France Pharmaceutical Membrane Filtration Market Size & Forecast:
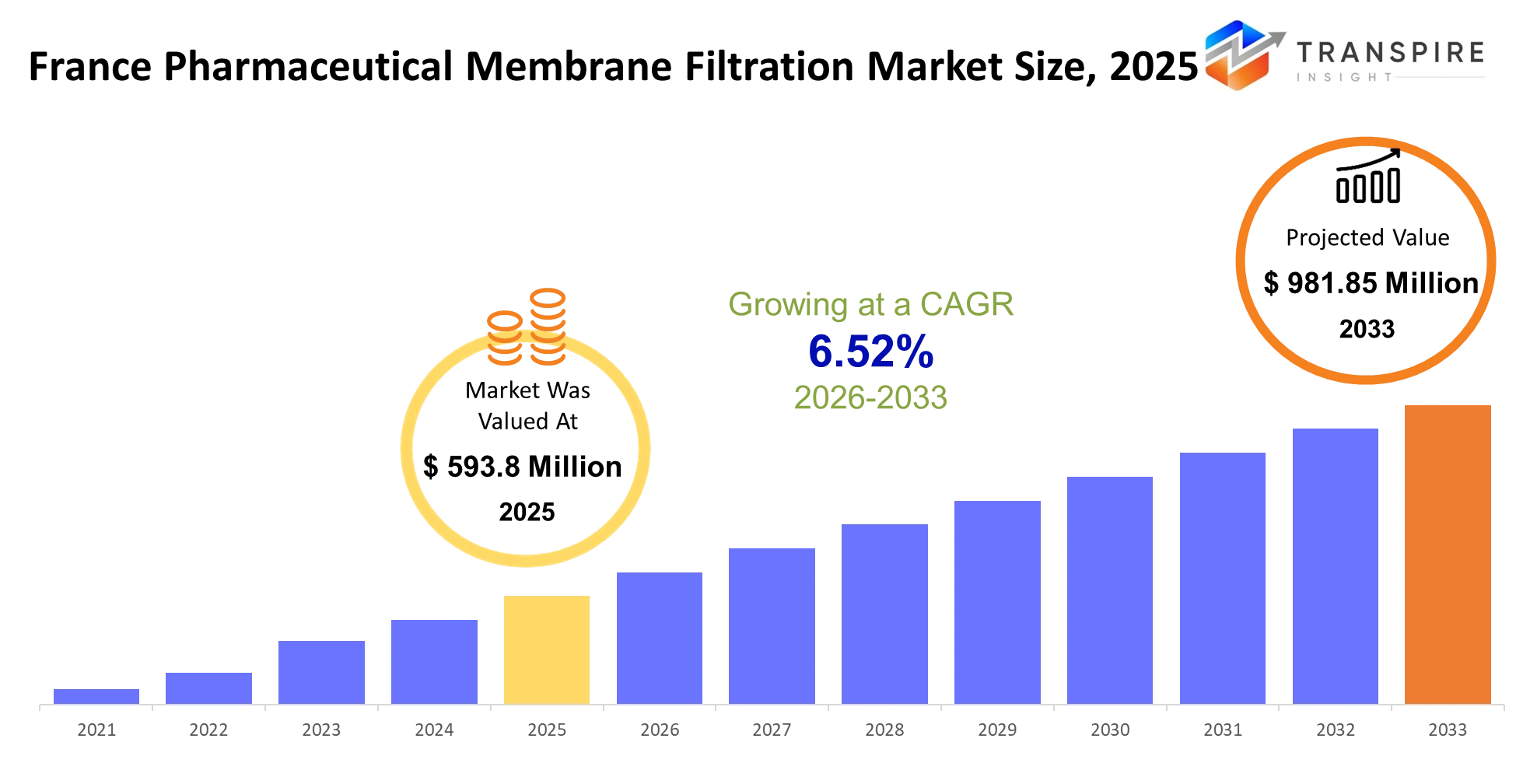
- France Pharmaceutical Membrane Filtration Market Size 2025: USD 593.8 Million
- France Pharmaceutical Membrane Filtration Market Size 2033: USD 981.85 Million
- France Pharmaceutical Membrane Filtration Market CAGR: 6.52%
- France Pharmaceutical Membrane Filtration Market Segments: By Type (Microfiltration, Ultrafiltration, Nanofiltration, Reverse Osmosis, Others); By Application (Sterile Filtration, Drug Processing, Biologics, Water Purification, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, CMOs, Others); By Material (Polymeric, Ceramic, Others).
To learn more about this report, Download Free Sample Report
France Pharmaceutical Membrane Filtration Market Summary
The France Pharmaceutical Membrane Filtration Market was valued at USD 593.8 Million in 2025. It is forecast to reach USD 981.85 Million by 2033. That is a CAGR of 6.52% over the period.
Pharmaceutical membrane filtration operates inside drug and biologics manufacturing facilities throughout France to conduct liquid separation and purification and sterilization of vaccines and injectable medications and advanced therapeutic treatments. The method functions as an essential security measure for sterile production lines and bioprocessing operations because it guarantees successful elimination of all harmful substances and microorganisms and undesired materials before products enter patient use.
Biopharmaceutical production growth during the past three to five years has created a market transformation which now favors single-use products and efficient membrane systems that operate at high performance. The COVID-19 vaccine manufacturing surge served as a primary catalyst which revealed existing capacity limitations and motivated manufacturers to implement faster-disposable filtration systems. The enforcement of stricter EU Good Manufacturing Practices and the rise of regulatory scrutiny created an increased demand for validated filtration systems which operate with high precision. The producers now prioritize three factors because these elements require investment in advanced membrane technologies: flexibility and contamination control and rapid production cycle times which ensure supply reliability and operational stability.
Key Market Insights
- The Western region of France controls the Pharmaceutical Membrane Filtration Market because of its strong biopharma clusters and its advanced manufacturing facilities.
- The Southern region of France experiences the fastest growth because of its increasing number of biotech startups and its expanding vaccine production facilities. The Paris region maintains a substantial market portion because it contains both pharmaceutical research facilities and regulatory bodies.
- The France Pharmaceutical Membrane Filtration Market depends on ultrafiltration membranes which industrial facilities use to separate proteins from biologics throughout their operations.
- Microfiltration systems hold the second-largest share, which functions to filter sterile injectable drug formulations through its established usage.
- The single-use filtration systems represent the market segment with the highest growth rate because customers demand production methods which provide flexible operation without risk of contamination.
- The main application for biopharmaceutical manufacturing supports the production of monoclonal antibodies and vaccine development pipelines. Vaccine filtration applications show rapid growth due to pandemic-driven capacity expansion and preparedness investments.
- The France Pharmaceutical Membrane Filtration Market experiences growth because drug formulation and sterile processing operations face increasing regulatory requirements.
- The strategic partnerships between companies and biotech firms are helping to develop filtration solutions that meet specific customer needs.
- The regional expansion of manufacturing facilities together with R&D centers enables companies to create supply systems which provide better service and increase their production capabilities.
- The technology upgrades which single-use systems receive today help facilities to achieve better contamination control together with expanded operational capabilities.
- The France Pharmaceutical Membrane Filtration Market is shifting its focus toward two advanced manufacturing methods which include continuous processing and modular biomanufacturing systems.
- The demand for filtration systems which meet regulatory standards according to EU GMP requirements and sterile manufacturing practices is increasing.
What are the Key Drivers, Restraints, and Opportunities in the France Pharmaceutical Membrane Filtration Market?
The biopharmaceutical market in France particularly for monoclonal antibodies vaccines and cell-based therapies drives the growth of the Pharmaceutical Membrane Filtration Market. Manufacturers had to update their old filtration systems because the post-pandemic manufacturing realignment together with new EU GMP sterilization standards created this need. The pharmaceutical industry now uses advanced membrane filtration systems to achieve better sterility control while decreasing production errors and increasing operational output. The industrial sector has increased its capital expenditures to establish high-capacity filtration systems, which drives continual demand for filter replacements and disposable products in their manufacturing operations.
Advanced membrane filtration systems face a major barrier because their operational costs and system validation expenses are extremely high. The deployment process becomes slower because organizations need to complete multiple testing procedures together with documentation requirements and quality assurance activities to meet strict regulatory validation standards. Pharmaceutical manufacturers must follow EU regulatory requirements, which creates a permanent structural obstacle that prevents the removal of this restriction. Budget limitations prevent smaller manufacturers and CDMOs from adopting new technologies, which decreases their market share and revenue growth, especially during the initial stages of biologics manufacturing.
Biotech clusters, which are emerging in Lyon and Toulouse, present a significant market opportunity through their implementation of single-use and continuous bioprocessing technologies. The bio manufacturing industry is expanding its investment in modular biomanufacturing facilities because these facilities provide faster production capabilities which enable companies to create larger quantities while maintaining lower contamination levels.
What Has the Impact of Artificial Intelligence Been on the France Pharmaceutical Membrane Filtration Market?
The France Pharmaceutical Membrane Filtration Market undergoes transformation through artificial intelligence which enhances control processes and operational stability and testing speed for biologics production facilities. The current filtration systems use artificial intelligence to automate the monitoring process which checks membrane conditions and observes pressure changes and measures flow rates throughout the entire system. The system reduces the need for human operators during essential processes of sterile filtration while it ensures that product quality remains stable throughout the manufacturing process of vaccines and biologics.
Machine learning enables organizations to create predictive models which forecast membrane fouling and filter degradation and unanticipated operational changes. The predictive maintenance scheduling system uses batch data and sensor input to create maintenance schedules which decrease unexpected equipment failures while optimizing equipment management throughout its life cycle. Early fault detection systems combined with optimized cleaning cycles help biopharma plants in France achieve better operational uptime and decrease their need to replace filters.
The integration of artificial intelligence into operations enables better yield maintenance because it adjusts filtration settings according to batch requirements. The system provides regulatory compliance support through its ability to produce validation logs which meet EU GMP standards thus decreasing administrative work and speeding up the audit process.
The main challenge lies in the expensive and intricate process of implementing artificial intelligence technology into existing membrane filtration systems that use traditional methods. The industry faces a major obstacle because most facilities lack access to high-quality standardized process information which results in reduced model performance and hinders industry-wide adoption.
Key Market Trends
- After 2020, biopharma manufacturers switched to single-use membranes because they wanted to decrease contamination risks and achieve faster production times than their previous reusable systems.
- Enterprises have needed to enhance their verification-heavy filtration systems because EU GMP regulations have become more stringent since 2021 which has resulted in rising use of advanced membrane technologies.
- The growth of vaccine manufacturing facilities after the COVID-19 pandemic created an urgent requirement for quick-installation filtration systems throughout French biologics manufacturing sites. The commercial production of monoclonal antibodies experienced a major increase in ultrafiltration technology usage throughout 2023.
- The contract development manufacturing organizations expanded their membrane filtration capabilities by more than 30 percent during the period from 2021 to 2025 in order to fulfill the biologics manufacturing requirements which they received from their clients.
- Manufacturing companies adopted automation systems at their filtration operations because they wanted to monitor their processes in real time while decreasing defects that occurred during production.
- The demand for modular bioprocessing systems increased dramatically during 2024 because these systems allowed small and mid-size biotech companies to adjust their production capacity according to their needs.
- After global disruptions, supply chain localization efforts increased which resulted in France-based production of filtration components and reduced dependency on imports.
- Organizations replaced multi-use filtration systems with disposable technologies because sustainability demands required them to decrease their use of cleaning chemicals.
France Pharmaceutical Membrane Filtration Market Segmentation
By Type
- Your training data includes information up to the month of October in the year 2023. The main technology used in the France Pharmaceutical Membrane Filtration Market operates through ultrafiltration which functions as an essential part of biologics and protein and vaccine manufacturing processes. The manufacturing of monoclonal antibodies requires manufacturers to use technology that controls molecular cut-off because the therapeutic molecules need scientists to control their movement. The injectable drug production process depends on microfiltration which serves as an essential technology that delivers both sterile filtration and pre-filtration functions throughout drug manufacturing operations.
- The demand for ultrafiltration technology has increased because of two factors which are the growing need for biologics production and the new European Union Good Manufacturing Practice regulations that require higher product quality and operational reliability. Nanofiltration and reverse osmosis function as dedicated technologies which produce buffer solutions and ultra-pure water needed for water-for-injection systems. The rising adoption of continuous bioprocessing leads to new standards for membrane integration which now includes multiple membrane types within a single production process.
- The market shows ultrafiltration as the leading technology which will dominate until the end of the forecast period while advanced purification applications will cause nanofiltration and reverse osmosis to increase their market share. Equipment developers are increasingly focusing on hybrid membrane systems that improve flux efficiency and reduce fouling. Investors demonstrate a preference for filtration technologies which allow easy scaling and modularization because these technologies satisfy the needs of both biologics production and automated manufacturing processes.
By Application
- The core segment of the France Pharmaceutical Membrane Filtration Market operates through biologics processing which maintains its dominant market share because production activities create vaccines and recombinant proteins and advanced therapies. The manufacturing process of injectable drugs uses sterile filtration as a mandatory requirement which organizations need to follow according to their regulatory compliance obligations.
- The growth of biologics depends on two factors which include the expanding number of clinical trials and the rising trend of pharmaceutical companies outsourcing their work to CDMOs that need large-scale adaptable filtration systems. The drug processing sector experiences continuous growth because pharmaceutical companies upgrade their outdated production facilities. Water purification stands as a permanent vital application which specifically serves the production needs of pharmaceutical-grade water systems used in manufacturing environments.
- The upcoming growth period will focus on biologics and sterile filtration applications because regulatory requirements become more stringent and production output increases. The market demand will drive manufacturers to develop integrated filtration solutions which enable operators to manage multiple operational processes through a single unified system. The market demand will push buyers to select technologies which decrease batch inconsistency while enhancing complete process efficiency from start to finish.
To learn more about this report, Download Free Sample Report
By End-User
- The France Pharmaceutical Membrane Filtration Market faces dominance from pharmaceutical companies because they require extensive production facilities and must meet their regulatory obligations. The biotech sector experiences fast growth because of new developments in cell and gene therapies and the production of small-scale biologics.
- Pharma companies maintain leadership through continuous investment in large-volume production facilities and established supply chains for sterile drug manufacturing. The biotech sector expands because companies need flexible manufacturing options and they receive more venture capital for developing new therapies. Pharmaceutical firms depend on contract manufacturing organizations to execute production needs through their global partnerships.
- Biotech companies and contract manufacturing organizations (CMOs) will increase their market share during the forecast period because the industry will experience higher outsourcing and people will demand more personalized medicine solutions. The demand for modular systems which can scale to different needs will increase because of this market transition. Equipment providers must develop systems which enable producers to switch between their miniaturized and large-scale manufacturing processes.
By Material
- The France Pharmaceutical Membrane Filtration Market shows material dominance through polymeric membranes which provide cost benefits and versatile compatibility and extensive usage in single-use systems. The high-temperature and chemically aggressive environments require specialized ceramic membranes which create a small but advanced technical segment.
- The market for polymeric membranes will expand because biologics manufacturers and vaccine producers increasingly adopt disposable filtration systems. Ceramic membranes serve specific industrial purposes that need strong materials which can endure multiple cleaning processes throughout the entire industrial purification operation. The market for specialized filtration needs currently has limited advanced materials but these materials are starting to receive more recognition.
- Polymeric membranes will maintain their market leadership because they match the needs of single-use bioprocessing operations. The market for ceramic membranes will see continuous growth in high-performance sectors that demand both durability and chemical protection. The competitive market landscape will develop through material innovation which enables manufacturers to achieve higher production rates while decreasing their system fouling levels.
What are the Key Use Cases Driving the France Pharmaceutical Membrane Filtration Market?
The primary application of biopharmaceutical manufacturing exists as the leading use case for France Pharmaceutical Membrane Filtration Market because the production process needs strict sterile separation and purification methods to create monoclonal antibodies, vaccines, and recombinant proteins. The application requires membrane filtration for all production batches because it serves as the essential method which manufacturers need to follow for both product safety and regulatory requirements.
The production of sterile injectable drugs and the clarification of buffer solutions have begun to develop additional application areas which particularly benefit contract development and manufacturing organizations. The end-user organizations need scalable filtration systems to handle their biologics production which they outsource while their order volume from global pharmaceutical firms continues to grow.
The new applications of this technology include processing of cell and gene therapies together with continuous bioprocessing platforms which need exact filtration technology to manage delicate biological substances. Although integration remains in its initial phase, the combination of clinical pipeline growth with European Union funding for advanced therapy initiatives will drive faster integration throughout the upcoming forecast period.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 593.8 Million |
|
Market size value in 2026 |
USD 631.2 Million |
|
Revenue forecast in 2033 |
USD 981.85 Million |
|
Growth rate |
CAGR of 6.52% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
France |
|
Key company profiled |
Merck Millipore, Pall Corporation, Sartorius, Danaher, GE Healthcare, 3M, Koch Membrane Systems, Thermo Fisher, Asahi Kasei, Toray, Pentair, Alfa Laval, Eaton, Veolia, GEA. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Microfiltration, Ultrafiltration, Nanofiltration, Reverse Osmosis, Others); By Application (Sterile Filtration, Drug Processing, Biologics, Water Purification, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, CMOs, Others); By Material (Polymeric, Ceramic, Others). |
Which Regions are Driving the France Pharmaceutical Membrane Filtration Market Growth?
The pharmaceutical membrane filtration market in France is led by Western France because the region contains both biopharmaceutical manufacturing facilities and modern industrial capabilities. The vaccination and biological production sites in Brittany and Pays de la Loire require specialized high-precision filtration systems to operate their existing facilities. The European Union Good Manufacturing Practice regulations mandate strict enforcement which leads manufacturers to implement approved sterile processing methods. The filtration component supplier network together with engineering services maintains operational leadership while enabling production facilities to adopt new technologies.
The pharmaceutical research and development centers of Île-de-France together with its international corporate offices create a stable secondary market. This area concentrates on developing new products through its research activities instead of manufacturing products which Western France does. The global pharmaceutical sector maintains consistent investments which create ongoing requirements for both laboratory and pilot-scale filtration equipment. The sector establishes dependable demand through its economic strength and stable regulations which support needs of new drug testing and validation processes.
The biotech industry in southern France experiences its most rapid growth because cities like Toulouse and Montpellier develop new biotech clusters. The adoption of modern membrane filtration technologies has increased due to recent funding for vaccine research centers and government-supported life science programs. Biotech parks have implemented infrastructure updates which enable them to increase their biological product output capacity at a quicker rate. This growth creates significant chances for businesses that want to enter markets and for people who want to invest in them.
Who are the Key Players in the France Pharmaceutical Membrane Filtration Market and How Do They Compete?
The France Pharmaceutical Membrane Filtration Market shows moderate competition because a few international life sciences and filtration companies hold most market share for high-demand bioprocessing solutions. The pharmaceutical industry prefers established systems which meet regulatory needs and provide permanent operational dependability to cheaper options which lack proven performance. The market competition exists because businesses develop unique technologies that improve their single-use systems and filtration performance and their automated bioprocess systems.
The vendor selection process for biopharma facilities depends on service capabilities which include validation support and lifecycle management. Merck KGaA runs its operations through MilliporeSigma to develop advanced membrane technologies and single-use bioprocess solutions which enable monoclonal antibody and vaccine production. The company protects its market advantage through complete filtration system connections which link all parts of their production process from beginning to end. The company achieves growth through ongoing development of its sterilizing-grade filter products and its local production support agreements with French biomanufacturers.
Danaher Corporation operates through its life sciences subsidiaries including Pall Corporation to provide advanced single-use filtration systems and comprehensive validation services. Sartorius develops its competitive advantage through its precise membrane technology and quick deployment systems which help contract development and manufacturing organizations handle their biologic production needs. Thermo Fisher Scientific grows its business through bioprocessing solutions which unify its filtration systems and analytical instruments and manufacturing services. Koch Membrane Systems specializes in membrane filtration solutions which serve various industrial applications.
Company List
- Merck Millipore
- Pall Corporation
- Sartorius
- Danaher
- GE Healthcare
- 3M
- Koch Membrane Systems
- Thermo Fisher
- Asahi Kasei
- Toray
- Pentair
- Alfa Laval
- Eaton
- Veolia
- GEA
Recent Development News
In February 2025, Thermo Fisher Scientific announced the acquisition of Solventum’s purification and filtration business for approximately USD 4.1 billion, strengthening its bioprocessing and pharmaceutical filtration portfolio. The acquisition enhances its competitive positioning in sterile drug manufacturing and membrane-based purification technologies.
Source: https://www.reuters.com
What Strategic Insights Define the Future of the France Pharmaceutical Membrane Filtration Market?
The France Pharmaceutical Membrane Filtration Market is currently making a structural shift that will result in high-continuity single-use systems which incorporate digital technology into bioprocessing systems during the next 5 to 7 years. The biologics pipeline expansion and EU sterility requirements make manufacturers need to establish faster validation processes together with systems which minimize contamination risks. Manufacturing systems now consider filtration processes as essential control points which operate within their interconnected production networks.
The supply chain problem exists because companies depend on a small number of worldwide suppliers who provide high-quality membrane materials which makes them vulnerable to interruptions caused by political events and shortages of essential materials. Although demand signals show strong customer interest the situation will limit production capacity. Biotech facilities in France now have a new business opportunity through AI-enabled membrane diagnostics which can become part of their ongoing bioprocessing operations. The development exists at an early stage but will transform maintenance practices and yield management standards.
French market participants need to establish local manufacturing partnerships with French companies while developing modular filtration systems which can operate with digital technology and meet regulatory needs for continuous production.
France Pharmaceutical Membrane Filtration Market Report Segmentation
By Type
- Microfiltration
- Ultrafiltration
- Nanofiltration
- Reverse Osmosis
- Others
By Application
- Sterile Filtration
- Drug Processing
- Biologics
- Water Purification
- Others
By End-User
- Pharma Companies
- Biotech Firms
- Research Institutes
- CMOs, Others
By Material
- Polymeric
- Ceramic
- Others
Frequently Asked Questions
Find quick answers to common questions.
The confirmed 2033 market size figure in USD 981.85 Million.
Key segments for the France Pharmaceutical Membrane Filtration Market are By Type (Microfiltration, Ultrafiltration, Nanofiltration, Reverse Osmosis, Others); By Application (Sterile Filtration, Drug Processing, Biologics, Water Purification, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, CMOs, Others); By Material (Polymeric, Ceramic, Others).
Major France Pharmaceutical Membrane Filtration Market players are Merck Millipore, Pall Corporation, Sartorius, Danaher, GE Healthcare, 3M, Koch Membrane Systems, Thermo Fisher, Asahi Kasei, Toray, Pentair, Alfa Laval, Eaton, Veolia, GEA.
The France Pharmaceutical Membrane Filtration Market size is USD 593.8 Million in 2025.
The France Pharmaceutical Membrane Filtration Market CAGR is 6.52% from 2026 to 2033.
- Merck Millipore
- Pall Corporation
- Sartorius
- Danaher
- GE Healthcare
- 3M
- Koch Membrane Systems
- Thermo Fisher
- Asahi Kasei
- Toray
- Pentair
- Alfa Laval
- Eaton
- Veolia
- GEA
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


