France Therapy Devices Market Size & Forecast:
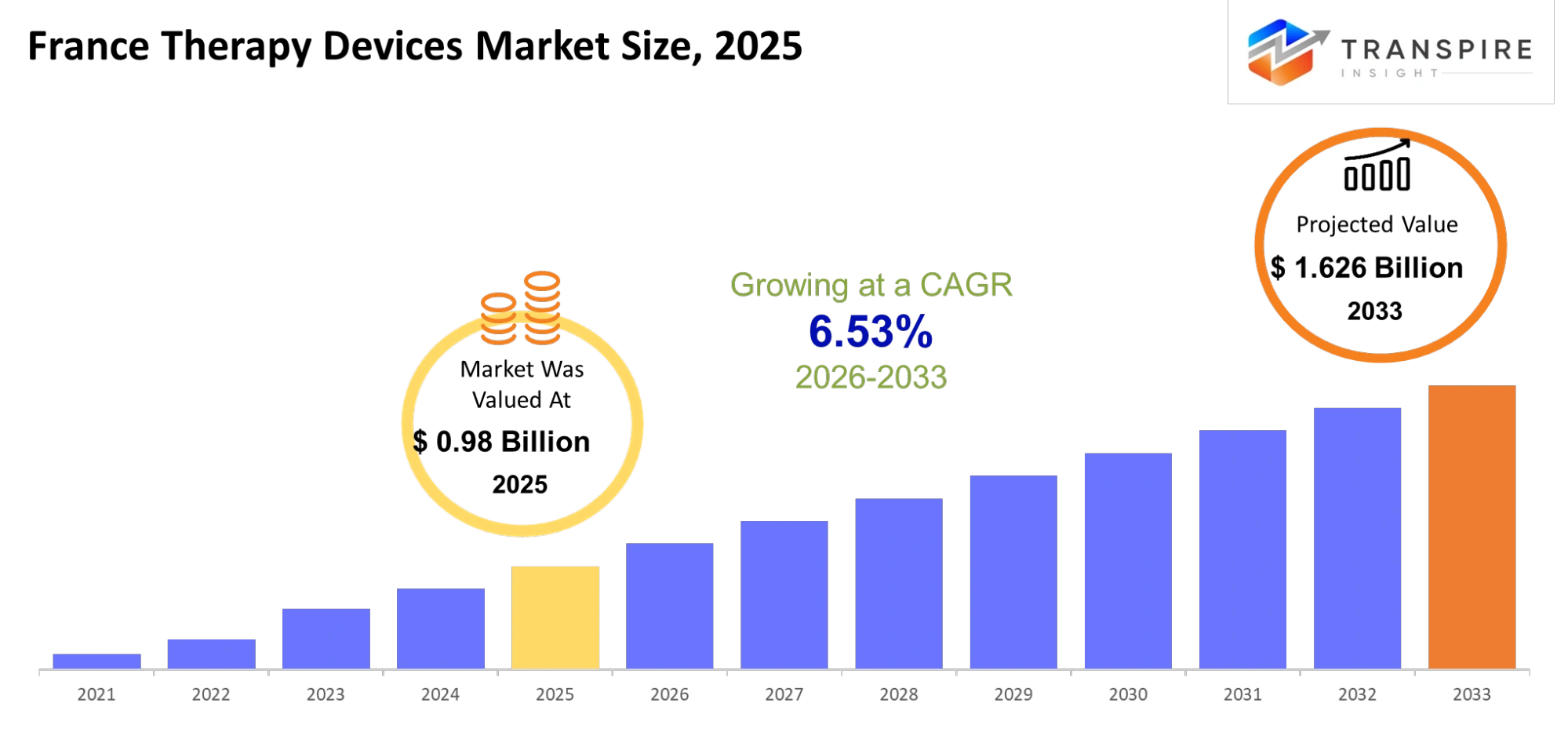
- France Therapy Devices Market Size 2025: USD 0.98 Billion
- France Therapy Devices Market Size 2033: USD 1.626 Billion
- France Therapy Devices Market CAGR: 6.53%
- France Therapy Devices Market Segments: By Type (Electrotherapy Devices, Respiratory Therapy Devices, Physical Therapy Devices, Radiation Therapy Devices, Others); By Application (Pain Management, Respiratory Disorders, Oncology, Rehabilitation, Neurological Disorders, Others); By End-User (Hospitals, Clinics, Rehabilitation Centers, Homecare, Specialty Centers, Others); By Technology (Wearable Devices, Portable Devices, Stationary Devices, Others)
To learn more about this report, Download Free Sample Report
France Therapy Devices Market Summary
The France Therapy Devices Market was valued at USD 0.98 Billion in 2025. It is forecast to reach USD 1.626 Billion by 2033. That is a CAGR of6.53%over the period.
The France Therapy Devices Market provides medical equipment for hospitals, which enables healthcare providers to treat patients who experience chronic pain and breathing difficulties and movement problems and who need to recover after surgical procedures. The devices enable faster patient recovery which allows for home treatment and ongoing medical care, which helps reduce hospital demands because of the aging population who require extended care.
The period between 2018 and 2021 saw a major market transformation which resulted in hospitals moving away from using fixed medical devices toward adopting portable medical devices that work with remote monitoring systems. The COVID-19 pandemic forced healthcare facilities to create decentralized treatment systems which enabled them to expand their home-based medical services. Manufacturers are now including digital monitoring systems together with telehealth functionalities in their products. Beyond acute care, the system generates increased income because it enables new applications for chronic disease management which results in more frequent device usage and steady demand from outpatient and homecare markets.
Key Market Insights
- The France Therapy Devices Market in 2025 will be more than 45% dominated by northern and urban areas because they have developed superior healthcare facilities.
- The southern region of France will experience its highest growth rate until 2033 because of the increasing availability of outpatient and homecare services.
- The market for respiratory therapy devices will reach a 30% market share in 2025 because the number of chronic respiratory diseases continues to increase.
- The market for pain management devices has reached its second biggest share because more people choose non-invasive treatment methods.
- The demand for rehabilitation and mobility devices has become the fastest-growing market segment because of both the elderly population and people who need help after surgical procedures.
- The market share for chronic disease management exceeds 40% because therapy devices make it possible to treat patients for extended periods without needing hospitalization.
- Home-based therapy applications represent the most rapidly expanding category because telehealth and remote monitoring capabilities have become increasingly popular.
- The hospital and clinic sector will control about 50% of the market in 2025 because they can treat many patients through their advanced medical facilities.
- Homecare settings represent the fastest-growing end-user segment, which exists because of their cost efficiency and the convenience they provide to patients. The France
- Therapy Devices Market displays intense competition between global companies which include Philips Healthcare and Medtronic and GE HealthCare and Siemens Healthineers and ResMed.
- Companies are developing new products which include portable therapy devices that use artificial intelligence technology to improve treatment results.
- The healthcare organizations in France are increasing device adoption through their efforts to establish strategic partnerships with healthcare providers.
- The regional expansion strategies will use their resources to serve both outpatient and homecare markets that currently lack proper service delivery.
- The competitiveness of companies increases through technology advancements that include IoT-enabled monitoring and data-driven therapy optimization.
What are the Key Drivers, Restraints, and Opportunities in the France Therapy Devices Market?
The France Therapy Devices Market exists because hospitals face capacity limitations which have been worsened by post-pandemic care reforms that provide better healthcare options to patients through decentralized delivery systems. French health authorities expanded reimbursement frameworks for home-based therapies, making devices such as portable respiratory and rehabilitation systems financially viable outside clinical settings. The policy change resulted in higher treatment adoption because it replaced hospital-based care with continuous treatment at home. The company earns additional revenue through its products because customers use its devices and maintenance services and pay for digital monitoring services that assist in treating patients over extended periods.
The European Union Medical Device Regulation (MDR) establishes an excessive regulatory burden which functions as a key obstacle to business operations. The process requires extensive clinical validation and post-market surveillance and documentation which results in increased costs and delays product development until sales begin. The regulatory framework establishes the structural barrier because it limits all options for achieving compliance through fast-track methods. The financial limitations faced by smaller companies result in product delays that prevent them from entering markets which results in reduced innovation capabilities and lost potential earnings across new treatment areas.
The integration of connected therapy devices with AI-powered remote patient monitoring platforms presents a significant business opportunity. Smart respiratory devices that monitor usage patterns for real-time therapy adjustments are becoming popular in outpatient medical facilities. Healthcare providers in France can implement personalized data-driven therapy solutions because the country has developed its digital health infrastructure and telemedicine capabilities.
What Has the Impact of Artificial Intelligence Been on the France Therapy Devices Market?
The artificial intelligence technology creates new methods for therapeutic device deployment in France because it combines automated systems with data-driven management to establish clinical and homecare environments. The connected devices which include CPAP systems and neurostimulation units and rehabilitation platforms now implement AI technology to automatically modify treatment based on actual patient information. The system collects data about how equipment gets used and how patients follow their treatment plan and how their body reacts to treatment which enables doctors to adjust their patient care from a distance without needing to perform any physical tasks. The system improves patient care by decreasing hospital visits while it enhances efficiency for all treatment processes.
The machine learning models provide healthcare professionals with tools to predict how equipment will function and what health results their patients will achieve. The AI algorithms enable predictions about machinery upkeep requirements while they identify initial indicators that patients will not follow their treatment schedule which enables healthcare providers to take action before their patients experience treatment decline. The use of predictive analytics in respiratory care helps healthcare professionals determine optimal airflow settings which results in better treatment outcomes and fewer medical issues. The new technologies produce higher equipment availability which leads to increased patient treatment compliance and reduced expenses throughout extended medical care.
The high costs of system integration together with the difficulties in standardizing data present a significant barrier that prevents organizations from implementing artificial intelligence technology. The majority of healthcare organizations continue to use incomplete digital systems which create obstacles for efficient data sharing and impede the growth of AI-based therapeutic solutions.
Key Market Trends
- The adoption of home-based therapy in France has increased since 2020 because more than 35% of chronic care patients will use portable or remote monitoring devices by 2025.
- Manufacturers have developed IoT systems that enable real-time data tracking and clinician dashboard access as replacements for their previous standalone devices which help improve therapy adherence and patient outcomes.
- The reimbursement reforms which came into effect after the COVID-19 pandemic introduced expanded coverage of remote therapy products which hospitals and homecare organizations started to use for their purchasing choices.
- The period from 2021 until 2025 saw a major rise in demand for portable respiratory devices which healthcare providers used to treat patients with long-term pulmonary conditions and those undergoing post-infection rehabilitation.
- Philips Healthcare and ResMed have developed cloud-connected therapy platforms which use software-based solutions to create a new competitive advantage in their market.
- The launch of AI-driven therapy devices in 2022 brought treatment customization and patient progress monitoring capabilities which reduced the need for clinical staff to conduct manual procedures.
- Hospitals are reallocating budgets toward outpatient and homecare ecosystems which will decrease their reliance on inpatient therapy infrastructure while creating new procurement methods.
- The supply chains have achieved partial localization after facing disruptions in 2021 because manufacturing hubs in Europe now serve as primary sources for essential components instead of depending on international supply networks.
- The period after 2023 brought more strategic alliances between device makers and telehealth providers which enabled seamless patient care delivery throughout France's digital health network.
- The EU MDR regulations which became stricter after 2021 have made it harder for companies to get new products approved because they now need more resources to meet clinical validation requirements and develop ongoing regulatory compliance solutions.
France Therapy Devices Market Segmentation
By Type
- The France Therapy Devices Market demonstrates respiratory therapy devices as their leading segment because patients with chronic respiratory conditions need continuous treatment which extends throughout their hospital stays and homecare periods. The establishment of home-based oxygen systems and CPAP systems with reimbursement options after 2020 led to increased adoption of pulmonary care as it achieved clinical importance. Manufacturers achieved market dominance by launching portable medical devices which support remote patient monitoring and enhance treatment compliance. The combination of medical needs and digital technology systems maintains high revenue generation through respiratory system sales.
- The demand for non-invasive treatment approaches has led to a steady growth of electrotherapy and physical therapy devices as patients increasingly choose rehabilitation methods which do not require surgical procedures or medication. Clinics and rehabilitation centers increasingly rely on these devices to improve recovery timelines and reduce patient dependency on medication. The development of wearable and programmable systems has brought better treatment accuracy which allows outpatient and homecare settings to use these systems. Radiation therapy devices maintain their specialized status because oncology needs drive development through advanced targeting systems which improve treatment accuracy.
To learn more about this report, Download Free Sample Report
By Application
- The application demand for pain management leads to its use because healthcare providers choose non-pharmacological methods to treat chronic diseases while minimizing opioid use. Electrotherapy and neurostimulation devices function as primary tools which provide exact and repeatable medical treatment that can be applied in both hospital environments and patient homes. After 2022, safer solutions for extended pain relief became the preferred choice because regulatory and clinical standards developed. The requirement for medical devices in chronic care leads to ongoing demand which creates opportunities for sustainable income streams.
- The need for continuous treatment in respiratory disorders drives the growth of rehabilitation applications which now focus on outpatient recovery methods. Pulmonary care devices and physiotherapy equipment allow healthcare professionals to track patient progress while patients recover at home without needing hospital treatment. The demand for specialized treatment systems in oncology applications maintains a constant level which supports demand for precision treatment systems in specialized centers. Advanced therapy devices now provide neurological disorder use cases which enable personalized treatment through data-driven methods to enhance patient results.
By End-User
- The usage of hospitals shows dominance because their advanced treatment systems are essential for handling intricate medical procedures and their capacity to treat numerous patients. The hospitals keep building complete treatment systems which enable them to deliver both acute and chronic medical services throughout their different medical departments. Hospitals adopt high-value specialized equipment because of strong reimbursement systems and their ability to hire expert workers. The existing position remains intact because operational expenses are pushing some medical treatments to become available through outpatient facilities and homecare services.
- The healthcare system is moving towards decentralized care delivery which creates a need for more clinics, rehabilitation centers and homecare facilities. Clinics provide basic medical services through their therapy sessions and follow-up appointments. The rehabilitation center creates demand for their services by providing structured recovery programs which help patients with post-surgical and mobility issues. Homecare services are experiencing rapid growth because patients want convenient treatments which cost less and can be used with portable medical devices that connect to the internet. Specialized treatment centers and niche medical providers are implementing cutting-edge systems to deliver specific therapies which lets them develop unique and profitable treatment options.
By Technology
- The healthcare systems of today adopt portable devices because they need flexible solutions that enable patients to receive treatment outside hospital settings. The devices enable patients to receive home-based therapy and outpatient treatment which helps them keep up their treatment schedule without needing to visit clinics. The period after 2020 experienced rapid growth when healthcare providers started using remote monitoring and telehealth services to deliver medical care. The industry maintains its market strength as manufacturers create more portable products through their development of smaller devices which include advanced connectivity options.
- The demand for wearable devices is increasing because people need continuous health monitoring and customized treatment methods to manage their chronic diseases. The systems provide real-time data monitoring with dynamic treatment adjustment capabilities which boost patient treatment compliance and medical results. Hospitals and specialized facilities depend on stationary devices to provide high-accuracy measurements and complex medical functions. The current technology environment is moving towards ecosystems that connect multiple device types with digital health systems to create better therapy methods that use data analysis.
What are the Key Use Cases Driving the France Therapy Devices Market?
The main application in France serves as a solution for managing chronic diseases which particularly affects patients with respiratory issues and persistent pain problems. The medical field uses CPAP systems and neurostimulation units as essential components for treating patients in both hospital and homecare environments. The primary need of this use case exists because it prevents hospital readmissions while allowing physicians to monitor patients over time which matches national healthcare expenditure management goals.
Healthcare providers use post-surgical rehabilitation and elderly mobility support systems to enable patients to receive treatment at outpatient clinics and assisted living facilities. Physiotherapy devices and wearable rehabilitation tools have gained popularity as medical professionals use them to help patients recover faster while gaining greater independence. Healthcare organizations are now adopting these new use cases because they want to transition towards outpatient treatment methods instead of using inpatient treatment facilities.
Artificial intelligence (AI) systems that provide personalized treatment solutions together with remote patient monitoring systems represent emerging use cases. Telehealth research programs are currently evaluating smart devices that use patient data to change treatment plans in real time. France's ongoing investment in digital health infrastructure together with its integrated care delivery systems shows promising developments for these applications.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 0.98 Billion |
|
Market size value in 2026 |
USD 1.044 Billion |
|
Revenue forecast in 2033 |
USD 1.626 Billion |
|
Growth rate |
CAGR of 6.53% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
France |
|
Key company profiled |
Philips, GE Healthcare, Siemens Healthineers, Medtronic, Abbott, Boston Scientific, Baxter, Becton Dickinson, Stryker, Zimmer Biomet, Fresenius, Hill-Rom, Nihon Kohden, Dräger, Omron . |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Electrotherapy Devices, Respiratory Therapy Devices, Physical Therapy Devices, Radiation Therapy Devices, Others); By Application (Pain Management, Respiratory Disorders, Oncology, Rehabilitation, Neurological Disorders, Others); By End-User (Hospitals, Clinics, Rehabilitation Centers, Homecare, Specialty Centers, Others); By Technology (Wearable Devices, Portable Devices, Stationary Devices, Others) |
Which Regions are Driving the France Therapy Devices Market Growth?
The Île-de-France region leads the France Therapy Devices Market, anchored by Paris's dense network of tertiary hospitals, research institutes, and specialist care centers. The device uptake at both public and private providers increases because of strong digital health policy support and home-based therapy reimbursement programs. The region benefits from advanced clinical infrastructure which allows for quick deployment of connected therapy systems and remote monitoring technologies. The presence of top healthcare organizations and research centers in the area drives continuous demand for advanced treatment solutions which medical facilities start using before their official launch.
The stable second contributor to the market operates through Auvergne-Rhône-Alpes which depends on Lyon as its main medical and industrial center. The capital region operates under different growth patterns because its development depends equally on hospital networks and rehabilitation centers and medical device manufacturing clusters. The region shows consistent investment in outpatient care and physiotherapy services which results in steady device usage throughout the year instead of sudden adoption surges. The region provides reliable income streams to the national market because of its economic stability and effective local healthcare system development.
Regional healthcare systems in Occitanie have started to develop which makes it the fastest expanding territory according to recent telemedicine system growth and healthcare system upgrade initiatives. The government has increased funding for rural healthcare access which has resulted in faster adoption of portable and home-based therapy equipment to remote and underserved regions. The region has also experienced more local healthcare organizations collaborating with digital health startups to create scalable systems for delivering remote healthcare services. The market opportunities between 2026 and 2033 will expand because the market will grow through decentralized healthcare solutions which new companies will enter.
Who are the Key Players in the France Therapy Devices Market and How Do They Compete?
The France Therapy Devices Market displays a semi-consolidated structure which allows international medical technology companies to maintain dominant market share while digital technological advancements and service delivery models create new competitive challenges. The existing market players safeguard their market share through device development which combines connectivity and analytics features with remote monitoring capabilities, which results in a shift towards integrated care systems as the new basis for competing with their products. The primary battleground has moved to technology differentiation and long-term patient management solutions, where recurring revenue from software and services is becoming as important as device sales. The existing healthcare system faces disruption from digital health companies and niche market specialists who develop patient-centered treatment systems that provide better flexibility compared to traditional healthcare approaches.
Philips Healthcare provides complete connected care ecosystems which enable cloud-based monitoring to function with its therapeutic devices that include sleep and respiratory equipment. The company achieves its unique position through its comprehensive platform system which connects patients with healthcare providers and medical data analysis tools to provide continuous patient monitoring outside of hospital environments. The company competes through technological advancements in neuromodulation and chronic disease management which help Medtronic establish physician credibility through its highly accurate medical devices and proven clinical effectiveness. GE HealthCare develops integrated hospital systems which use therapy equipment to build complete diagnostic and patient monitoring systems, thus enabling hospitals to provide consistent patient treatment throughout their facilities.
The company establishes its market position through combined digital health solutions and artificial intelligence research which create integrated systems that link medical imaging with diagnostic testing and therapeutic treatment procedures. ResMed develops as a major player in the home healthcare market through its respiratory devices which connect to the internet, while tracking system progress, which helps patients maintain their treatment schedule. Telehealth solutions across the entire market enable partnerships with various medical organizations to deliver their services.
Company List
- Philips
- GE Healthcare
- Siemens Healthineers
- Medtronic
- Abbott
- Boston Scientific
- Baxter
- Becton Dickinson
- Stryker
- Zimmer Biomet
- Fresenius
- Hill-Rom
- Nihon Kohden
- Dräger
- Omron
Recent Development News
In January 2026, Medtronic secured favorable reimbursement approval for its EVOLUT FX+ aortic valve device in France. This decision by French health authorities expands patient access and accelerates adoption of advanced cardiovascular therapy devices.https://mtrconsult.com
In July 2025, Medtronic and Philips entered a multi-year strategic partnership to expand patient monitoring technologies. The collaboration integrates next-generation monitoring solutions, strengthening connected therapy ecosystems and improving clinical outcomes.https://news.medtronic.com
What Strategic Insights Define the Future of the France Therapy Devices Market?
The France Therapy Devices Market is currently entering an operational framework which distributes treatment through continuous data-driven medical services that replace conventional treatment methods. The current market trajectory exists because home-based medical treatment receives reimbursement and healthcare facilities develop their connected health systems to create new methods for assessing value throughout patient treatment.
The European Union faces an invisible risk through its existing regulatory system and data governance rules which create separate pathways that hinder connected therapy platforms from expanding internationally while raising cost burdens on device manufacturers who develop their digital systems.
The rehabilitation and neuromodulation devices field now presents new possibilities through its implementation of AI-powered adaptive therapy systems which will benefit outpatient treatment facilities and rural healthcare centers that lack qualified medical personnel.
To maintain market position companies need to develop platforms which enable equipment and software as well as data services to work together while building partnerships with telehealth providers to establish customer relationships that produce continuous business revenue.
France Therapy Devices Market Report Segmentation
By Type
- Electrotherapy Devices
- Respiratory Therapy Devices
- Physical Therapy Devices
- Radiation Therapy Devices
- Others
By Application
- Pain Management
- Respiratory Disorders
- Oncology
- Rehabilitation
- Neurological Disorders
- Others
By End-User
- Hospitals
- Clinics
- Rehabilitation Centers
- Homecare
- Specialty Centers
- Others
By Technology
- Wearable Devices
- Portable Devices
- Stationary Devices
- Others
Frequently Asked Questions
Find quick answers to common questions.
The confirmed 2033 market size figure is USD 1.626 Billion.
Key segments for the France Therapy Devices Market areBy Type (Electrotherapy Devices, Respiratory Therapy Devices, Physical Therapy Devices, Radiation Therapy Devices, Others); By Application (Pain Management, Respiratory Disorders, Oncology, Rehabilitation, Neurological Disorders, Others); By End-User (Hospitals, Clinics, Rehabilitation Centers, Homecare, Specialty Centers, Others); By Technology (Wearable Devices, Portable Devices, Stationary Devices, Others).
Major France Therapy Devices Market players are Philips, GE Healthcare, Siemens Healthineers, Medtronic, Abbott, Boston Scientific, Baxter, Becton Dickinson, Stryker, Zimmer Biomet, Fresenius, Hill-Rom, Nihon Kohden, Dräger, Omron .
The France Therapy Devices Market size is USD USD 0.98 Billion in 2025.
The France Therapy Devices Market CAGR is 6.53% from 2026 to 2033.
- Philips
- GE Healthcare
- Siemens Healthineers
- Medtronic
- Abbott
- Boston Scientific
- Baxter
- Becton Dickinson
- Stryker
- Zimmer Biomet
- Fresenius
- Hill-Rom
- Nihon Kohden
- Dräger
- Omron
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)