Europe Protein Therapeutics Market Size & Forecast:
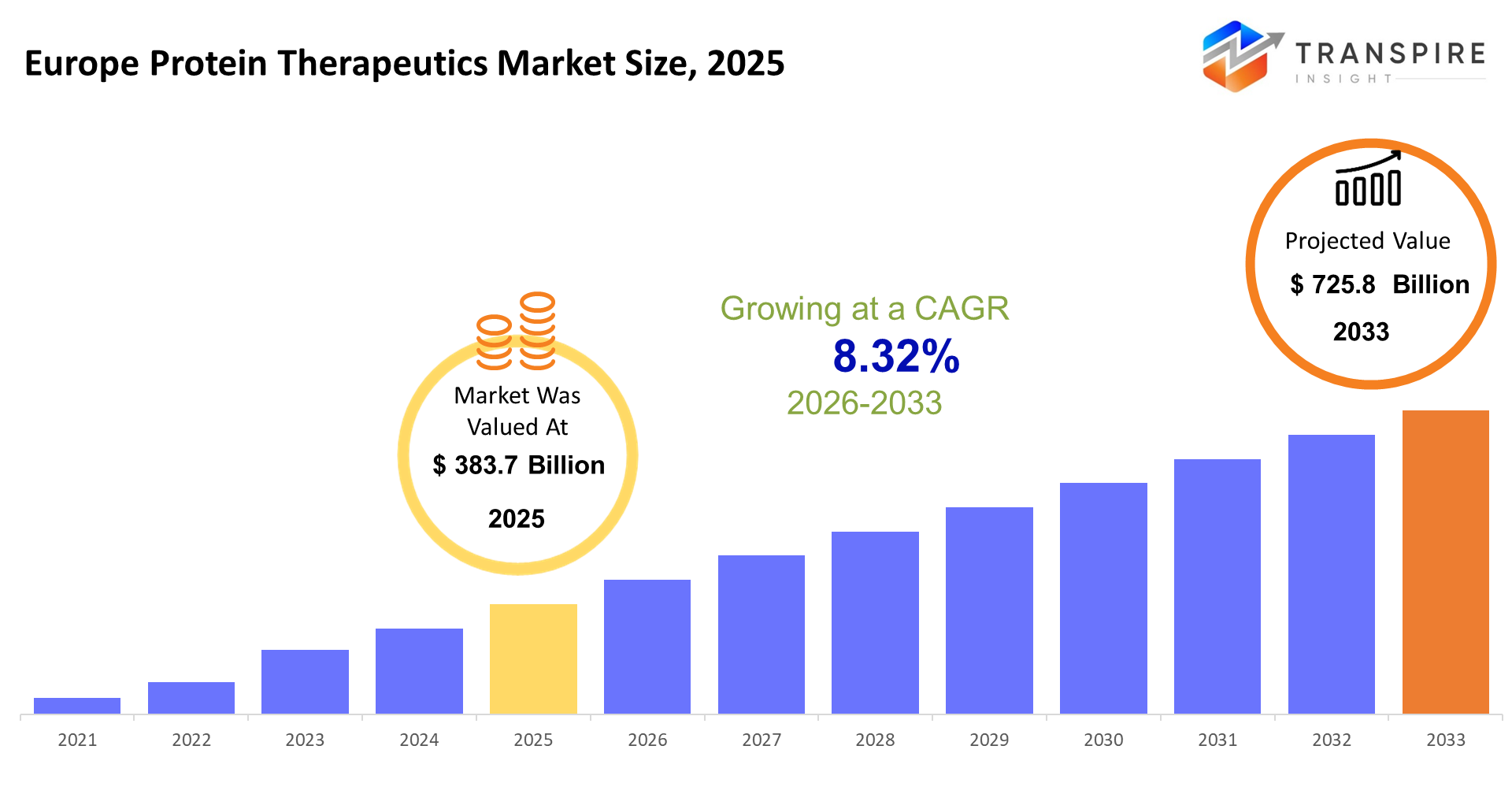
- Europe Protein Therapeutics Market Size 2025: USD 383.7 Billion
- Europe Protein Therapeutics Market Size 2033: USD 725.8 Billion
- Europe Protein Therapeutics Market CAGR: 8.32%
- Europe Protein Therapeutics Market Segments: By Type (Monoclonal Antibodies, Insulin, Fusion Proteins, Hormones, Others), By Application (Cancer, Diabetes, Autoimmune Diseases, Infectious Diseases, Others), By End-User (Hospitals, Clinics, Research Institutes, Pharma Companies, Others), By Distribution (Hospital Pharmacies, Retail Pharmacies, Others).
To learn more about this report, Download Free Sample Report
Europe Protein Therapeutics Market Summary:
The Europe Protein Therapeutics Market size is estimated at USD 383.7 Billion in 2025 and is anticipated to reach USD 725.8 Billion by 2033, growing at a CAGR of 8.32% from 2026 to 2033. European protein therapeutic solutions treat diseases through their ability to replace biological functions which exist beyond the control of small-molecule medications. The system enables doctors to provide precise treatment for cancer and autoimmune disorders and rare genetic diseases which lack effective standard treatments.
The market has developed through three to five years because healthcare organizations now choose biologic treatments instead of using all-purpose medications. The development of recombinant protein engineeringand biosimilar creationhas enabled organizations to enhance their production methods and make more affordable goods. The COVID-19 pandemic served as an essential factor which enabled European countries to establish their biologics production facilities and fast-track their regulatory processes.
The current growth process shows that businesses are adopting new technologies at a faster speed while receiving more funding support from insurance companies. The system allows more patients to receive treatment while manufacturers need to improve their production methods and product life cycle handle process.
Key Market Insights
- Germany maintains its leading position in the Europe Protein Therapeutics Market by controlling more than 22% of the market share which will exist in 2025 because of its robust biopharmaceutical infrastructure together with its research and development expenditures.
- France will experience its most rapid growth period until 2030 because of our government-supported financial programs for biotechnology and our increasing number of clinical trials.
- The market for monoclonal antibodies will reach its peak in 2025 when it achieves a 48% market share because these antibodies serve as essential treatments for both oncology and autoimmune diseases.
- The second largest market segment consists of insulin and growth factors which together generate about 21% of total market share because of their usage in treating chronic diseases.
- The segment of biosimilars will experience the highest growth rate among all segments because cost-containment strategies will drive its growth at a high single-digit rate until 2030.
- The Europe Protein Therapeutics Market sees more than 35% of its market share dedicated to oncology applications which receive support from the rising use of biologics-based cancer treatments.
- The fastest-growing application segment includes rare diseases which receive treatment through accelerated approvals and orphan drug incentives that enhance their usage.
- Hospitals control approximately 50% of the market share in 2025 because they need to administer biologics and provide specialized medical treatment.
- The end-user segment of specialty clinics experiences its most rapid growth because decentralized treatment and outpatient biologics delivery systems have become more common.
What are the Key Drivers, Restraints, and Opportunities in the Europe Protein Therapeutics Market?
Your training includes data until the month of October in the year 2023. The primary force driving market growth for protein therapeutics in Europe operates through the development of precision biologics which depends on advances in recombinant DNA technology and protein engineering. The European Medicines Agency along with other regulatory bodies has established easier paths to approval for biologics and biosimilars after the industry expanded its operations during the COVID-19 pandemic. The combination of regulatory standards with manufacturing processes has enabled faster product development which results in better market performance. Pharmaceutical companies are now developing targeted treatments at a faster pace which results in increased adoption of these treatments for patients with oncology and autoimmune diseases and produces higher revenue from each patient.
The manufacturing process of biologics presents a twofold challenge because it requires high financial resources and operational expertise. The production of protein therapeutics demands specialized equipment through bioreactors and cold-chain systems and stringent quality management procedures which make it harder to scale operations than to produce small-molecule drugs. The production process contains these restrictions because they need extensive financial resources for resolution. The health systems throughout Southern and Eastern Europe experience delayed market entry because price-sensitive customers face reimbursement restrictions which limit their ability to purchase products.
Biosimilars provide a significant future opportunity to create wider market access which will result in increased product demand. After biological patents expire leading companies are creating cost-effective biosimilar development pipelines together with their local production facilities. The development of biosimilars has improved economic factors through increased production in Poland and Spain. The European Protein Therapeutics Market will experience increased patient access together with faster product replacements and a more competitive pricing system through this industry transition.
What Has the Impact of Artificial Intelligence Been on the Europe Protein Therapeutics Market?
Artificial intelligence together with advanced digital technologies now begins to transform protein therapeutics by establishing better control methods for complex biologics production processes and enhancing research discovery operations. Companies use artificial intelligence powered control systems to manage bioreactor operations which include cell culture conditions and nutrient feed rates and contamination detection. The method enables production for commercial use because it decreases batch variations while producing steady results in both yield and quality. Manufacturing process digital twins create real-time simulations which enable operators to modify settings before errors start to impact production results thus improving their ability to meet regulatory requirements while minimizing production errors.
Machine learning models enable value chain organizations to develop forecasting abilities. Organizations adopt algorithms to predict protein folding stability and they use the algorithms for optimal molecular design and patient data-based clinical response pattern prediction. Predictive maintenance models in manufacturing environments anticipate equipment failure within bioprocessing units which leads to enhanced operational efficiency through reduced unplanned system outages. The applications produced specific measurable benefits which included shorter production cycles and higher batch success rates that resulted in lower operational costs.
The main obstacle exists because biological data stays divided into many parts which require special protection measures. Large-scale AI deployment faces obstacles because of limited standardized datasets and high system integration costs which hinder complete commercial operations for legacy systems.
Key Market Trends
- Biologics research since 2021 has shifted its focus towards monoclonal antibodies and cell-derived proteins because Roche now emphasizes oncology biologics development instead of traditional small molecule drugs.
- The period from 2022 to 2025 experienced an increase in biosimilar usage because Germany and Italy implemented stricter cost control measures which aimed to decrease their biologics expenses.
- The investments made after the 2020 pandemic allowed European countries to build new biologics manufacturing facilities which helped them produce medical proteins more quickly while decreasing their need to import essential protein therapies.
- The European Medicines Agency and other regulatory bodies have decreased the time needed for biologics approval since 2023, which helps new treatments reach the market sooner and generate revenue for their developers.
- The last three years have seen pharmaceutical companies such as Novartis form more alliances with biotech startups because they want to use new protein engineering technologies which will help them create new products more quickly.
- The introduction of orphan drug incentives after 2021 made rare disease therapeutics more attractive to research and development because it directed research efforts towards specialized protein treatments that compete with few market players.
- The implementation of AI-powered drug discovery systems in 2022 has enabled faster protein design processes which now require 30% less time, which leads to improved drug development processes and decreased clinical trial failure rates.
- The period from 2023 to 2026 will see decentralized clinical trials become more common in Europe, which will speed up patient enrollment and provide protein therapies to areas that currently lack sufficient medical services.
- The past years have seen a major rise in cold chain logistics funding, which has strengthened the dependable distribution of biologics while decreasing the amount of product waste that occurs during cross-border shipments throughout Europe.
Europe Protein Therapeutics Market Segmentation
By Type
The largest market share for monoclonal antibodies originates from their classification because they demonstrate high clinical effectiveness when used to treat both cancer and autoimmune diseases which results in continuous approval of new treatment applications. The insulin and growth hormone market maintains its strength because it fulfills the chronic disease treatment requirements of diabetes patients. The market for fusion proteins and recombinant proteins experiences moderate demand because their development process requires greater investment and technical difficulty.
The healthcare systems make their service adoption decisions based on the distinct manufacturing processes and pricing structures that exist within each sub-segment. Biosimilar monoclonal antibodies will experience growth during the upcoming period because patent expiration enables more companies to launch competing products. Manufacturers need to improve their production processes and pricing methods in response to this change while investors concentrate on biologics platforms that can be expanded.
By Application
The application of cancer treatment shows its foremost importance because targeted biologics have become widely used to enhance patient survival rates while minimizing unintended side effects. The second largest disease category consists of autoimmune diseases which receive support through extended treatment periods and stable patient demographics. Diabetes requires constant demand for insulin treatments yet its growth remains slower when compared to biologics-driven market segments.
The field of infectious diseases together with rare medical conditions has developed into specialized markets which yield significant value through their accelerated approval processes and funding for technological advancements. The upcoming medical research path will use precision medicine methods that combine protein-based therapeutics with genetic testing results. This approach requires businesses to develop unique product lines while creating new clinical testing methods.
By End-User
Hospitals hold the biggest market share because they require controlled administration with special infrastructure and monitoring capabilities for their complex biologics. Healthcare systems are experiencing rapid growth in both clinics and specialty centers because they are moving toward treating patients in outpatient settings while reducing costs. The early-stage development and clinical validation process depends on research institutes and pharmaceutical companies which create a long-term market supply chain.
Each end-user segment shows how treatment complexity affects patient accessibility. The demand patterns will change because specialty clinics and home-based biologics administration will expand, which will lead manufacturers to create better stable formulations and simpler delivery systems.
By Distribution
The need for cold chain handling and special storage requirements and controlled dispensing procedures for biologics makes hospital pharmacies the main distribution method. Retail pharmacies hold a smaller but growing share as biosimilars and less complex protein therapeutics become more accessible for outpatient use. Specialty distributors now create new distribution channels to help direct delivery systems while they enhance supply chain performance.
The methods used for distribution depend on the reimbursement systems and the available infrastructure across different regions. The upcoming developments will introduce digital tracking systems together with integrated logistics networks which will enhance inventory management systems and decrease waste materials while helping both service providers and product manufacturers.
What are the Key Use Cases Driving the Europe Protein Therapeutics Market?
The primary function of protein therapeutics in oncology treatment involves using monoclonal antibodies to target tumors while activating the immune response. The application generates maximum demand because hospitals choose therapies which enhance patient survival rates and decrease harmful effects to patients, resulting in increased funding support and ongoing usage in medical practice.
Specialty clinics and outpatient care facilities currently utilize insulin treatment to manage both autoimmune disorders and diabetes. Patients with rheumatoid arthritis require continuous treatment through regular use of their prescribed biologic medications. The need for insulin therapies remains constant because older people in Europe develop chronic diseases at high rates.
The development of new applications for medical research now includes two fields: rare genetic disorders and personalized medicine. Medical researchers now use protein engineering to develop targeted treatments for specific genetic mutations, while basic research work investigates protein-based antiviral treatments which have strong potential to bring about valuable specialized applications.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 383.7 Billion |
|
Market size value in 2026 |
USD 414.7 Billion |
|
Revenue forecast in 2033 |
USD 725.8 Billion |
|
Growth rate |
CAGR of 8.32% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain and Rest of Europe) |
|
Key company profiled |
Roche, Pfizer, Novartis, Sanofi, Amgen, AbbVie, Eli Lilly, Johnson & Johnson, Merck, AstraZeneca, Bristol Myers Squibb, GSK, Takeda, Bayer, Biogen |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Monoclonal Antibodies, Insulin, Fusion Proteins, Hormones, Others), By Application (Cancer, Diabetes, Autoimmune Diseases, Infectious Diseases, Others), By End-User (Hospitals, Clinics, Research Institutes, Pharma Companies, Others), By Distribution (Hospital Pharmacies, Retail Pharmacies, Others) |
Which Regions are Driving the Europe Protein Therapeutics Market Growth?
The market in Western Europe operates from Germany and France and the United Kingdom as its main market drivers because these nations provide complete healthcare funding and clear regulatory frameworks. The European Medicines Agency operates a centralized approval system which enables companies to launch their biologics products more quickly. Research institutes and biotech companies and major pharmaceutical companies form a strong network that enables ongoing technological progress. The advanced reimbursement systems enable patients to receive treatment while maintaining high treatment volumes which results in constant revenue flow.
Southern Europe maintains its position as a reliable market contributor while Spain and Italy work to achieve operational cost savings through their slow development of biologics. Biosimilars receive early market acceptance in this region because its healthcare systems prioritize budget management over expensive original biologics. The market experiences steady growth because chronic diseases remain persistent and public health services operate through organized systems. The region maintains dependable operational procedures through its established procurement methods and extended patient treatment schedules which include established procedures.
Poland and Hungary lead Eastern Europe as its fastest-expanding area. The region has experienced increased adoption after local biologics production facilities began operating and the European Union increased its healthcare system funding. Western European regulatory standards enable better access to modern treatment options for patients who operate in cost-sensitive environments which treat their medical needs with biosimilars. The period from 2026 to 2033 will create significant market entry possibilities for manufacturers and investors who want to increase output and establish production facilities in local markets.
Who are the Key Players in the Europe Protein Therapeutics Market and How Do They Compete?
The competitive landscape shows moderate consolidation because large pharmaceutical groups control a major market share while small biotech companies develop their own specific market solutions. Incumbents defend position through scale in manufacturing and established regulatory pathways, while new entrants compete on specialized protein engineering and faster development cycles. The competition between businesses centers on their ability to produce biologics through advanced technology resources which require intricate production methods and rigorous quality assessment procedures. Companies that balance their innovation efforts with cost management practices achieve measurable benefits in both premium product markets and biosimilar product markets.
Roche uses its advanced monoclonal antibody platforms in oncology to develop patient-specific treatments through its advanced diagnostic systems, which enhance patient care. Novartis achieves market differentiation by developing next-generation biologics and gene-based protein therapies, which its strategic acquisitions increase through various partnership agreements. Sanofi uses its extensive production capacity in diabetes and immunology to create biosimilars, which enable the company to control costs across various European distribution networks.
AstraZeneca develops its competitive advantage through its oncological and rare disease biologics research, which uses international clinical research networks to speed up its drug approval process. GlaxoSmithKline concentrates its research efforts on immunology and specialty therapeutics, which it develops through partnerships with biotechnology companies that provide access to new protein discovery technologies. Regional manufacturing facilities and partnerships in research and development enable these companies to grow their business while achieving faster product delivery and stronger protection for their future product range.
Company List
- Roche
- Pfizer
- Novartis
- Sanofi
- Amgen,
- AbbVie
- Eli Lilly
- Johnson & Johnson
- Merck
- AstraZeneca
- Bristol Myers Squibb
- GSK
- Takeda
- Bayer
- Biogen
Recent Development News
In May 2026, Archimed to take Esperion Therapeutics private in a $1.1B deal: European investment firm Archimed announced a major acquisition deal to take Esperion Therapeutics private in a transaction valued up to $1.1 billion. The deal reflects continued consolidation interest in protein-based cardiovascular therapies and biologics-linked pipelines in Europe–US biotech ecosystems.
Source: https://www.reuters.com
In April 2026, Sensorion raises €60M with Sanofi backing: French biotech Sensorion secured €60 million funding, including investment from Sanofi, strengthening its protein and gene-therapy pipeline focused on congenital deafness. The capital will accelerate clinical development of therapies targeting genetic protein deficiencies in hearing disorders.
Source: https://www.lemonde.fr
What Strategic Insights Define the Future of the Europe Protein Therapeutics Market?
The European Protein Therapeutics Market will establish its future operations through three scientific elements which include precise biologics development and decentralized production methods and integrated systems that connect diagnostic tests with treatment selection. The field advances because researchers develop new protein engineering methods while healthcare systems experience increasing demands to pay for surgical procedures that produce observable results for patients. The upcoming 5 to 7 years will witness a transition from volumetric measurements of biologics value to advanced therapeutic approaches which use biomarker-based treatment methods for their medical pricing and reimbursement systems.
The manufacturing process faces dual challenges which include restrictions on essential production materials and bioprocess resources and specific limitations related to cell culture media and special research chemicals. The production process faces two main threats which include supply chain interruptions and dependence on specific suppliers, which together limit production capacity while creating price fluctuations that reduce profit margins, despite high market demand.
The development of continuous biomanufacturing platforms provides an emerging opportunity for economic growth in Ireland, which possesses both the necessary infrastructure and supportive government policies. Companies should establish their manufacturing systems through flexible plant designs and strategic supplier agreements to maintain production capabilities and manage expenses for an extended period.
Europe Protein Therapeutics Market Report Segmentation
By Type
- Monoclonal Antibodies
- Insulin
- Fusion Proteins
- Hormones
- Others
By Application
- Cancer
- Diabetes
- Autoimmune Diseases
- Infectious Diseases
- Others
By End-User
- Hospitals
- Clinics
- Research Institutes
- Pharma Companies
- Others
By Distribution
- Hospital Pharmacies
- Retail Pharmacies
- Others
Frequently Asked Questions
Find quick answers to common questions.
The Approximate Europe Protein Therapeutics Market size for the Market will be USD 725.8 Billion in 2033.
The key Segments of the Europe Protein Therapeutics Market are By Type (Monoclonal Antibodies, Insulin, Fusion Proteins, Hormones, Others), By Application (Cancer, Diabetes, Autoimmune Diseases, Infectious Diseases, Others), By End-User (Hospitals, Clinics, Research Institutes, Pharma Companies, Others), By Distribution (Hospital Pharmacies, Retail Pharmacies, Others).
Major Players in the Europe Protein Therapeutics Market are Roche, Pfizer, Novartis, Sanofi, Amgen, AbbVie, Eli Lilly, Johnson & Johnson, Merck, AstraZeneca, Bristol Myers Squibb, GSK, Takeda, Bayer, Biogen.
The Current Market size of the Europe Protein Therapeutics Market is USD 383.7 Billion in 2025.
The Europe Protein Therapeutics Market CAGR is 8.32%.
1. Introduction
1.1. Report Description
1.2. Overview of the Europe Protein Therapeutics Market: Definition
1.3. Market Research Scope
1.4. Market Covered: Regional Scope
1.5. Years Considered for The Study
2. Research Methodology
2.1. Description
2.1.1. Market Research Process
2.1.2. Information Procurement
2.1.3. Data Analysis
2.1.4. Market Formulation & Validation
3. Executive Summary
3.1. Key Insight of the Study
3.2. Segmentation Outlook By Type
3.3. Segmentation Outlook By Application
3.4. Segmentation Outlook By End-User
3.5. Segmentation Outlook By Distribution
3.6 Segmentation Outlook by Region
4. Europe Protein Therapeutics Market – Industry Outlook
4.1. Impact of COVID-19 on the Market
4.2. Market Attractiveness Analysis
4.2.1. Market Attractiveness Analysis By Component
4.2.2. Market Attractiveness Analysis by Region
4.3. Industry Swot Analysis
4.3.1. Strength
4.3.2. Weakness
4.3.3. Opportunities
4.3.4. Threats
4.4. Porter's Five Forces Analysis
4.4.1. Threat of New Entrants
4.4.2. Bargaining Power of Suppliers
4.4.3. Bargaining Power of Buyers
4.4.4. Threat of Substitutes
4.4.5. Industry Rivalry
4.5. Pointers Covered at the Micro Level
4.5.1. Customers
4.5.2. The Supply and Demand Side
4.5.3. Shareholders and Investors
4.5.4. Media, Advertising, and Marketing
4.6. Pointers Covered at the Macro Level
4.6.1. Economic Factors
4.6.2. Technological Advancements
4.6.3. Regulatory Environment
4.6.4. Societal and Cultural Trends
4.7. Value Chain
4.7.1. Raw Material Sourcing
4.7.2. Manufacturing/Processing
4.7.3. Quality Control and Testing
4.7.4. Packaging and Distribution
4.7.5. End-Use Segment 4S
4.8. Impact of AI Across Leading Economies
5. Market Overview and Key Dynamics
5.1. Market Dynamics
5.2. Drivers
5.2.1. Driver 1
5.2.2. Driver 2
5.3. Restraints and Challenges
5.3.1. Restraints and Challenges 1
5.3.2. Restraints and Challenges 2
5.4. Opportunities
5.4.1. Opportunity 1
5.4.2. Opportunity 2
6. Global Europe Protein Therapeutics Market Insights and Forecast Analysis
6.1. Global Europe Protein Therapeutics Market Analysis and Forecast
7. Europe Protein Therapeutics Market Insights & Forecast Analysis, By Type – 2021 to 2033
7.1. Europe Protein Therapeutics Market Analysis and Forecast, By Type
7.1.1. Monoclonal Antibodies
7.1.2. Insulin
7.1.3. Fusion Proteins
7.1.4. Hormones
7.1.5. Others
8. Europe Protein Therapeutics Market Insights & Forecast Analysis, By Application – 2021 to 2033
8.1. Europe Protein Therapeutics Market Analysis and Forecast, By Application
8.1.1. Cancer
8.1.2. Diabetes
8.1.3. Autoimmune Diseases
8.1.4. Infectious Diseases
8.1.5. Others
9. Europe Protein Therapeutics Market Insights & Forecast Analysis, By End-User – 2021 to 2033
9.1 Europe Protein Therapeutics Market Analysis and Forecast, By End-User
9.1.1. Hospitals
9.1.2. Clinics
9.1.3. Research Institutes
9.1.4. Pharma Companies
9.1.5. Others
10. Europe Protein Therapeutics Market Insights & Forecast Analysis, By Distribution – 2021 to 2033
10.1 Europe Protein Therapeutics Market Analysis and Forecast, By Distribution
10.1.1. Hospital Pharmacies
10.1.2. Retail Pharmacies
10.1.3. Others
11 Europe Protein Therapeutics Market Insights & Forecast Analysis, By Region – 2021 to 2033
11.1 Europe Protein Therapeutics Market, By Region
11.2 Europe Protein Therapeutics Market, By Type
11.2.1 Europe Protein Therapeutics Market, By Type, Revenue (USD Billion), (2021 -2033), CAGR (%) (2026-2033)
11.3 Europe Protein Therapeutics Market, By Application
11.3.1 Europe Protein Therapeutics Market, By Application, Revenue (USD Billion), (2021 -2033), CAGR (%) (2026-2033)
11.4 Europe Protein Therapeutics Market, By End-User
11.4.1 Europe Protein Therapeutics Market, By End-User, Revenue (USD Billion), (2021 -2033), CAGR (%) (2026-2033)
11.5 Europe Protein Therapeutics Market, By Distribution
11.5.1 Europe Protein Therapeutics Market, By Distribution , Revenue (USD Billion), (2021 -2033), CAGR (%) (2026-2033)
11.6 Europe Protein Therapeutics Market Insights & Forecast Analysis, BY Segmentation and Country – 2021 - 2033
11.7 Europe Protein Therapeutics Market, By Country
11.7.1 Germany
11.7.2 United Kingdom
11.7.3 France
11.7.4 Italy
11.13.5 Spain
11.13.6 Rest of Europe
12 Europe Protein Therapeutics Market: Competitive Landscape
12.1 Competitive Rivalry and Division
12.2 Company Market Share Analysis
12.3 Europe Protein Therapeutics Market: Top Winning Strategies
12.4 Europe Protein Therapeutics Market: Competitive Heatmap Analysis
13 Europe Protein Therapeutics Market: Company Profiles
13.1 Roche
13.1.1 Overview of Business
13.1.2 Economic Performance of the Company
13.1.3 Key Executives
13.1.4 Portfolio of Products
13.1.5 Company Strategy Mapping
13.2 Pfizer
13.3 Novartis
13.4 Sanofi
13.5 Amgen,
13.6 AbbVie
13.7 Eli Lilly
13.8 Johnson & Johnson
13.9 Merck
13.10 AstraZeneca
13.11 Bristol Myers Squibb
13.12 GSK
13.13 Takeda
13.14 Bayer
13.15 Biogen
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


