Japan Biopharmaceutical Market Size & Forecast:
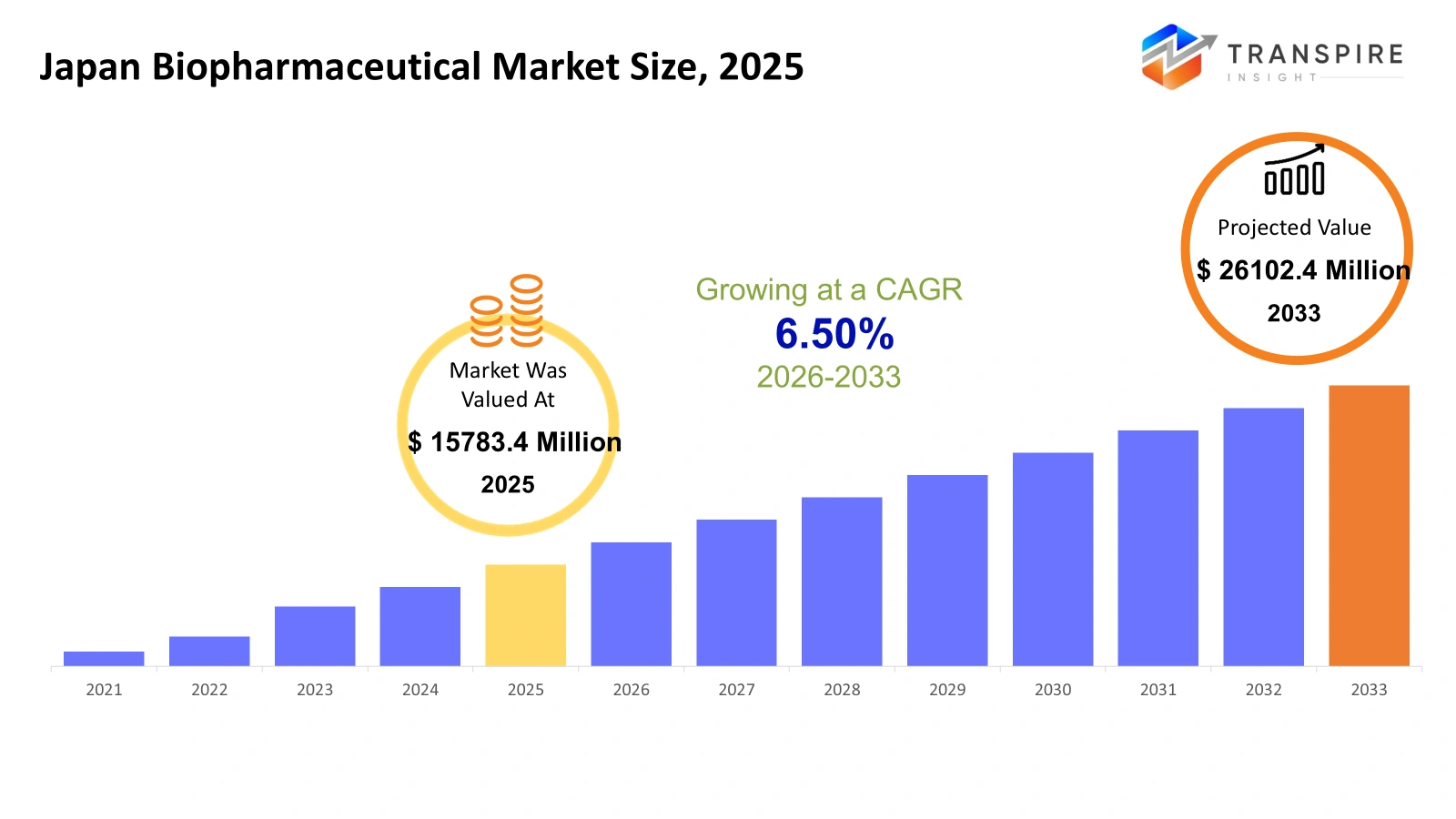
- Japan Biopharmaceutical Market Size 2025: USD 15783.4 Million
- Japan Biopharmaceutical Market Size 2033: USD 26102.4 Million
- Japan Biopharmaceutical Market CAGR: 6.50%
- Japan Biopharmaceutical Market Segments: By Product Type (Monoclonal Antibodies, Vaccines, Recombinant Proteins, Cell & Gene Therapies), By Application (Oncology, Autoimmune Diseases, Infectious Diseases, Metabolic Disorders).
To learn more about this report, Download Free Sample Report
Japan Biopharmaceutical Market Summary:
The Japan biopharmaceutical market size is estimated at USD 15783.4 Million in 2025 and is anticipated to reach USD 26102.4 Million by 2033, growing at a CAGR of 6.50% from 2026 to 2033. Japan's biopharmaceutical industry experiences fast development because of three factors, which include its ageing population and increased need for sophisticated medical treatments and government backing of innovation. Companies are investing heavily in biologics and cell and gene therapies and precision medicine to address complex diseases. Domestic companies and international partners work together to enhance research and development processes. The implementation of regulatory reforms creates a faster process for drug approvals. Japan has become a major centre for advanced biopharmaceutical research throughout Asia.
Key Market Trends & Insights:
- The growing number of elderly people in Japan requires new advanced medical treatments for patients with persistent health problems and diseases that develop with age. This situation needs new biological medical products and extended-duration medical treatments.
- The current system provides companies with better ways to develop products through its dual process of streamlined approval paths and flexible regulatory requirements. The government programmes help organisations establish new medical treatments in the market at faster rates.
- Regional businesses now establish partnerships with international biopharmaceutical companies to exchange knowledge and equipment. The research process benefits from these partnerships because they help scientists discover new information and create international connections.
- The medical field shows increased interest in developing customised therapies that use patients' genetic data. This medical method leads to better patient results while decreasing the probability of unwanted medical reactions.
- The public sector, together with the private sector, currently allocates substantial financial resources toward research and development efforts. The ongoing financial support enables Japan to develop innovative technologies which will enhance its global competitiveness.
- The combination of artificial intelligence with data analysis techniques leads to better outcomes in both drug development processes and clinical research operations. The digital tools improve both patient treatment processes and organisational operation effectiveness.
Japan Biopharmaceutical Market Segmentation
By Product Type
- Monoclonal Antibodies: The Japanese biopharmaceutical market uses monoclonal antibodies because these products provide targeted treatment which achieves high success rates. The treatment approach backs their widespread application as an effective solution for both cancer and autoimmune diseases. Healthcare organisations across the country adopt these technologies because of their ongoing research development and their successful clinical outcomes.
- Vaccines: Vaccines function as an essential healthcare category because the Japanese government emphasises preventive medical services. The market expands because people understand vaccines better, the government operates immunisation campaigns, and vaccine technology improves. Recent global health issues made people demand vaccines, which led to more local vaccine production and accelerated new vaccine development.
- Recombinant Proteins: Proteins purified from the bodies of other organisms could be used as medicine. The treatment method provides reliable results, which makes it an important component of present-day medical practices. Japanese researchers conduct studies while manufacturers develop better production methods to make their products more effective and create new medical uses.
- Cell & Gene Therapies: The Japanese government provides active support for regenerative medicine, which causes this field to attract more researchers. Cell and gene therapies provide effective treatment options for patients who suffer from uncommon and difficult-to-treat medical conditions. With supportive laws and rising financial backing, Japan is becoming a worldwide centre for research and business activities related to innovative treatment methods.

To learn more about this report, Download Free Sample Report
By Application
- Oncology: The field of oncology shows strong market growth because of increasing cancer cases which are currently affecting Japan. Biopharmaceuticals serve as the primary treatment method for targeted cancer therapies, which lead to better survival rates and less severe side effects. The market segment experiences continuous growth because of ongoing advancements in immunotherapy and precision medicine, which show increasing significance for the business.
- Autoimmune Diseases: The rising number of autoimmune disorders requires treatment with modern biologic therapies, which drives treatment demand. Traditional therapies do not provide the same level of treatment benefits as biopharmaceuticals, which deliver precise and enduring therapeutic effects. Japanese patients experience constant growth in treatment options because of better diagnostic methods and increased patient knowledge.
- Infectious Diseases: Vaccines and innovative therapies depend on biopharmaceuticals, which serve as essential tools for controlling infectious disease outbreaks. Japan possesses an effective healthcare system which enables rapid outbreak control measures. The ongoing research activities, together with the preparedness programmes, ensure that effective treatment methods will be developed, which results in consistent demand for this market sector.
- Metabolic Disorders: Japan faces increasing healthcare demands because of growing diabetes and other metabolic disorders, which require effective treatment options. The market widely utilises biopharmaceutical products such as insulin and enzyme therapies. The pharmaceutical industry experiences continuous growth in this market segment because of better patient outcomes resulting from ongoing drug development progress.
Country Insights
The biopharmaceutical market in Japan combines two distinct elements, which are advanced research capabilities and well-developed medical systems. The demand for medical treatments keeps growing because the elderly population increases their need for care, which includes treating chronic conditions and unusual diseases and age-related medical problems. The government's funding initiatives establish a vital framework because they implement policies which accelerate drug development processes while backing research for next-generation medical solutions in areas such as regenerative medicine and precision therapies.
The country of Japan hosts both established international pharmaceutical companies and an increasing number of emerging biotech startups. The academic institutions, industry organisations, and international companies work together to create new technologies which will enhance the efficiency of bringing new medical treatments to the marketplace. The country maintains high standards for drug development and manufacturing through its commitment to safety and quality and its drive for technological progress.
Japan has dedicated itself to research and development activities while digital health technologies have become more popular to establish itself as a biopharmaceutical hub for the Asia-Pacific region.
Recent Development News
Japan Approves First iPS Cell-Based Therapies: Japan has taken a historic step by approving treatments derived from induced pluripotent stem (iPS) cells. This breakthrough highlights the country’s leadership in regenerative medicine and opens new doors for treating heart failure and Parkinson’s disease.
Japan Forms New Biotechnology Working Group: The Japanese government launched a biotechnology working group to boost supply chains and funding. This initiative aims to strengthen the country’s global competitiveness in advanced therapeutics.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 15783.4 Million |
|
Market size value in 2026 |
USD 16792.8 Million |
|
Revenue forecast in 2033 |
USD 26102.4 Million |
|
Growth rate |
CAGR of 6.50% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Japan |
|
Key company profiled |
Takeda Pharmaceutical Company Ltd., Daiichi Sankyo Company Ltd., Astellas Pharma Inc., Chugai Pharmaceutical Co. Ltd., Eisai Co. Ltd., Fujifilm Diosynth Biotechnologies, Kyowa Kirin Co. Ltd., Otsuka Holdings Co. Ltd., Pfizer Inc., Roche Holding AG, Novartis AG, Sanofi SA, Johnson & Johnson, Amgen Inc., Bristol Myers Squibb. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Product Type (Monoclonal Antibodies, Vaccines, Recombinant Proteins, Cell & Gene Therapies), By Application (Oncology, Autoimmune Diseases, Infectious Diseases, Metabolic Disorders). |
Key Japan Biopharmaceutical Company Insights
The biopharmaceutical market in Japan develops through the combined efforts of established pharmaceutical companies and new biotech startups. Leading companies develop their competitive edge by investing in advanced therapies, which include biologics and cell and gene therapies and precision medicine. Companies use partnerships and licensing agreements and joint research programmes to expand their international operations. The smaller biotech companies drive innovation through their development of specialized solutions. Japanese companies preserve their status as a leading biochemical research centre through their ongoing research funding and dedication to maintaining high-quality standards.
Company List
- Takeda Pharmaceutical Company Ltd.
- Daiichi Sankyo Company Ltd.
- Astellas Pharma Inc.
- Chugai Pharmaceutical Co. Ltd.
- Eisai Co. Ltd.
- Fujifilm Diosynth Biotechnologies
- Kyowa Kirin Co. Ltd.
- Otsuka Holdings Co. Ltd.
- Pfizer Inc.
- Roche Holding AG
- Novartis AG
- Sanofi SA
- Johnson & Johnson
- Amgen Inc.
- Bristol Myers Squibb
Japan Biopharmaceutical Market Report Segmentation
By Product Type
- Monoclonal Antibodies
- Vaccines
- Recombinant Proteins
- Cell & Gene Therapies
By Application
- Oncology
- Autoimmune Diseases
- Infectious Diseases
- Metabolic Disorders
Frequently Asked Questions
Find quick answers to common questions.
The approximate Japan Biopharmaceutical Market size for the Market will be USD 26102.4 Million in 2033.
The key segments of the Japan Biopharmaceutical Market are By Product Type (Monoclonal Antibodies, Vaccines, Recombinant Proteins, Cell & Gene Therapies), By Application (Oncology, Autoimmune Diseases, Infectious Diseases, Metabolic Disorders).
Major players in the Japan Biopharmaceutical Market are Takeda Pharmaceutical Company Ltd., Daiichi Sankyo Company Ltd., Astellas Pharma Inc., Chugai Pharmaceutical Co. Ltd., Eisai Co. Ltd., Fujifilm Diosynth Biotechnologies, Kyowa Kirin Co. Ltd., Otsuka Holdings Co. Ltd., Pfizer Inc., Roche Holding AG, Novartis AG, Sanofi SA, Johnson & Johnson, Amgen Inc., Bristol Myers Squibb.
The current market size of the Japan Biopharmaceutical Market is USD 15783.4 Million in 2025.
The Japan Biopharmaceutical Market CAGR is 6.50%.
- Takeda Pharmaceutical Company Ltd.
- Daiichi Sankyo Company Ltd.
- Astellas Pharma Inc.
- Chugai Pharmaceutical Co. Ltd.
- Eisai Co. Ltd.
- Fujifilm Diosynth Biotechnologies
- Kyowa Kirin Co. Ltd.
- Otsuka Holdings Co. Ltd.
- Pfizer Inc.
- Roche Holding AG
- Novartis AG
- Sanofi SA
- Johnson & Johnson
- Amgen Inc.
- Bristol Myers Squibb
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


