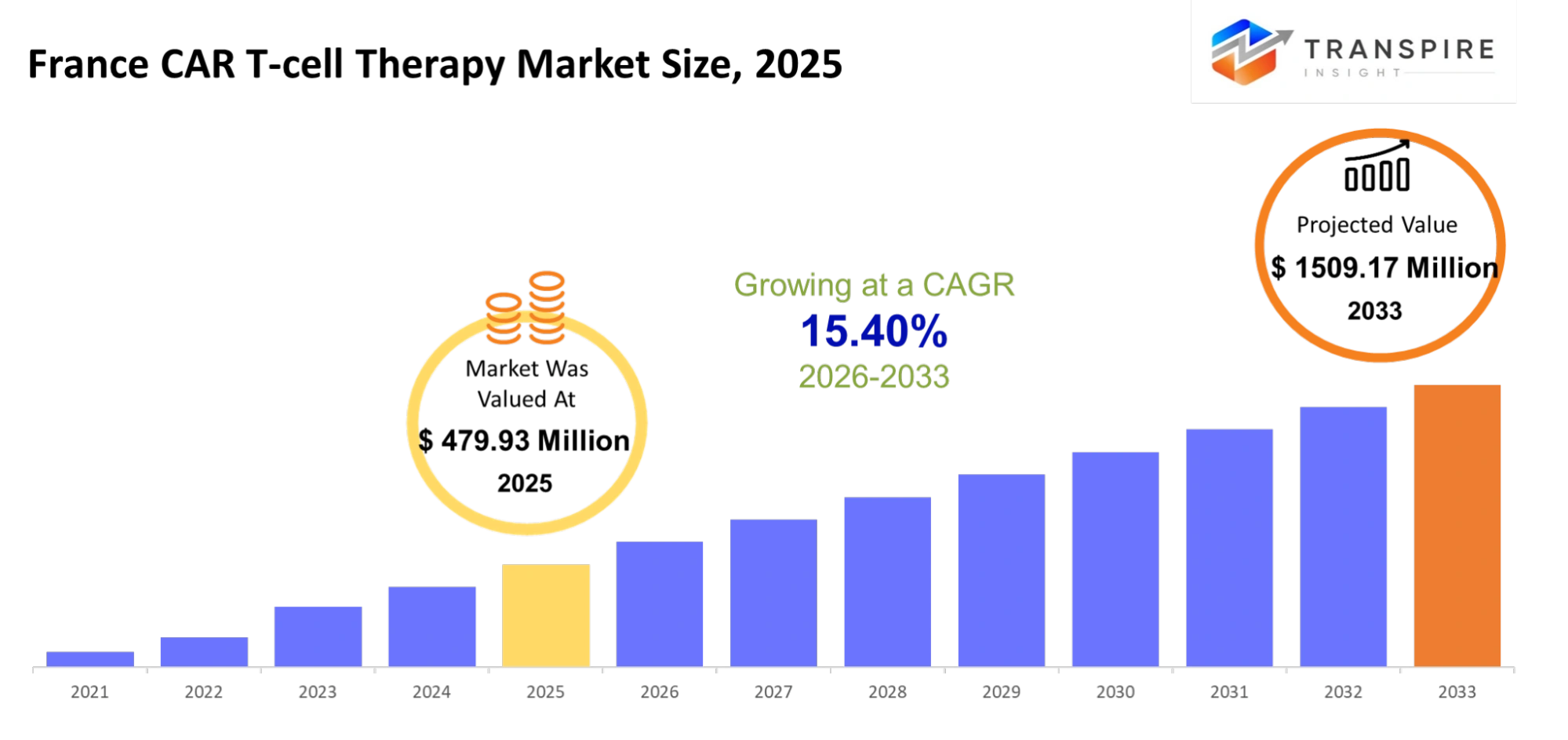
France CAR T-cell Therapy Market Size & Forecast:
- France CAR T-cell Therapy Market Size 2025: USD 479.93 Million
- France CAR T-cell Therapy Market Size 2033: USD 1509.17 Million
- France CAR T-cell Therapy Market CAGR: 15.40%
- France CAR T-cell Therapy Market Segments: By Application (Leukemia, Lymphoma, Multiple Myeloma), By End User (Hospitals, Cancer Treatment Centers),
To learn more about this report, Download Free Sample Report
France CAR T-cell Therapy Market Summary:
The France CAR T-cell Therapy Market size is estimated at USD 479.93 Million in 2025 and is anticipated to reach USD 1509.17 Million by 2033, growing at a CAGR of 15.40% from 2026 to 2033.The growth of personalized medicine together with the increasing number of clinical trials and the strong healthcare system in France which supports CAR‑T therapy development and delivery will drive market expansion.
The market mainly operates through autologous CAR-T cells which hospitals and specialized cancer centers use to deliver treatment because of the complex nature of therapy administration. Gilead Sciences together with Novartis and Bristol-Myers Squibb and Amgen and Merck support R&D through their partnerships to improve CAR-T therapies by developing better treatment methods which enhance safety and treatment availability. The combination of attractive regulatory conditions and new reimbursement systems together with increased medical knowledge about CAR-T treatments will lead to faster market growth in France which will become one of the main European markets for advanced cellular immunotherapies.
Key Market Trends & Insights:
- Rising Adoption in Oncology – Hospitals and cancer centers now use CAR‑T therapies as standard treatment for hematological cancers which include leukemia and lymphoma and multiple myeloma. Hospitals and cancer centers now use CAR‑T treatments because of their high treatment success rates and customized medical treatment methods.
- Focus on Autologous and Next-Generation Therapies – The current market shows autologous CAR‑T therapies as the main therapy which uses cells from patients because next-generation allogeneic and gene-edited CAR‑T products will enable doctors to treat patients with prebuilt systems that need less time to provide treatment and can handle more patients. The current segment will experience growth through the new innovative products that will come to market.
- Expanding Clinical Trials and Approvals – France has seen a surge in CAR‑T clinical trials targeting both blood cancers and solid tumors. Ongoing research and regulatory approvals will create new treatment options which will make next-generation CAR‑T therapies more accessible to patients.
- Hospital and Specialized Center Infrastructure – Hospitals and cancer treatment centers represent the only locations which can deliver CAR‑T therapies because of the complex requirements for preparing and storing and administering these treatments. Investment in infrastructure together with trained personnel and logistics ensures safe and effective therapy delivery which remains critical for market expansion and patient access.
- Regulatory Support and Reimbursement Evolution – CAR‑T therapy adoption receives support through French governmental regulations which establish favorable policies and develop new reimbursement systems. The measures decrease patient expenses while they motivate businesses to invest in R&D and they support the use of new immunotherapies in regular healthcare operations throughout the nation.
France CAR T-cell Therapy Market Segmentation
By Target Antigen
- CD19: The most active antigen used in CAR‑T therapies for B-cell cancers which include acute lymphoblastic leukemia and particular forms of lymphoma. The established clinical outcomes of this treatment make it the most suitable option for CAR‑T developers who operate in France.
- BCMA:BCMA-targeted CAR‑T therapies show increasing usage among multiple myeloma patients who experience both relapsed and refractory forms of the disease. The strong clinical responses from ongoing research in France lead to growing interest in BCBA which functions as an essential antigen for developing next-generation CAR‑T therapies.
- Others: The category covers new antigens under research which include CD22 CD20 and mesothelin for both blood cancers and solid tumors. The treatments present new methods which enable CAR‑T therapies to treat patients who have not responded to standard treatment methods which creates market expansion possibilities for the French industry.
To learn more about this report, Download Free Sample Report
By Application
- Leukemia:The use of CAR-T therapies for acute lymphoblastic leukemia treatment helps patients with relapsed or refractory leukemia achieve complete remission. The clinical evidence together with the specialized infrastructure hospitals and oncology centers in France has established safe practices for treatment preparation and delivery and post-treatment monitoring which follows treatment administration.
- Lymphoma: Both B-cell and other lymphoma types are treated with CD19 and other targeted CAR‑T therapies. The adoption of therapies throughout France is growing because of increased clinical success and ongoing clinical trials and patient requirements particularly in specialized hospitals and cancer treatment centers which have experienced medical teams.
- Multiple Myeloma: The use of BCMA-targeted CAR‑T therapies has become a new treatment option for multiple myeloma which medical professionals are introducing into their practice. French oncology centers are starting to use personalized cellular immunotherapy treatments which help patients who do not respond to standard therapies.
By End User
- Hospitals:Major hospitals with oncology and hematology departments function as the primary operational centers for CAR-T treatment delivery. The hospitals function as essential medical facilities which provide safe facilities for patient treatment while enabling advanced immunotherapy programs in France through their role in research and regulatory requirements.
- Cancer Treatment Centers: The dedicated cancer centers together with research institutes treat patients with both CAR-T therapy and post-treatment monitoring through their clinical research projects. The centers provide specialized medical services which help increase the usage of CAR-T therapies while they enable patients to access the newest cellular treatment innovations.
Country Insights
France has become the top European market for CAR-T cell therapies because its strong healthcare system and active clinical research facilities and increasing rates of blood cancer diseases. The government and healthcare authorities actively promote advanced immunotherapies through favorable regulatory frameworks which provide funding support and create streamlined reimbursement pathways that encourage both local and international pharmaceutical companies to expand operations in the country.
Hospitals and specialized cancer treatment centers serve as primary treatment hubs which provide the necessary facilities and medical knowledge to conduct CAR-T therapy treatments. Clinical trials for both approved and next-generation CAR-T products are increasing their testing for leukemia and lymphoma and multiple myeloma. The combination of patient demand between supportive policies and active research and development activities establishes France as a central location for CAR-T therapy development and implementation throughout Europe.
Recent Development News
May 2025: The partnership between Astraveus and NecstGen will create a CAR-T manufacturing operation which will begin in May 2025. The French company Astraveus SAS established a strategic partnership with NecstGen which is known as The Netherlands Center for the Clinical Advancement of Stem Cell and Gene Therapies. The two organizations will work together to demonstrate a manufacturing platform which operates at high production rates while achieving lower operational costs to develop CAR-T treatments.
February 2025: Allo‑CAR‑T Research Advancement in France
The researchers from Institut Curie, Gustave Roussy, CNRS, and Inserm discovered gene combinations which decrease the immune system's ability to destroy allogeneic CAR-T cells. Their findings published in Nature Biomedical Engineering show that they advance development of "off-the-shelf" CAR-T products which show better treatment results.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 479.93 Million |
|
Market size value in 2026 |
USD 553.83 Million |
|
Revenue forecast in 2033 |
USD 1509.17 Million |
|
Growth rate |
CAGR of 15.40% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
France |
|
Key company profiled |
Novartis, Gilead Sciences, Bristol Myers Squibb, Johnson & Johnson, Fate Therapeutics, Cellectis, Autolus Therapeutics, Bluebird Bio, Legend Biotech, Allogene Therapeutics, Precision BioSciences, CRISPR Therapeutics, Takeda, Kite Pharma, TCR² Therapeutics. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Target Antigen (CD19, BCMA, Others), Application (Leukemia, Lymphoma, Multiple Myeloma), End User (Hospitals, Cancer Treatment Centers). |
Key France CAR T-cell Therapy Market Company Insights
The France CAR‑T cell therapy market is dominated by global biopharmaceutical leaders and innovative biotech firms which develop and sell advanced cellular immunotherapy products. The companies Novartis Gilead Sciences Bristol Myers Squibb Johnson Johnson and Kite Pharma develop their CAR‑T therapies through approved products which treat hematological cancers specifically leukemia and lymphoma and multiple myeloma using CD19 and BCMA targeting methods. French oncology centers use clinical expertise and hospital partnerships and localized manufacturing methods to expand their product distribution.
Fate Therapeutics Cellectis Autolus Therapeutics Bluebird Bio Legend Biotech Allogene Therapeutics Precision BioSciences CRISPR Therapeutics Takeda and TCR² Therapeutics are specialized companies which develop next-generation CAR‑T platforms and gene-edited and allogeneic therapies and novel target antigens. The clinical trials R&D partnerships with hospitals and research organizations and regulatory activities that the organization conducts in France create a framework for boosting innovation and enhancing patient access to advanced CAR‑T therapies.
Company List
- Novartis
- Gilead Sciences
- Bristol Myers Squibb
- Johnson & Johnson
- Fate Therapeutics
- Cellectis
- Autolus Therapeutics
- Bluebird Bio
- Legend Biotech
- Allogene Therapeutics
- Precision BioSciences
- CRISPR Therapeutics
- Takeda
- Kite Pharma
- TCR² Therapeutics
France CAR T-cell Therapy Market Report Segmentation
By Target Antigen
- CD19
- BCMA
- Others
By Application
- Leukemia
- Lymphoma
- Multiple Myeloma
By End User
- Hospitals
- Cancer Treatment Centers
Frequently Asked Questions
Find quick answers to common questions.
The approximate France CAR T-cell Therapy Market size for the market will be USD 1509.17 Million in 2033.
Key segments for the France CAR T-cell Therapy Market are Target Antigen (CD19, BCMA, Others), Application (Leukemia, Lymphoma, Multiple Myeloma), End User (Hospitals, Cancer Treatment Centers).
Major France CAR T-cell Therapy Market players are Novartis, Gilead Sciences, Bristol Myers Squibb, Johnson & Johnson, Fate Therapeutics, Cellectis, Autolus Therapeutics, Bluebird Bio, Legend Biotech, Allogene Therapeutics, Precision BioSciences, CRISPR Therapeutics, Takeda, Kite Pharma, TCR² Therapeutics.
The France CAR T-cell Therapy Market size is USD 479.93 Million in 2025.
The France CAR T-cell Therapy Market CAGR is 15.40%.
- Novartis
- Gilead Sciences
- Bristol Myers Squibb
- Johnson & Johnson
- Fate Therapeutics
- Cellectis
- Autolus Therapeutics
- Bluebird Bio
- Legend Biotech
- Allogene Therapeutics
- Precision BioSciences
- CRISPR Therapeutics
- Takeda
- Kite Pharma
- TCR² Therapeutics
Recently Published Reports
-
May 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033