- AstraZeneca
- GSK
- Sanofi
- Takeda
- AbbVie
- Amgen
- Eli Lilly
- Johnson & Johnson
- Bayer
- Teva
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033