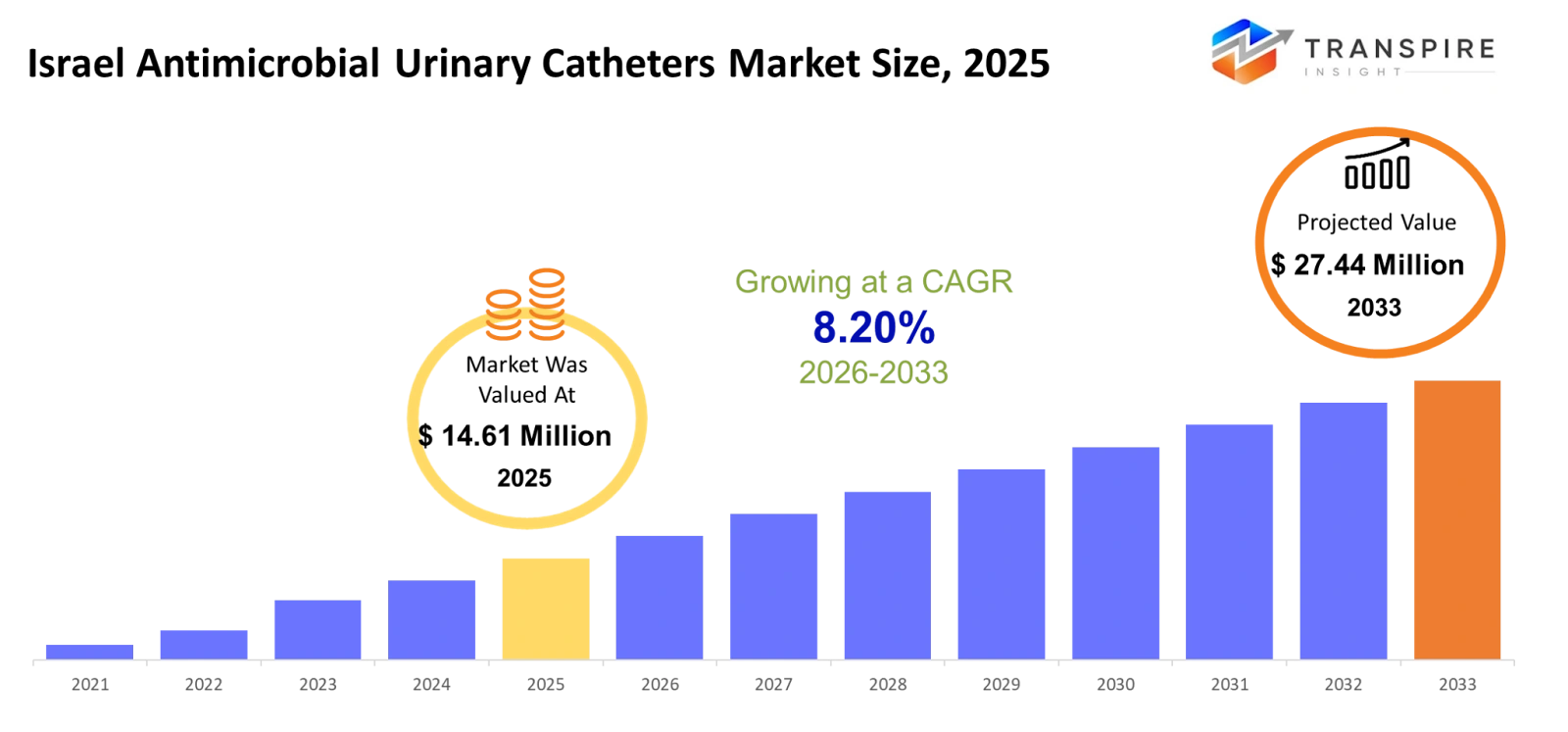
Israel Antimicrobial Urinary Catheters Market Size & Forecast:
- Israel Antimicrobial Urinary Catheters Market Size 2025: USD 14.61 Million
- Israel Antimicrobial Urinary Catheters Market Size 2033: USD 27.44 Million
- Israel Antimicrobial Urinary Catheters Market CAGR: 8.20%
- Israel Antimicrobial Urinary Catheters Market Segments: By Product Type (Silver-Alloy Catheters, Antibiotic-Coated Catheters), By Application (Short-Term Catheterization, Long-Term Catheterization), By End-User (Hospitals, Clinics, Homecare Settings).
To learn more about this report, Download Free Sample Report
Israel Antimicrobial Urinary Catheters Market Summary:
The Israel Antimicrobial Urinary Catheters Market size is estimated at USD 14.61 Million in 2025 and is anticipated to reach USD 27.44 Million by 2033, growing at a CAGR of 8.20% from 2026 to 2033.
The antimicrobial urinary catheters’ market in Israel would be classified under the advanced medical device segment. This is because the major issue would be infection control. This is also because the hospitals would be continuing with the trend of using coated catheters. This is because the patients would be educated about the types of catheters. Moreover, hospital-acquired infections would lead to severe penalties. Today, medical professionals would prefer materials that prevent bacterial colonization rather than the use of antibiotics for the purpose of defending against these microorganisms. Today, there would be a higher emphasis on quality certifications. The manufacturers would be producing silver alloy catheters as well as antimicrobial polymer catheters. This would be in accordance with the demands of the hospitals with a need for shorter patient stay. In the future, the regulations would be stringent. This would be because the suppliers would have to provide evidence of clinical benefits as well as the effectiveness of the products, which would be used.
Key Market Trends & Insights:
The hospitals in Israel will maintain their official preference for infection-resistant catheter technology because their clinical responsibility requirements will not diminish. Procurement teams are moving toward devices that demonstrate reduced complication rates in real settings. Healthcare administrators will focus on preventing catheter-associated infections in intensive care unit settings which will create demand for evidence-backed coatings.
Patients will become more informed and selective, which will lead hospitals to implement solutions that enhance comfort and maintain safety. Catheters which provide extended use capabilities while creating minimal discomfort for users will receive increasing interest from medical professionals. This shift will encourage manufacturers to focus on biocompatible materials that balance antimicrobial performance with patient-friendly design features.
The development of smarter catheter systems will proceed through technological advancements which will lead to the creation of new products that incorporate sensor technology and monitoring systems. The adoption of digital tools by Israeli healthcare institutions will enable them to develop new technological solutions. Companies will invest in research collaborations which will result in the development of devices that combine infection control features with real-time usage monitoring capabilities.
The Israeli government will implement stricter regulatory requirements which will demand clinical testing confirmation and post-marketing product assessment. Authorities will require manufacturers to deliver stronger proof of product effectiveness before they will grant initial product approvals or product renewal requests. This regulation will lead manufacturers to establish higher operational standards while they must disclose their operational performance metrics to both hospitals and healthcare decision-making bodies.
Israel Antimicrobial Urinary Catheters Market Segmentation
By Product Type
Silver-Alloy Catheters- The use of silver alloy catheters is expected to increase steadily in hospital settings, with these products being the choice of medical professionals to reduce the risk of infections during critical care and surgical recovery periods.
Antibiotic- Coated Catheters- Antibiotic-coated catheters will continue to be in the spotlight when there is a need to specifically target bacteria. Medical professionals will use these catheters on patients with a higher susceptibility to infections while being careful about the issue of antibiotic resistance.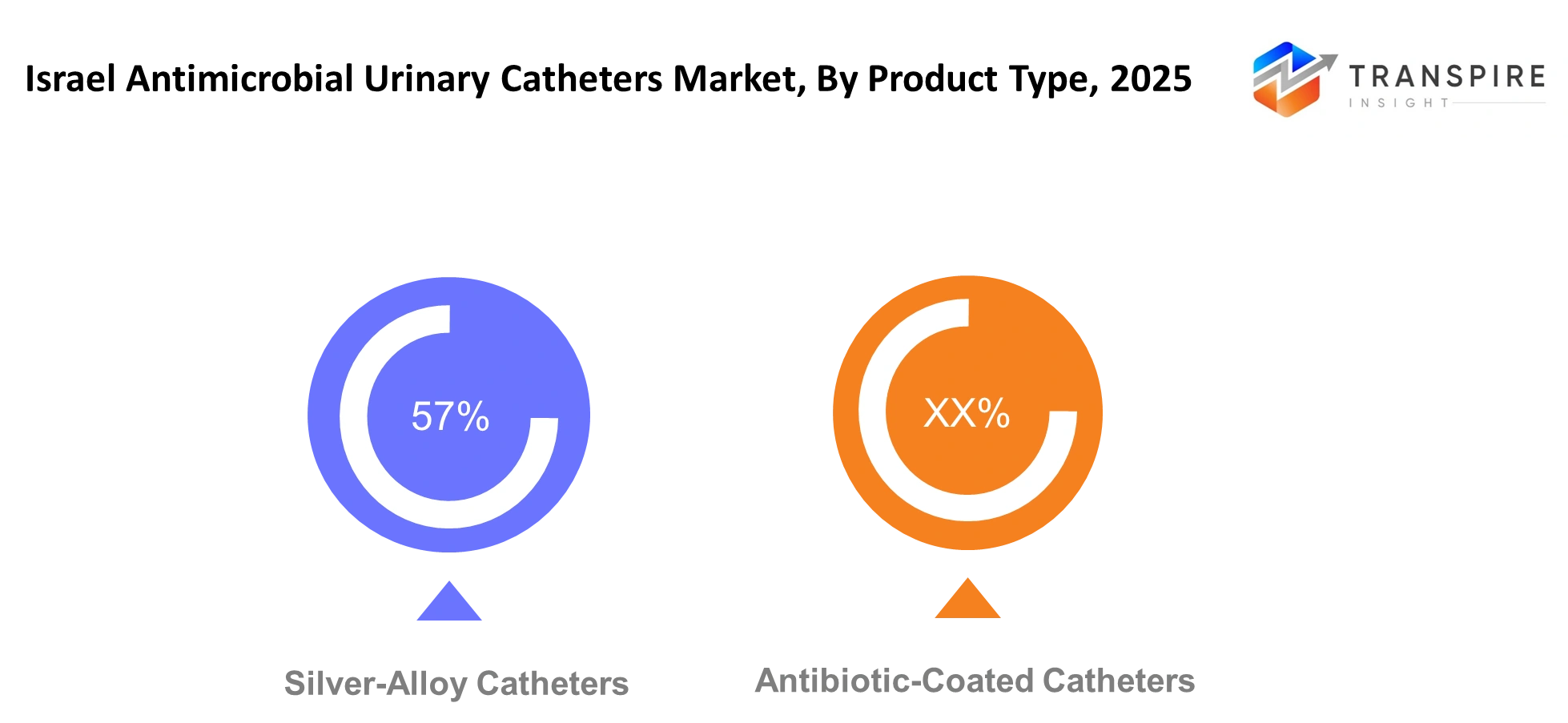
To learn more about this report, Download Free Sample Report
By Application
Short-Term Catheterization- Short-term catheterization will continue to be used in both surgical procedures and emergency medical treatment facilities. Hospitals will choose antimicrobial products which provide protection against infections during their short duration of use particularly for monitoring patients after surgery and treating urgent medical situations.
Long-Term Catheterization- The process of long-term catheterization will require development of new advanced antimicrobial solutions which can protect patients throughout extended use. The devices will be created to minimize irritation and biofilm development and multiple infections, which will enhance patient comfort and treatment outcomes.
By End-User
Hospitals- The majority of the usage will be in the hospitals, and the reason for that is the sheer volume of patients they have to deal with, and the infection control practices they have to follow require more stringent measures. The organization will continue to be committed to the best in antimicrobial catheter technology, and the reason for that is the need to follow regulatory requirements and to provide the best in clinical services.
Clinics- As outpatient procedures become more common clinics will start using antimicrobial catheter solutions. Healthcare providers will concentrate on developing affordable products which maintain patient safety standards while keeping their operational costs low in small medical facilities.
Homecare Settings- Patients will use antimicrobial catheters at home because more people will receive treatment outside hospital facilities. The products will include design features which enable users to operate them safely while minimizing their chances of developing infections during extended self-treatment periods.
Country Insights
The Israeli healthcare system will maintain its dedication to disease control which serves as the country's main healthcare focus while encouraging hospitals to implement antimicrobial catheter solutions. Public and private healthcare providers will achieve standardized operations through their coordinated efforts while centralized purchasing policies will determine product selection based on clinical performance and cost effectiveness and patient safety results across various treatment environments.
Urban medical centers will drive early adoption of advanced catheter technologies, supported by higher budgets and access to specialized expertise. The smaller regional facilities will start using the solutions when their prices become stable. The different speeds of progress will determine how suppliers develop their strategies to enable them to use flexible pricing and distribute their products through various healthcare settings.
Innovation culture at the local level will be a factor when implementing the development of new catheters. Research institutions and startups in Israel will be working with worldwide manufacturers to implement pilot programs and clinical trials. This will promote the use of the next generation of antimicrobial materials to meet the changing demands of the clinical community.
Recent Development News
New Product Launches Supporting Infection Prevention- The recent developments in Israel demonstrate that urology departments are implementing stronger infection control measures. The introduction of Utipro® Plus for treating urinary tract infections in January 2026 demonstrated that healthcare facilities were developing new preventive treatment methods while creating new medical devices, which led to increased demand for antimicrobial catheter products.
Global Technology Advancements Influencing Local Adoption- Research and development work on antimicrobial coatings will drive progress in catheter technology which enters Israel. The new material development which includes better antibiofilm coatings and advanced surface protection methods will enhance product durability and performance. Healthcare providers will use these advancements to make purchasing decisions because they need catheters that can resist infections for extended periods.
Policy and Research Funding Focus on Antimicrobial Resistance- Israel's international antimicrobial resistance research program participation in 2026 shows how the country has shifted its policy to focus on infection prevention technologies. The government funding programs will support the research and development of new medical technologies which include antimicrobial devices because they need to develop preventive medical solutions like coated urinary catheters for use in healthcare settings.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 14.61 Million |
|
Market size value in 2026 |
USD 15.81 Million |
|
Revenue forecast in 2033 |
USD 27.44 Million |
|
Growth rate |
CAGR of 8.20% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Israel |
|
Key company profiled |
Becton Dickinson and Company, Teleflex Incorporated, Coloplast A/S, B. Braun Melsungen AG, Hollister Incorporated, ConvaTec Group plc, Cook Medical LLC, Medtronic plc, Boston Scientific Corporation, C. R. Bard Inc., Cardinal Health Inc., WellLead Medical Co. Ltd., AngioDynamics Inc., Advin Health Care, Romsons Group. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Product Type (Silver-Alloy Catheters, Antibiotic-Coated Catheters), By Application (Short-Term Catheterization, Long-Term Catheterization), By End-User (Hospitals, Clinics, Homecare Settings). |
Key Israel Antimicrobial Urinary Catheters Market Company Insights
The major players in the Israel antimicrobial urinary catheters market would be focused on delivering clinical reliability and material innovation. The multinational companies would be well represented in the market by forming associations with local healthcare facilities in Israel. The positioning strategy for the players would be based on their ability to deliver infection reduction, longevity, and conformance to tight regulatory and procurement requirements.
Company List
- Becton Dickinson and Company
- Teleflex Incorporated
- Coloplast A/S
- B. Braun Melsungen AG
- Hollister Incorporated
- ConvaTec Group plc
- Cook Medical LLC
- Medtronic plc
- Boston Scientific Corporation
- C. R. Bard Inc.
- Cardinal Health Inc.
- WellLead Medical Co. Ltd.
- AngioDynamics Inc.
- Advin Health Care
- Romsons Group
Israel Antimicrobial Urinary Catheters Market Report Segmentation
By Product Type
- Silver-Alloy Catheters
- Antibiotic-Coated Catheters
By Application
- Short-Term Catheterization
- Long-Term Catheterization
By End-User
- Hospitals
- Clinics
- Homecare Settings
Frequently Asked Questions
Find quick answers to common questions.
The Approximate Israel Antimicrobial Urinary Catheters Market size for the Market will be USD 27.44 Million in 2033.
Key Segments for the Israel Antimicrobial Urinary Catheters Market By Product Type (Silver-Alloy Catheters, Antibiotic-Coated Catheters), By Application (Short-Term Catheterization, Long-Term Catheterization), By End-User (Hospitals, Clinics, Homecare Settings).
Major Players in the Israel Antimicrobial Urinary Catheters Market are Becton Dickinson and Company, Teleflex Incorporated, Coloplast A/S, B. Braun Melsungen AG, Hollister Incorporated, ConvaTec Group plc, Cook Medical LLC, Medtronic plc, Boston Scientific Corporation, C. R. Bard Inc., Cardinal Health Inc., WellLead Medical Co. Ltd., AngioDynamics Inc., Advin Health Care, Romsons Group.
The Current Israel Antimicrobial Urinary Catheters Market size is USD 14.61 Million in 2025.
The Israel Antimicrobial Urinary Catheters Market CAGR is 8.20%.
- Becton Dickinson and Company
- Teleflex Incorporated
- Coloplast A/S
- B. Braun Melsungen AG
- Hollister Incorporated
- ConvaTec Group plc
- Cook Medical LLC
- Medtronic plc
- Boston Scientific Corporation
- C. R. Bard Inc.
- Cardinal Health Inc.
- WellLead Medical Co. Ltd.
- AngioDynamics Inc.
- Advin Health Care
- Romsons Group
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


