France Microbiome Therapeutics Market Size & Forecast:
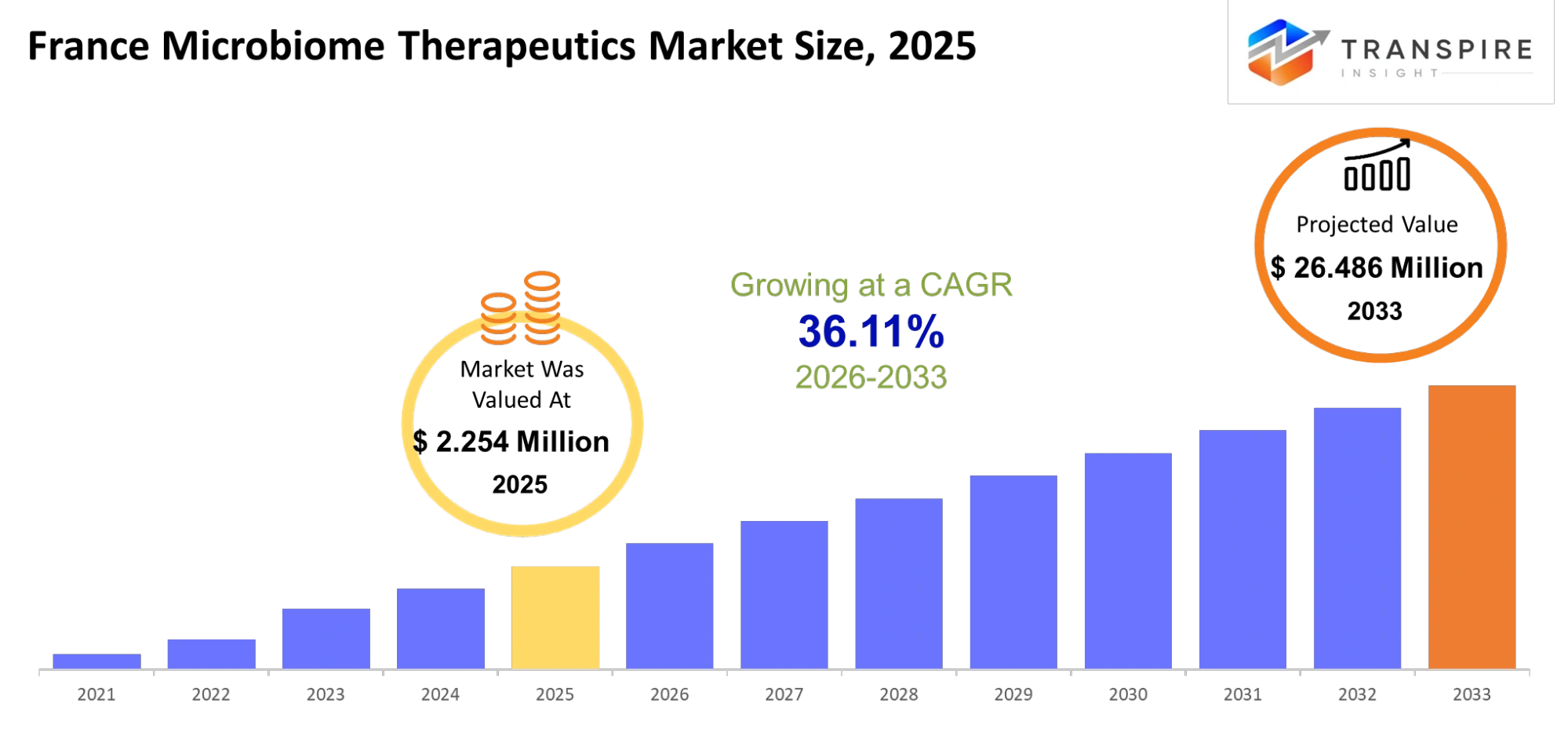
- France Microbiome Therapeutics Market Size 2025: USD 2.254 Million
- France Microbiome Therapeutics Market Size 2033: USD 26.486 Million
- France Microbiome Therapeutics Market CAGR: 36.11%
- France Microbiome Therapeutics Market Segments: By Type (Probiotics, Prebiotics, Synbiotics, Fecal Microbiota Transplants, Others); By Application (Gastrointestinal Disorders, Metabolic Disorders, Immune Diseases, Oncology, Others); By End-User (Hospitals, Clinics, Research Institutes, Consumers, Others); By Distribution (Hospital Pharmacies, Retail Pharmacies, Online, Others)
To learn more about this report, Download Free Sample Report
France Microbiome Therapeutics Market Summary
The France Microbiome Therapeutics Market was valued at USD 2.254 Million in 2025. It is forecast to reach USD 26.486 Million by 2033. That is a CAGR of 36.11% over the period.
The France microbiome therapeutics market focuses on using targeted microbial treatments to restore or modify the human microbiome to manage conditions such gastrointestinal disorders and metabolic diseases and immune dysfunction. The system enables clinicians to advance their practice because they can now use it to diagnose and treat the root causes of gut and immune system disorders.
The market has undergone fundamental changes during the last three to five years because experimental microbiota research has transitioned into clinically validated live biotherapeutic products which researchers support through advancements in sequencing and strain engineering. The post-COVID research boom in clinical microbiome studies became the main factor which led to increased funding and accelerated research progress from laboratory discoveries until they reached clinical testing.
Microbiome therapies have become established treatment options which hospitals and biotech companies now use to treat resistant medical cases according to the current trend. The investment in live biotherapeutic products has increased because regulatory frameworks for these products have started to become established, which results in quicker product launch timelines and better financial returns from advanced clinical trials.
Key Market Insights
- Ile-de-France will maintain its position as the leading region in 2025 with approximately 45 percent of the market because it possesses strong biotech clusters and hospital research networks and advanced microbiome research facilities.
- The region of Occitanie will experience its highest growth rate during the period from 2023 to 2033 because of its expanding biotech incubators located in Toulouse and Montpellier together with the increasing availability of early-stage funding.
- The market share for live biotherapeutic products reaches its highest point because these treatments serve as the primary solution for hospitals that treat gastrointestinal and immune disorders.
- The France Microbiome Therapeutics Market reaches its highest market share through live biotherapeutic products which treat gastrointestinal and immune system disorders.
- The second largest market segment of fecal microbiota transplantation-based therapies exists because this treatment shows established clinical success against recurrent C. difficile infections.
- The growth of engineered microbiome therapies today moves forward through two main factors: synthetic biology platforms that enhance strain design accuracy and increasing market demand which will boost growth from 2024.
- The treatment of gastrointestinal disorders will reach a market share of 50 percent in 2025 because patients need treatment for IBS and inflammatory bowel disease.
- The fastest-growing applications develop through metabolic and immune-related disorders which microbiome and gut-brain research studies support.
- Hospitals lead the market with their 55 percent end-user adoption because healthcare facilities need to monitor complex microbiome treatments which demand special administration methods.
- The biotech companies Seres Therapeutics and Enterome and Ferring Pharmaceuticals and Nestlé Health Science and BiomX create new solutions through their partnerships and development of engineered strains.
- The France Microbiome Therapeutics Market provides companies with competitive benefits through their ability to use AI for microbiome mapping and speed up clinical trials and develop partnerships.
What are the Key Drivers, Restraints, and Opportunities in the France Microbiome Therapeutics Market?
The France microbiome therapeutics market develops because clinical validation of live biotherapeutic products progresses throughout their development. Researchers can use technological advancements to perform exact microbial treatments which target gastrointestinal and immune system disorders. The increasing clinical trial success rates drive hospitals and biotech companies to implement microbiome-based therapies which result in more funding and faster development of new treatments. The lack of common regulatory standards and manufacturing standards for live biotherapeutic products creates a major obstacle which hampers industry development.
The existing structural barrier prevents businesses from achieving consistent production levels while ensuring product safety and effectiveness during different production runs. The absence of clear regulatory guidelines leads to prolonged approval processes which decreases investor trust and hampers the development of widespread clinical practices. The scientific achievements of multiple therapies have not yet translated into financial success because they remain in lengthy testing periods.
Engineered microbiome therapies that use synthetic biology and AI-based strain design represent a significant business opportunity. The Île-de-France and Occitanie research clusters now use computational biology systems to create clinical testing processes. AI-based microbiome mapping provides precise solutions for treating gut diseases. The progress of these technologies will enable development of personalized treatment solutions which will produce a new profitable market for investors and biotech firms.
What Has the Impact of Artificial Intelligence Been on the France Microbiome Therapeutics Market?
The France microbiome therapeutics market experiences transformation through artificial intelligence which enhances microbial data analysis, therapeutic candidate identification, and clinical pipeline optimization. The AI-driven bioinformatics platforms enable automatic microbiome sequencing analysis which decreases the time needed for researchers to identify functional microbial strains that cause diseases including inflammatory bowel disease and metabolic disorders. The process has created better results for biotherapeutic product developers who work in their initial phase of product development.
Machine learning models enable healthcare professionals to forecast patient reactions through microbiome composition analysis which assists them in selecting appropriate patients for clinical trials. The predictive tools assist trial design optimization by detecting probable responders which results in increased success rates and decreased development expenses. In manufacturing and formulation, AI supports process optimization by modeling strain stability and fermentation conditions, improving batch consistency and reducing production variability.
The operational activities of companies result in faster candidate selection processes and better trial execution, with some research pipelines experiencing early-stage screening times that decrease by 20 to 30 percent. The absence of extensive standardized microbiome databases throughout France and Europe represents the primary barrier at this point. The existing data fragmentation hinders model development while it simultaneously delays medical microbiome AI system implementation across hospitals.
Key Market Trends
- The France Microbiome Therapeutics Market experienced its first clinical-stage pipeline growth when 60% of its research activities reached that stage between 2020 and 2025.
- The regulatory agencies in France established better live biotherapeutic regulations after 2022 which helped scientists obtain product approvals and attract more funding to their biotech research.
- The hospitals adopted new treatment guidelines in 2023 to treat recurrent gastrointestinal disorders through microbiome-based treatments while decreasing their dependence on extended antibiotic treatment.
- The adoption of AI-powered microbiome sequencing technology developed quickly after 2023 which led to improvements in strain identification results and accelerated discovery processes for therapeutic candidates.
- Enterome and Ferring Pharmaceuticals expanded their research activities from 2024 to 2025 when they evaluated their microbiome-based therapies against immune system disorders.
- The funding of synthetic biology startups changed after 2022 when investors focused their funds on engineered microbial strain development and targeted delivery technology.
- The Occitanie biotech cluster experienced fast growth after 2023 because of research activities from Toulouse research institutes and increased government funding for microbiome research.
- The demand for personalized microbiome treatments increased after 2024 because patients with inflammatory bowel disease and metabolic disorders required specific treatment solutions.
- The number of CDMO manufacturing partnerships increased after 2023 which enabled biotech companies to scale their production of live biotherapeutics while decreasing their product development times.
- The rate of successful clinical validations improved after 2022 which led to higher investor trust and better progress tracking for companies developing microbiome-based therapeutics.
France Microbiome Therapeutics Market Segmentation
By Type
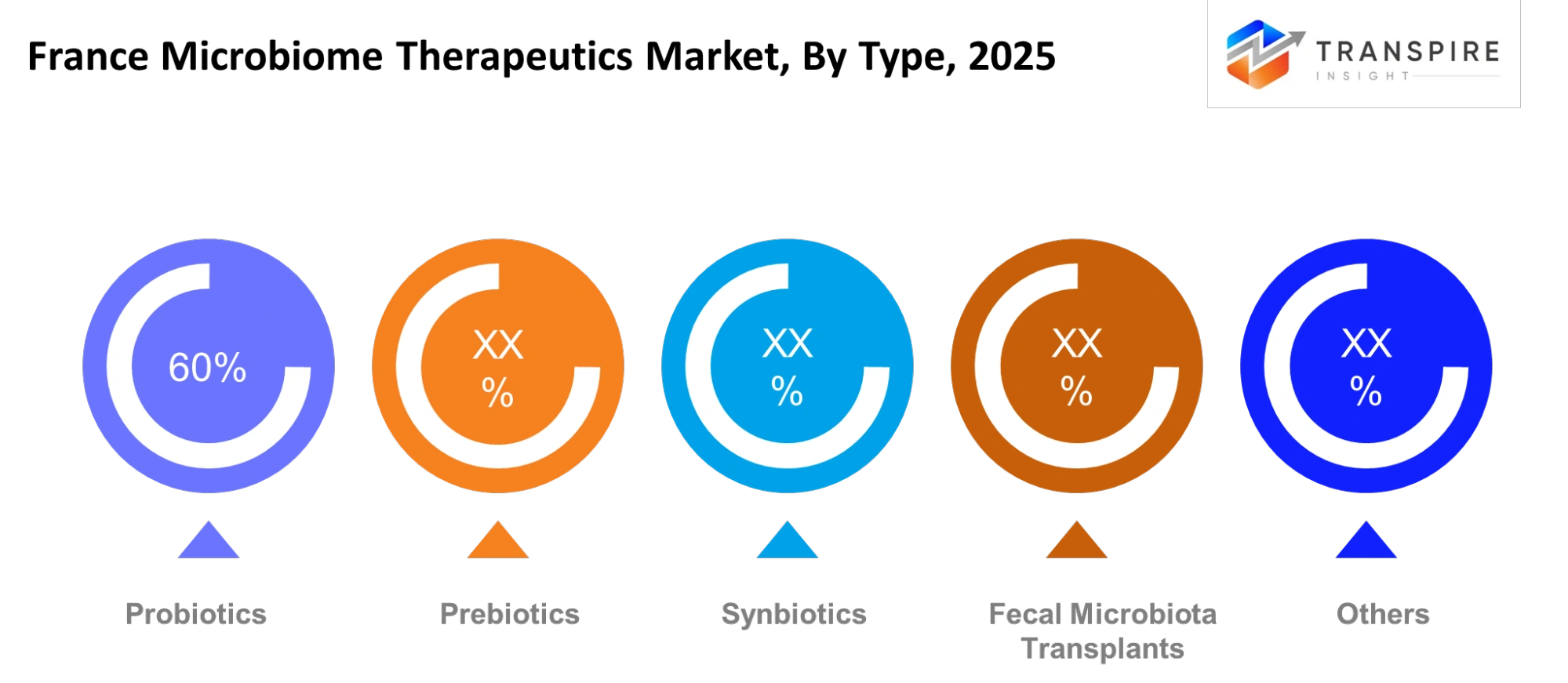
- Probiotics hold the largest share because established clinical usage in gut health management supports widespread adoption across gastrointestinal and preventive care applications. Hospitals and clinics prefer probiotic-based interventions because these treatments demonstrate safety and provide simple administration methods and strong evidence to restore microbial balance. The early dominance of this sector over advanced microbiome therapies occurred because historical treatments were more accessible and encountered fewer regulatory obstacles.
- This segment experiences growth because its products are used as complementary treatments in antibiotic recovery programs and chronic digestive disorder therapies. The market entry for engineered solutions faces challenges because their current accuracy falls short of the existing microbial solutions.Probiotics will maintain steady demand during the forecast period while their market share will decrease because advanced therapeutic modalities will receive increased clinical acceptance.
To learn more about this report, Download Free Sample Report
By Application
- The application field shows strong gastrointestinal disorder presence because microbiome treatments receive solid clinical proof for their effectiveness against IBS and inflammatory bowel disease. The existing hospital treatment routes for patients who show high disease rates create a situation which generates steady need for medical services. The medical field operates between established practices and ongoing research which creates a sustainable flow of patients needing treatment.
- The replacement of antibiotics in chronic gut conditions with microbiome-based treatments drives business expansion. The various treatment methods used by different patients lead to unpredictable results. The need for services will continue to grow throughout time but will start to shift when microbiome therapies begin treating other diseases.
By End-User
- The hospital sector maintains its highest market share because hospitals need to manage live biotherapeutic products and advanced microbiome treatments through controlled administration. The capability of hospitals to deliver complex treatments enables them to maintain their position as market leaders through their diagnostic testing and clinical oversight functions. The established infrastructure provides a reliable method to treat patients with severe and chronic conditions.
- The number of clinics is increasing because outpatient microbiome treatments have become more standardized and their administration has become less complex. The decentralized care model enables hospitals to provide treatment to a larger number of patients who need medical care outside of hospital settings. The market for medical products experiences growth because patients can now access safer treatment methods which become available through simplified treatment methods.
By Distribution
- Hospital pharmacies control distribution because they need to follow strict storage rules and their system for distributing live biotherapeutic products. The inpatient treatment systems require integration to guarantee safe patient care and hospital regulations. The channel operates as a vital link to deliver advanced microbiome therapies and prescription-based microbiome treatments.
- Retail pharmacies handle an increasing volume of probiotic and prebiotic products distribution because preventive health adoption rates continue to rise. The public uses products because they find them easy to access and use. The market for high-value products remains restricted because advanced therapies are not widely available.
What are the Key Use Cases Driving the France Microbiome Therapeutics Market?
Hospitals use microbiome-based treatments to treat IBS and inflammatory bowel disease which serves as the primary application for gastrointestinal disorder control. The strongest factor driving adoption occurs through hospital settings which use established clinical evidence to deliver treatment under secure conditions that produce identifiable results.
Clinics and specialty centers now use microbiome interventions to treat chronic conditions in patients with metabolic diseases and immune system disorders. Hospitals are increasingly adopting oncology support therapy which uses microbiome modulation to enhance patient immunotherapy results while decreasing side effects from treatment.
Scientists study gut-brain axis connections to brain and mental health disorders which shows potential for neurological treatment development while dermatological medicine uses microbiome restoration to treat skin disorders. Research institutes and biotech firms are actively testing these areas through early-stage clinical trials, positioning them for future commercialization.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 2.254 Million |
|
Market size value in 2026 |
USD 3.061 Million |
|
Revenue forecast in 2033 |
USD 26.486 Million |
|
Growth rate |
CAGR of 36.11% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
France |
|
Key company profiled |
Seres Therapeutics, Vedanta Biosciences, Ferring Pharmaceuticals, Pfizer, Novartis, Roche, Enterome, MaaT Pharma, Finch Therapeutics, Synlogic, Rebiotix, 4D Pharma, Evelo Biosciences, Yakult, Nestle |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Probiotics, Prebiotics, Synbiotics, Fecal Microbiota Transplants, Others); By Application (Gastrointestinal Disorders, Metabolic Disorders, Immune Diseases, Oncology, Others); By End-User (Hospitals, Clinics, Research Institutes, Consumers, Others); By Distribution (Hospital Pharmacies, Retail Pharmacies, Online, Others) |
Which Regions are Driving the France Microbiome Therapeutics Market Growth?
The France microbiome therapeutics market depends on Île-de-France because the region contains numerous biomedical research institutes and biotechnology companies and advanced clinical trial facilities. Research activities in early microbiome studies and their practical applications stem from hospitals and academic institutions located in Paris. The combination of substantial government funding for precision medicine research and the nearby presence of regulatory agencies enables rapid product approvals and product development processes. The system facilitates faster transitions from laboratory research to clinical testing which strengthens the area's research capabilities.
The pharmaceutical manufacturing industry and life sciences sector of Auvergne-Rhône-Alpes create a strong foundation that establishes the region as a reliable second market position. Lyon serves as a primary location for bioproduction activities and clinical research activities because international pharmaceutical companies maintain their ongoing financial support. This region prioritizes production capabilities and industrial operational stability instead of focusing on initial phase research and development which distinguishes it from the capital region. The country can rely on this system because it maintains consistent economic strength and implements long-term plans for industrial growth.
Occitanie experiences the highest growth rate because biotech startup activities in Toulouse and Montpellier continue to rise while public funding for life sciences research increases. The expansion of research parks and academic partnerships has improved the region's ability to conduct early microbiome research. The 2022 regional support program enhancements have resulted in more efficient startup creation and clinical trial operations. The region provides market entrants and investors with high-growth opportunities because they can access new microbiome technologies while encountering limited market competition.
Who are the Key Players in the France Microbiome Therapeutics Market and How Do They Compete?
The France microbiome therapeutics market displays a competitive environment which includes various specialized biotech companies and international pharmaceutical firms that compete against each other. The initial development of new products depends on biotech startups while established pharmaceutical companies work to advance their approved medicines through testing and market distribution. The competition operates through technological proficiency which includes strain engineering capabilities and clinical validation results and regulatory advancement but the treatments focus on complex diseases with high value.
MaaT Pharma competes through its focus on microbiome ecosystem restoration therapies for oncology patients undergoing stem cell transplantation. The company establishes its unique market position through standardized microbiota-based drug candidates which utilize advanced manufacturing methods to produce live biotherapeutics. The company expands through European clinical trial collaborations and oncology-focused hospital partnerships which enhance its position in hospital-based microbiome treatment operations.
Enterome develops precision immunotherapies which use microbiome data to treat cancer and autoimmune disorders. The company gains its competitive edge through its antigen discovery platforms which connect gut microbiota patterns with immune system response mechanisms. The company expands through strategic partnerships with international pharmaceutical firms which enable them to develop upcoming immunotherapy products together.
Ferring Pharmaceuticals uses its Rebiotix subsidiary to market fecal microbiota therapies which treat recurrent infections. The company uses its advanced clinical achievements and regulatory microbiome restoration product approvals to create unique market value. The company expands through its European regulatory procedures and the development of manufacturing technologies which enable production capacity upgrades.
- Seres Therapeutics
- Vedanta Biosciences
- Ferring Pharmaceuticals
- Pfizer
- Novartis
- Roche
- Enterome
- MaaT Pharma
- Finch Therapeutics
- Synlogic
- Rebiotix
- 4D Pharma
- Evelo Biosciences
- Yakult
- Nestle
Recent Development News
In November 2025, MaaT Pharma announced a €9.1 million capital increase to support development and commercialization of its lead microbiome therapy Xervyteg in Europe. The funding strengthens late-stage clinical progress and prepares the company for regulatory submission and potential market launch in 2026.
https://www.maatpharma.com
In September 2025, MRM Health secured €55 million in Series B financing to accelerate clinical development of its microbiome-based biotherapeutic pipeline and expand manufacturing capabilities. The investment supports advancement of engineered microbiome therapies targeting inflammatory and metabolic diseases across Europe.
https://en.wikipedia.org
What Strategic Insights Define the Future of the France Microbiome Therapeutics Market?
The France microbiome therapeutics market displays a competitive environment which includes various specialized biotech companies and international pharmaceutical firms that compete against each other. The initial development of new products depends on biotech startups while established pharmaceutical companies work to advance their approved medicines through testing and market distribution. The competition operates through technological proficiency which includes strain engineering capabilities and clinical validation results and regulatory advancement but the treatments focus on complex diseases with high value.
MaaT Pharma competes through its focus on microbiome ecosystem restoration therapies for oncology patients undergoing stem cell transplantation. The company establishes its unique market position through standardized microbiota-based drug candidates which utilize advanced manufacturing methods to produce live biotherapeutics. The company expands through European clinical trial collaborations and oncology-focused hospital partnerships which enhance its position in hospital-based microbiome treatment operations.
Enterome develops precision immunotherapies which use microbiome data to treat cancer and autoimmune disorders. The company gains its competitive edge through its antigen discovery platforms which connect gut microbiota patterns with immune system response mechanisms. The company expands through strategic partnerships with international pharmaceutical firms which enable them to develop upcoming immunotherapy products together.
Ferring Pharmaceuticals uses its Rebiotix subsidiary to market fecal microbiota therapies which treat recurrent infections. The company uses its advanced clinical achievements and regulatory microbiome restoration product approvals to create unique market value. The company expands through its European regulatory procedures and the development of manufacturing technologies which enable production capacity upgrades.
France Microbiome Therapeutics Market Report Segmentation
By Type
- Probiotics
- Prebiotics
- Synbiotics
- Fecal Microbiota Transplants
- Others
By Application
- Gastrointestinal Disorders
- Metabolic Disorders
- Immune Diseases
- Oncology
- Others
By End-User
- Hospitals
- Clinics
- Research Institutes
- Consumers
- Others
By Distribution
- Hospital Pharmacies
- Retail Pharmacies
- Online
- Others
Frequently Asked Questions
Find quick answers to common questions.
The confirmed 2033 market size figure in USD 26.486 Million.
Key segments for the France Microbiome Therapeutics Market are By Type (Probiotics, Prebiotics, Synbiotics, Fecal Microbiota Transplants, Others); By Application (Gastrointestinal Disorders, Metabolic Disorders, Immune Diseases, Oncology, Others); By End-User (Hospitals, Clinics, Research Institutes, Consumers, Others); By Distribution (Hospital Pharmacies, Retail Pharmacies, Online, Others).
Major France Microbiome Therapeutics Market players are Seres Therapeutics, Vedanta Biosciences, Ferring Pharmaceuticals, Pfizer, Novartis, Roche, Enterome, MaaT Pharma, Finch Therapeutics, Synlogic, Rebiotix, 4D Pharma, Evelo Biosciences, Yakult, Nestle .
The France Microbiome Therapeutics Market size is USD 2.254 Million in 2025.
The France Microbiome Therapeutics Market CAGR is 36.11% from 2026 to 2033.
- Seres Therapeutics
- Vedanta Biosciences
- Ferring Pharmaceuticals
- Pfizer
- Novartis
- Roche
- Enterome
- MaaT Pharma
- Finch Therapeutics
- Synlogic
- Rebiotix
- 4D Pharma
- Evelo Biosciences
- Yakult
- Nestle
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)