France Live Biotherapeutic Products And Microbiome CDMO Market Size & Forecast:
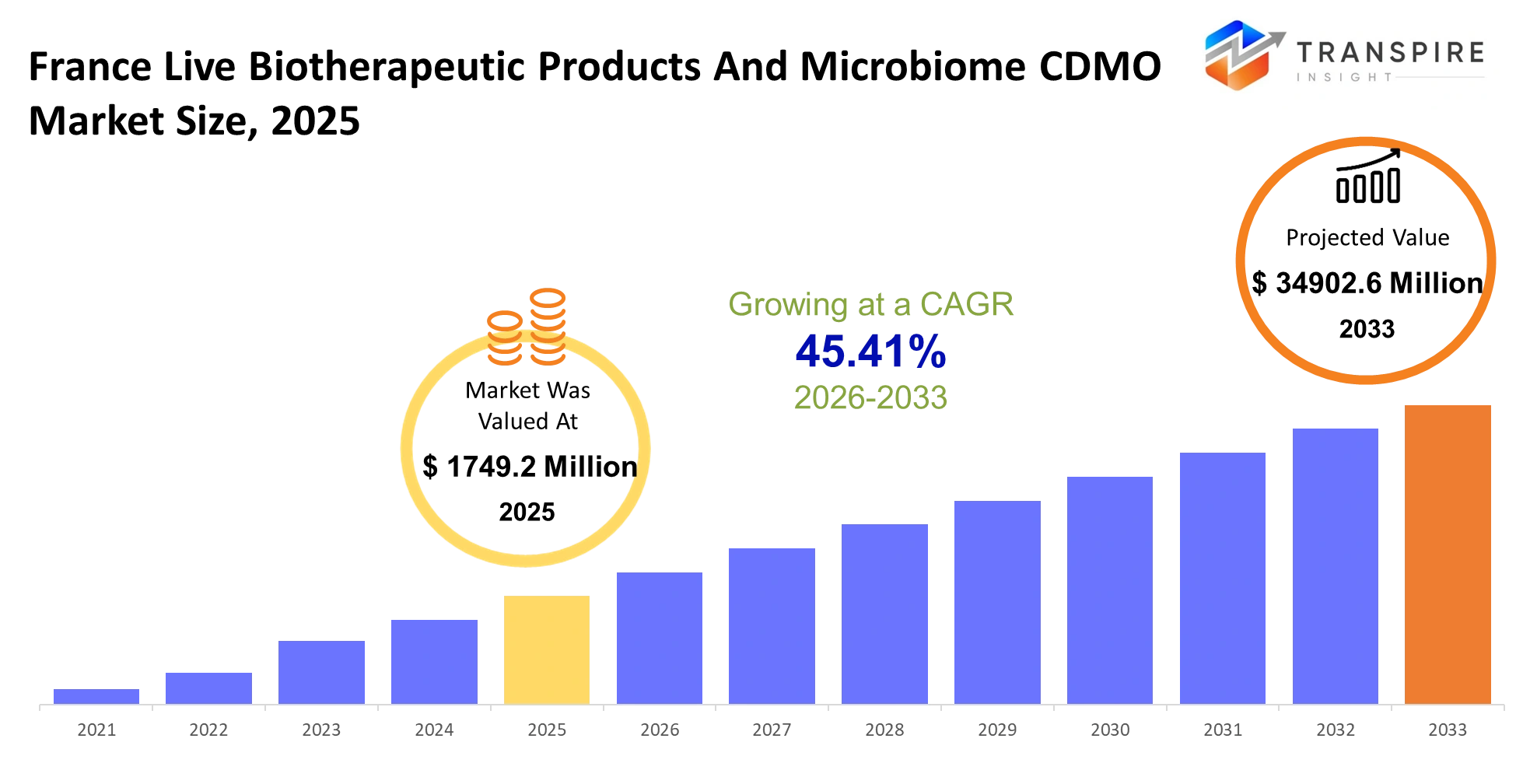
- France Live Biotherapeutic Products And Microbiome CDMO Market Size 2025: USD 1749.2 Million
- France Live Biotherapeutic Products And Microbiome CDMO Market Size 2033: USD 34902.6 Million
- France Live Biotherapeutic Products And Microbiome CDMO Market CAGR: 45.41%
- France Live Biotherapeutic Products And Microbiome CDMO Market Segments: By Type (Drug Development Services, Manufacturing Services, Analytical Services, Others); By Application (Microbiome Therapeutics, Clinical Trials, Drug Discovery, Personalized Medicine, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Hospitals, Others); By Scale (Preclinical, Clinical, Commercial, Others).
To learn more about this report, Download Free Sample Report
France Live Biotherapeutic Products And Microbiome CDMO Market Summary
The France Live Biotherapeutic Products And Microbiome CDMO Market was valued at USD 1749.2 Million in 2025. It is forecast to reach USD 34902.6 Millionby 2033. That is a CAGR of45.41% over the period.
The France Live Biotherapeutic Products and Microbiome CDMO Market is heading toward integrated manufacturing systems that operate as a single platform for development and analytics and commercial production. The increasing complexity of live microbial therapies and the requirement for complete regulatory accountability drive the market need for CDMOs which can control product development from start to finish.Competitors who combine bioprocessing knowledge with data-based optimization methods and expandable systems will gain market advantage during the next five to seven years.
The supply of raw materials and microbial strains presents a hidden danger because it restricts access to rare or extremely delicate microbial consortia. The unavailability of standardized biological materials hinders batch production reliability which results in industrial capacity expansion delays because of production stoppages during times of high demand.The development of microbiome production processes with precision medicine systems creates new business prospects in locations that develop patient-specific treatment solutions. CDMOs which partner with diagnostic companies and data solution providers will obtain production contracts which require tailored manufacturing services.
The market needs companies to develop adaptable production facilities which enable multiple product operations while using advanced analytics to enhance scaling processes and maintain consistent results.The CDMO business operates with increased revenue certainty because it receives extended manufacturing arrangements instead of temporary project-based work.
Key Market Insights
- The French market holds the largest share of the European microbiome CDMO market because its biotech infrastructure enables its companies to produce 28% of the market share in Western Europe during 2025.
- The Paris and Lyon clusters together with government life sciences funding programs create a base for their contract manufacturing operations to drive innovation.
- The contract manufacturing market will reach 52% share from contract manufacturing services because live biotherapeutics need special anaerobic facilities for their production process.
- Early-stage strain optimization and formulation development needs to drive contract development services to become the second-largest market share holder.
- The integrated CDMO platforms will remain the fastest-growing market segment until 2033 because biotech companies choose complete outsourcing solutions. The 2025 applications of gastrointestinal disorders will reach 46% market share because researchers focus their clinical studies on gut microbiome treatments.
- The application of oncology microbiome therapies develops into the fastest-expanding field because scientists increase research studies which investigate the microbiome connection to immunotherapy results.
- The pipeline activity for metabolic and autoimmune indications is increasing which creates new possibilities for developing microbiome-based treatments.
- The France Live Biotherapeutic Products And Microbiome CDMO Market sees biopharmaceutical companies take over 60% of the market because they outsource their complex biologics production needs.
- The end-user segment of emerging biotech startups experiences the fastest growth because these startups depend on CDMOs to compensate for their lack of in-house production capacity.
- Early-stage microbiome research collaborations create niche demand which academic and research institutes fulfill through their research activities.
- The strategic partnerships between biotech firms and CDMOs establish a pathway for clinical operations to transition into commercial operations while they secure contracts that bring in revenue for a long time.
- The growing number of GMP-certified facilities throughout France enables companies to increase their operational capacity while they maintain their compliance with regulatory requirements.
- Companies obtain a competitive advantage because they provide complete service packages which include research and development as well as clinical manufacturing services and commercialization support.
- The company achieves market differentiation through its ongoing investment in microbiome analytics and strain engineering research which establishes its position in the market.
What are the Key Drivers, Restraints, and Opportunities in the France Live Biotherapeutic Products And Microbiome CDMO Market?
The France Live Biotherapeutic Products and Microbiome CDMO Market experiences its primary growth because clinical-stage microbiome therapeutics develop rapidly through improvements in sequencing technologies and strain-specific targeting techniques. The development of these technologies enables scientists to advance microbiome research from its early exploration phase into successful drug development pipelines which successfully target gastrointestinal and oncology diseases. Biotech companies encounter both capacity restrictions and compliance requirements after their candidates enter Phase II and III testing. The need for specialized CDMOs with anaerobic manufacturing capabilities and GMP-certified facilities has increased because research organizations require research work which transforms into manufacturing contracts that generate consistent income for long periods.
The most significant restraint is the complex and evolving regulatory framework governing live biotherapeutic products in Europe. These therapies employ living organisms which require strict containment and stability testing and batch consistency evaluation whereas traditional biologics do not need these measures. The regulatory system for this industry has not established clear standards for product classification and safety evaluation which causes extended approval times and raised development expenses. The structural barrier needs extended time for resolution because it requires multiple agencies to achieve operational agreement through extensive testing which delays market introduction and reduces short-term income generation.
A major opportunity lies in the integration of AI-driven microbiome analytics with CDMO manufacturing platforms. The companies that develop data-based solutions for strain optimization and predictive formulation development will achieve faster product development times and higher chances of clinical success. The partnerships between French microbiome startups and advanced analytics firms enable personalized therapeutic design through their mutual work.
What Has the Impact of Artificial Intelligence Been on the France Live Biotherapeutic Products And Microbiome CDMO Market?
The France Live Biotherapeutic Products and Microbiome CDMO Market experiences operational changes through artificial intelligence which creates automated systems that manage advanced biological production processes. The CDMOs now use AI-based control systems to track fermentation parameters including oxygen sensitivity and pH stability and microbial growth throughout the process. The systems perform automatic process parameter adjustments which decrease the need for human operators while they decrease the variability of live biotherapeutic production batches.
Machine learning models provide predictive modeling capabilities which manufacturing environments use across their entire operations. The predictive maintenance algorithms help facilities detect equipment failures which cause unplanned downtime by monitoring bioreactor and containment system early signs of failure. AI-based analytics enable process optimization through forecasting yield fluctuations and discovering the best culture parameters which leads to better batch results and decreased material waste.
The technologies boost operational efficiency by increasing production time while they help companies meet GMP regulations and decrease batch processing costs through their operational benefits. CDMOs who implement data-based decision-making processes experience measurable improvements in batch success rates and faster scaling operations
The implementation process faces limitations because companies need both quality biological data and affordable AI solutions for their existing manufacturing systems. The predictive models create accuracy problems because they depend on incomplete data which hampers smaller facilities from using them.
Key Market Trends
- The microbiome drug pipelines in France have reached their current state of development after entering Phase II and III trials because CDMOs need to build additional GMP-certified anaerobic manufacturing facilities.
- The period between 2020 and 2025 witnessed a complete shift in outsourcing behavior when more than 65% of biotech companies chose to work with end-to-end CDMO providers instead of using multiple fragmented service companies.
- The implementation of closed-system bioprocessing technologies by CDMOs since 2022 has enhanced their ability to control contamination while producing oxygen-sensitive microbial strains with consistent quality.
- The introduction of clear live biotherapeutic guidelines by France and EU regulatory agencies after 2021 created less uncertainty for businesses but resulted in higher expenses for compliance and increased documentation needs.
- The partnership between Evotec and microbiome startups has resulted in faster co-development processes since 2023 because their collaborations enable quicker transitions from research to clinical production.
- Digital bioprocessing tools together with AI-powered analytics solutions have become popular since 2022 because they make it possible to forecast production results while decreasing the occurrence of batch failures throughout CDMO operations.
- The COVID-19 pandemic transformed investment trends which led to a 40% rise in microbiome funding throughout France between 2021 and 2024.
- CDMOs now focus on modular facility design as their main priority since 2023 because this design lets them reach production targets at different speeds while producing multiple live biotherapeutic products.
- The entrance of international companies into France created tougher competition for local CDMOs, who needed to establish their unique identities through complete service solutions and their proficiency in microbiome research.
- The demand for microbiome-based therapeutic approaches after 2022 extended beyond their previous gastrointestinal applications to include both oncology and immunology fields because clinical evidence started to prove their effectiveness..
France Live Biotherapeutic Products And Microbiome CDMO Market Segmentation
By Type
- The manufacturing services sector generates the highest revenue because live biotherapeutic production requires both specialized anaerobic environments and containment systems and GMP-certified facilities. The developers face two main obstacles which include high capital requirements and strict regulatory standards that force them to use established CDMOs for their production needs. The manufacturing providers maintain a steady flow of work because the system creates permanent requirements and extended business agreements.
- The drug development services segment ranks as the second largest operational sector because increasing clinical-stage pipelines create demand for strain optimization and formulation design and stability testing. The regulatory environment requires product characterization and batch consistency testing which has increased the demand for analytical services. The forecast period will see integrated service platforms that provide development and analytics and manufacturing services grow because these platforms enable providers to obtain more valuable contracts while keeping their customers.

To learn more about this report, Download Free Sample Report
By Application
- Microbiome therapeutics hold the largest market share because research primarily targets digestive and immune system disorders. Pharmaceutical companies now develop specific microbial treatment solutions which emerged after 2020 through advancements in sequencing and biomarker discovery methods. The segment maintains its revenue base because additional candidates reach the final testing stage.
- Clinical trials operate as the second most significant application because Phase II and III studies increase throughout France and all of Europe. The development of personalized medicine has become the most rapidly expanding field because advances enable doctors to create custom treatments through microbiome analysis. The upcoming growth of the industry will rely on clinical validation success because it will create new treatment possibilities and draw funding for microbiome-based medical systems.
By End-User
- Pharma companies account for the largest share because large-scale drug developers require reliable CDMO partners to manage complex biologics manufacturing and regulatory compliance. Established firms prefer to outsource work because it helps them minimize their capital costs and enables faster product delivery to customers. This behavior creates stronger relationships between companies and their service providers because it establishes permanent supply contracts which bring in consistent income streams.
- Biotech firms represent the fastest-growing end-user segment as startups dominate early innovation in microbiome science but lack production infrastructure. Research institutes contribute to early-stage demand through collaborations and pilot-scale studies. Hospitals remain a smaller segment but show gradual involvement in clinical validation programs which may expand as microbiome therapies move closer to routine medical practice.
By Scale
- Most microbiome candidates operate in Phase II and III pipelines which establish clinical-scale production as the leading approach. The current stage demands both flexible manufacturing capabilities and complete adherence to requirements which results in ongoing need for CDMO expertise. The period after 2021 has experienced faster progress from preclinical research to clinical development because of increased funding and better regulatory understanding.
- The market for commercial-scale manufacturing has become the fastest expanding sector because specific therapies need substantial production capacity to reach market approval. Preclinical services continue to support early innovation but they generate only a minor part of the total revenue. The market will reach its mature phase through commercial-scale capacity expansion, which will also enable businesses to establish long-term supply agreements and international distribution arrangements.
What are the Key Use Cases Driving the France Live Biotherapeutic Products And Microbiome CDMO Market?
The primary application of microbiome therapeutics exists in the treatment of gastrointestinal diseases, which include inflammatory bowel disease and Clostridioides difficile infections. Drug developers depend on CDMOs to scale oxygen-sensitive microbial strains under strict GMP conditions, which creates consistent demand for specialized manufacturing and formulation services.
Biotech companies and research institutions are increasing their clinical trial support and drug discovery applications. Early-stage companies use CDMOs to conduct strain screening and biomarker validation while developing pilot-scale production capabilities which enable their candidates to progress through regulated trial pathways. Researchers are studying oncology-focused microbiome research to understand how gut bacteria affect immunotherapy results.
The development of personalized medicine produces a new application area which uses microbiome profiling to create customized treatment solutions for individual patients. The healthcare industry develops preventive applications which use microbiome modulation to treat metabolic disorders, and these applications will create new business opportunities as clinical testing results become more reliable.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 1749.2 Million |
|
Market size value in 2026 |
USD 2539.8 Million |
|
Revenue forecast in 2033 |
USD 34902.6 Million |
|
Growth rate |
CAGR of 45.41% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
France |
|
Key company profiled |
Lonza, Catalent, Evotec, WuXi AppTec, Samsung Biologics, Thermo Fisher, Charles River, Recipharm, AGC Biologics, Boehringer Ingelheim, Fujifilm Diosynth, Curia, AbbVie, Pfizer CentreOne, Patheon. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Drug Development Services, Manufacturing Services, Analytical Services, Others); By Application (Microbiome Therapeutics, Clinical Trials, Drug Discovery, Personalized Medicine, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Hospitals, Others); By Scale (Preclinical, Clinical, Commercial, Others). |
Which Regions are Driving the France Live Biotherapeutic Products And Microbiome CDMO Market Growth?
Biotech organizations and regulatory bodies and research facilities locate their operations in Île-de-France which enables the region to dominate the market. Public funding programs and national health authority access for clinical approval processes, which operate from Paris, create a base for the city to establish its controlling position. The system which connects CDMOs and academic laboratories and venture capital companies enables the development of microbiome treatments to proceed through their innovative process and market expansion. The system enables companies to travel their product from early research through to clinical production, which helps the region maintain its top position.
The Auvergne-Rhône-Alpes region maintains its position as a second stable region which includes Lyon as its primary center for biopharmaceutical production. )The region has developed its industrial base and trained workforce, which enables its economy to operate without depending on the capital-centered approach used in Île-de-France. The combination of consistent funding from mid-sized CDMOs and established connections to European supply chains produces dependable manufacturing capabilities. The company can expect constant income, which comes mainly from its clinical-scale manufacturing and process development services.
Occitanie has become the fastest-growing region because of its recent investments into biotechnology parks and its government-supported innovation hubs which exist in Toulouse and Montpellier. The region has experienced growth since 2022 because of its research facility expansion and microbiome startup support programs. Academic institutions and private companies now work together to build new pipelines which depend on CDMO assistance. The region presents strong growth opportunities for new businesses looking to expand at lower costs, while it establishes itself as a future center for innovation till 2033.
Who are the Key Players in the France Live Biotherapeutic Products And Microbiome CDMO Market and How Do They Compete?
The France Live Biotherapeutic Products and Microbiome CDMO Market shows moderate concentration because both global CDMOs and specialized biotech service providers compete for market share. The industry serves as a dual marketplace where established companies maintain their market share through their extensive size and regulatory knowledge and their pre-existing customer relationships while new companies enter the market through their specialized expertise in microbiome research. The core of competition between businesses centers on their ability to deliver integrated technology and services which customers now prefer because they seek partners that provide complete development and analytics and manufacturing solutions through one system. The competitive landscape in France research hubs uses geographic proximity to research centers which enables organizations to achieve faster collaboration and regulatory alignment.
Lonza uses its advanced biologics manufacturing systems and its effective compliance procedures to provide GMP-compliant live biotherapeutics which its clients can scale up to commercial production. Evotec develops technology-based partnerships which combine AI-enabled drug discovery with manufacturing support to accelerate clinical progress. Recipharm establishes manufacturing flexibility while expanding its operations throughout Europe to deliver affordable production services to mid-sized biotech companies.
Catalent uses its manufacturing capacity and its ability to transition products from clinical trials to commercial production to compete in the market for microbiome therapies. Aenova uses its expertise in formulation development together with its innovative dosage solutions to create unique product stabilization and delivery methods. These companies enhance their market position through facility growth and partnerships with microbiome startups and ongoing investments in unique anaerobic processing solutions.
Company List
- Lonza
- Catalent
- Evotec
- WuXi AppTec
- Samsung Biologics
- Thermo Fisher
- Charles River
- Recipharm
- AGC Biologics
- Boehringer Ingelheim
- Fujifilm Diosynth
- Curia
- AbbVie
- Pfizer CentreOne
- Patheon
Recent Development News
In January 2026, Lonza reported strong financial growth and raised its 2026 outlook following expansion of its CDMO platform. This reflects rising demand for advanced biologics and microbiome manufacturing capacity across Europe.
Source: https://pharmasource.global
In June 2024, Danone, Michelin, DMC Biotechnologies, and Crédit Agricole Centre France entered a partnership to establish the Biotech Open Platform. This initiative strengthens France’s microbial fermentation capabilities, supporting scalable microbiome production and CDMO ecosystem growth.
Source: https://www.globenewswire.com
What Strategic Insights Define the Future of the France Live Biotherapeutic Products And Microbiome CDMO Market?
The France Live Biotherapeutic Products and Microbiome CDMO Market is heading toward integrated manufacturing systems that operate as a single platform for development and analytics and commercial production. The increasing complexity of live microbial therapies and the requirement for complete regulatory accountability drive the market need for CDMOs which can control product development from start to finish.Competitors who combine bioprocessing knowledge with data-based optimization methods and expandable systems will gain market advantage during the next five to seven years.
The supply of raw materials and microbial strains presents a hidden danger because it restricts access to rare or extremely delicate microbial consortia. The unavailability of standardized biological materials hinders batch production reliability which results in industrial capacity expansion delays because of production stoppages during times of high demand.
The development of microbiome production processes with precision medicine systems creates new business prospects in locations that develop patient-specific treatment solutions. CDMOs which partner with diagnostic companies and data solution providers will obtain production contracts which require tailored manufacturing services.
The market needs companies to develop adaptable production facilities which enable multiple product operations while using advanced analytics to enhance scaling processes and maintain consistent results.
France Live Biotherapeutic Products And Microbiome CDMO Market Report Segmentation
By Type
- Drug Development Services
- Manufacturing Services
- Analytical Services
- Others
By Application
- Microbiome Therapeutics
- Clinical Trials
- Drug Discovery
- Personalized Medicine
- Others
By End-User
- Pharma Companies
- Biotech Firms
- Research Institutes
- Hospitals
- Others
By Scale
- Preclinical
- Clinical
- Commercial
- Others
Frequently Asked Questions
Find quick answers to common questions.
The confirmed 2033 market size figure is USD 34902.6 Million.
Key segments for the France Live Biotherapeutic Products And Microbiome CDMO Market are [By Type (Drug Development Services, Manufacturing Services, Analytical Services, Others); By Application (Microbiome Therapeutics, Clinical Trials, Drug Discovery, Personalized Medicine, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Hospitals, Others); By Scale (Preclinical, Clinical, Commercial, Others).
Major France Live Biotherapeutic Products And Microbiome CDMO Market players are Lonza, Catalent, Evotec, WuXi AppTec, Samsung Biologics, Thermo Fisher, Charles River, Recipharm, AGC Biologics, Boehringer Ingelheim, Fujifilm Diosynth, Curia, AbbVie, Pfizer CentreOne, Patheon.
The France Live Biotherapeutic Products And Microbiome CDMO Market size is USD 1749.2 Million in 2025.
The France Live Biotherapeutic Products And Microbiome CDMO Market CAGR is 45.41% from 2026 to 2033.
- Lonza
- Catalent
- Evotec
- WuXi AppTec
- Samsung Biologics
- Thermo Fisher
- Charles River
- Recipharm
- AGC Biologics
- Boehringer Ingelheim
- Fujifilm Diosynth
- Curia
- AbbVie
- Pfizer CentreOne
- Patheon
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


