France Pharmaceutical Contract Manufacturing Market Size & Forecast:
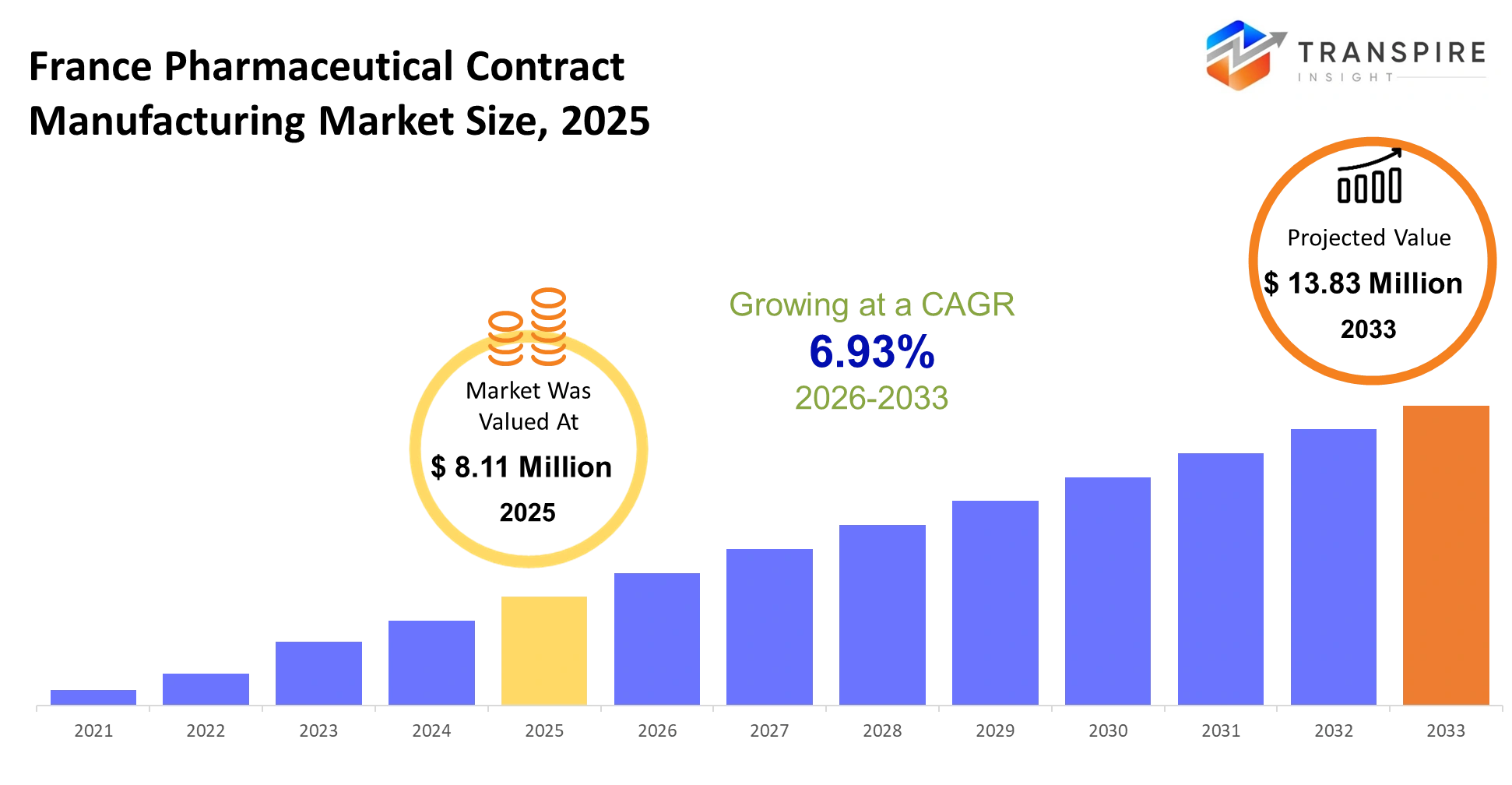
- France Pharmaceutical Contract Manufacturing Market Size 2025: USD 8.11 Million
- France Pharmaceutical Contract Manufacturing Market Size 2033: USD 13.83 Million
- France Pharmaceutical Contract Manufacturing Market CAGR: 6.93%
- France Pharmaceutical Contract Manufacturing Market Segments: Service Type (API Manufacturing, Finished Dosage Form Manufacturing, Packaging Services), End User (Pharmaceutical Companies, Biotechnology Firms).
To learn more about this report, Download Free Sample Report
France Pharmaceutical Contract Manufacturing Market Summary:
The France Pharmaceutical Contract Manufacturing Market size is estimated at USD 8.11 Million in 2025 and is anticipated to reach USD 13.83 Million by 2033, growing at a CAGR of 6.93% from 2026 to 2033. The pharmaceutical and biotechnology companies are increasing their outsourcing activities to achieve cost efficiency and scalability and regulatory expertise.Contract manufacturing organizations (CMOs) and contract development and manufacturing organizations (CDMOs) deliver essential services which support both drug development and commercial operations while allowing companies to focus on their research and innovative activities. The rising need for advanced drug formulations together with biologics and specialty medicines has increased the demand for external manufacturing services throughout the United States.
France establishes itself as an ideal site for pharmaceutical manufacturing services because of its robust healthcare infrastructure and skilled workforce and its regulatory frameworks which provide support to businesses. The market expansion is driven by three factors which include increasing demand for generic drugs and the growth of clinical pipelines and rising investments in advanced therapies which include cell and gene treatments. The market will experience growth during the upcoming years because manufacturing processes will become more efficient through technological advancements which create the need for versatile production systems that maintain high standards of quality.
Key Market Trends & Insights:
- Companies use Contract Manufacturing Organizations and Contract Development and Manufacturing Organizations to achieve lower operational expenses while they enhance productivity and gain access to specialized resources needed for developing advanced pharmaceutical products that require extensive manufacturing capabilities.
- The market experiences significant development because of the rising need for biologics and advanced therapies which include cell and gene treatments. The products require advanced production methods and specialized knowledge which leads companies to work with contract manufacturers who can deliver top quality production that meets regulatory requirements and operational demands.
- The demand for contract manufacturing services throughout France increases as generic drug production grows. Pharmaceutical companies increasingly turn to external partners for their generic drug manufacturing needs because branded drug patents become invalid and this approach helps them stay competitive while decreasing expenses and speeding up product development without compromising their need to follow strict regulations and achieve quality standards.
- Manufacturing processes now achieve better efficiency through technological improvements which include continuous manufacturing and automated systems and digital technology. French contract manufacturers implement these technological solutions to enhance their operational procedures while reducing mistakes and delivering better product results to their pharmaceutical customers in a highly competitive market.
- The pharmaceutical industry needs to comply with strict regulatory demands and quality requirements which creates market constraints. Contract manufacturers establish trust with their customers through their investments in compliance standards and certification processes and their dedication to building quality control systems which meet international regulations. This investment enhances client trust while establishing France as a trustworthy manufacturing destination.
France Pharmaceutical Contract Manufacturing Market Segmentation
Service Type
- API Manufacturing:The France market shows substantial growth for Active Pharmaceutical Ingredient (API) manufacturing because of increasing needs for high-quality bulk drug production. Pharmaceutical companies increasingly outsource API production because it helps them reduce costs, achieve regulatory compliance, and obtain advanced synthesis technologies which are necessary for creating complex and high-potency compounds.
- Finished Dosage Form Manufacturing:The market for this segment is expanding because more people want to buy tablets, capsules, injectables, and other dosage types. Companies rely on contract manufacturers for their formulation development and scale-up needs and commercial production requirements which allows them to launch products faster while achieving better operational results and maintaining high-quality and safety standards.
- Packaging Services:The packaging services market is expanding rapidly because more people need secure and compliant and innovative drug packaging solutions. Contract manufacturers provide primary packaging and secondary packaging and labeling and serialization services to protect product safety and enable traceability and meet European regulatory requirements.
To learn more about this report, Download Free Sample Report
End User
- Pharmaceutical Companies: Pharmaceutical companies represent the largest end-user segment as they increasingly outsource manufacturing to focus on core competencies like research and marketing. Contract manufacturing allows them to reduce production costs while dealing with capacity limitations and it enables them to bring products to market faster without compromising their production quality standards.
- Biotechnology Firms:The biotech industry is experiencing rapid growth because of the increasing need for developing biologics and biosimilars and advanced therapy solutions. These companies depend on specialized CDMOs to handle their complex manufacturing needs because they do not have their own capacity for large-scale production.
Country Insights
France functions as a well-established European market for pharmaceutical contract manufacturing because its healthcare system and research infrastructure and pharmaceutical companies create favorable conditions for business operations. The country attracts companies from both domestic and international markets because it has a skilled workforce and strong regulatory systems and developed infrastructure, which enables them to obtain dependable manufacturing solutions.
France targets strengthening its domestic pharmaceutical production capabilities because this will help decrease import dependency and improve supply chain resilience. The market expands through government initiatives and biologics and advanced therapies investments and increased industry and research institution collaboration. France has become a principal European contract manufacturing center because of its growing drug innovation focus and rising demand for generic and specialty medications.
Recent Development News
In March 2026, Pharmaceutical companies are increasingly delaying drug launches in Europe, including France, due to uncertainties around U.S. pricing policies. The trend is diminishing contract manufacturing demand because companies need to reconsider their production plans and market entry methods throughout various European countries.
In January 2026, Thermo Fisher Scientific secured new pharmaceutical manufacturing contracts as firms shift production toward the United States. The industry trend shows evidence of transforming outsourcing practices which will decrease Europe's portion of worldwide contract manufacturing operations.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 8.11 Million |
|
Market size value in 2026 |
USD 8.653 Million |
|
Revenue forecast in 2033 |
USD 13.83 Million |
|
Growth rate |
CAGR of 6.93% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
France |
|
Key company profiled |
Lonza, Catalent, Recipharm, Fareva, Samsung Biologics, WuXi AppTec, Patheon, Thermo Fisher Scientific, Aenova Group, Boehringer Ingelheim, AbbVie Contract Manufacturing, Fujifilm Diosynth Biotechnologies, PCI Pharma Services, Cambrex, CordenPharma. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
Service Type (API Manufacturing, Finished Dosage Form Manufacturing, Packaging Services), End User (Pharmaceutical Companies, Biotechnology Firms). |
Key France Pharmaceutical Contract Manufacturing Market Company Insights
The France pharmaceutical contract manufacturing market features a mix of global CDMO leaders and strong regional players, all competing through advanced capabilities, global reach, and regulatory expertise. The market leaders Lonza, Catalent, and Thermo Fisher Scientific control the industry through their complete service offerings which include drug development and biologics manufacturing and commercial-scale production. The companies invest substantial resources to develop advanced technologies which include cell and gene therapy platforms, which enable them to develop complex therapeutics while meeting international regulatory standards.
European and France-based players such as Fareva, Recipharm, and Aenova Group play a crucial role in strengthening regional manufacturing capacity, particularly in oral solid dosage, sterile injectables, and packaging services. The companies Samsung Biologics, WuXi AppTec, and Fujifilm Diosynth Biotechnologies all expand internationally their operations which creates greater rivalry through their extensive biologics manufacturing facilities. The companies Boehringer Ingelheim, AbbVie Contract Manufacturing, PCI Pharma Services, Cambrex, and CordenPharma create a competitive environment which drives innovation through their focus on specialized services and API manufacturing and high-potency drug development.
Company List
- Lonza
- Catalent
- Recipharm
- Fareva
- Samsung Biologics
- WuXi AppTec
- Patheon
- Thermo Fisher Scientific
- Aenova Group
- Boehringer Ingelheim
- AbbVie Contract Manufacturing
- Fujifilm Diosynth Biotechnologies
- PCI Pharma Services
- Cambrex
- CordenPharma
France Pharmaceutical Contract Manufacturing Market Report Segmentation
By Service Type
- API Manufacturing
- Finished Dosage Form Manufacturing
- Packaging Services
By End User
- Pharmaceutical Companies
- Biotechnology Firms
Frequently Asked Questions
Find quick answers to common questions.
The approximate France Pharmaceutical Contract Manufacturing Market size for the market will be USD 13.83 Million in 2033.
Key segments for the France Pharmaceutical Contract Manufacturing Market are Service Type (API Manufacturing, Finished Dosage Form Manufacturing, Packaging Services), End User (Pharmaceutical Companies, Biotechnology Firms).
Major France Pharmaceutical Contract Manufacturing Market players are Lonza, Catalent, Recipharm, Fareva, Samsung Biologics, WuXi AppTec, Patheon, Thermo Fisher Scientific, Aenova Group, Boehringer Ingelheim, AbbVie Contract Manufacturing, Fujifilm Diosynth Biotechnologies, PCI Pharma Services, Cambrex, CordenPharma.
The France Pharmaceutical Contract Manufacturing Market size is USD 8.11 Million in 2025.
The France Pharmaceutical Contract Manufacturing Market CAGR is 6.93%.
- Lonza
- Catalent
- Recipharm
- Fareva
- Samsung Biologics
- WuXi AppTec
- Patheon
- Thermo Fisher Scientific
- Aenova Group
- Boehringer Ingelheim
- AbbVie Contract Manufacturing
- Fujifilm Diosynth Biotechnologies
- PCI Pharma Services
- Cambrex
- CordenPharma
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033