France Pre-clinical Scientific Research Medical Device Market Size & Forecast:
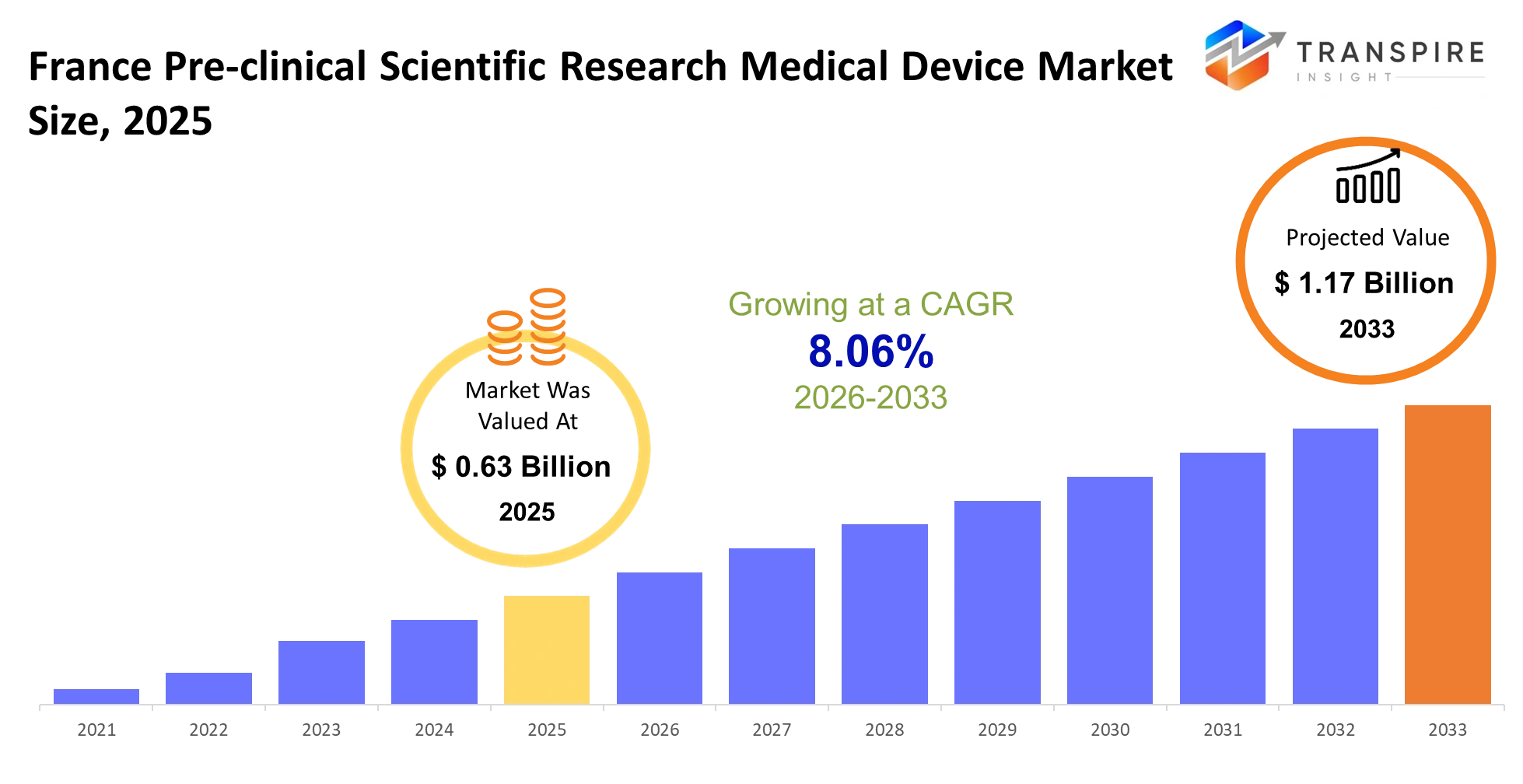
- France Pre-clinical Scientific Research Medical Device Market Size 2025: USD 0.63 Billion
- France Pre-clinical Scientific Research Medical Device Market Size 2033: USD 1.17 Billion
- France Pre-clinical Scientific Research Medical Device Market CAGR: 8.06%
- France Pre-clinical Scientific Research Medical Device Market Segments: By Type (Imaging Devices, Analytical Instruments, Monitoring Devices, Others); By Application (Drug Discovery, Toxicology Studies, Disease Research, Preclinical Trials, Others); By End-User (Research Institutes, Pharma Companies, Biotech Firms, Academic Institutions, Others); By Technology (AI-enabled Devices, Advanced Imaging, Others).
To learn more about this report, Download Free Sample Report
France Pre-clinical Scientific Research Medical Device Market Summary
The France Pre-clinical Scientific Research Medical Device Market was valued at USD 0.63 Billion in 2025. It is forecast to reach USD 1.17 Billion by 2033. That is a CAGR of 8.06% over the period.
The France Pre-clinical Scientific Research Medical Device Market enables researchers to conduct safety and efficacy tests because it provides them with facilities to assess biological responses before they start their human trials. The system enables laboratories to conduct experimental research through its imaging systems and analytical tools and monitoring devices which help decrease clinical risks while boosting trial success rates. Pharmaceutical companies require this function because their development processes have become more complicated and their budget requirements have increased.
The market experienced a structural transformation during the last three to five years which moved toward AI-driven high-resolution imaging solutions that replaced complete manual testing processes. The COVID-19 pandemic created major disruptions which led to the discovery of operational bottlenecks that existed in preclinical study timelines which in turn increased funding for both automated systems and digital laboratory environments.
The organizations now focus on generating data more quickly while ensuring that their results can be reproduced which leads to more organizations using advanced technology systems. The process leads to research work becoming more efficient while development times decrease which results in organizations making bigger investments and the market continuing to grow.
Key Market Insights
- Northern France dominates with over 35% share in 2025, supported by dense research clusters and pharmaceutical R&D hubs.
- The region of Southern France experiences its fastest growth period between now and 2033 because biotech startups and government-sponsored innovation initiatives are expanding.
- The market shows 40% share for imaging devices in 2025 because researchers need high-resolution tools for preclinical visualization.
- The second-largest market share belongs to analytical instruments because scientists need to conduct more biomarker and molecular tests.
- The segment of AI-enabled monitoring devices experiences the fastest growth between 2025 and 2033 because it tracks real-time data.
- The drug discovery market holds 45% share in 2025 because pharmaceutical companies develop new products and research biologics.
- The field of disease research shows the fastest expansion because more scientists study uncommon and long-lasting medical conditions.
- Pharma companies control approximately 50% market share because they invest heavily in research and development and possess superior testing facilities.
- Biotech companies experience their fastest growth as an end-user segment because they receive venture capital and create innovative products.
- Major companies in the market include Thermo Fisher Scientific, PerkinElmer Danaher Corporation Bruker Corporation and Agilent Technologies.
- Companies invest in AI-driven product development to improve data precision and enhance their research capabilities.
- The research institutes form strategic partnerships to help their products achieve validation while entering new markets. The European R&D hubs enable our company to extend its operations which brings us closer to our customers.
- The use of advanced imaging and automation technologies enables researchers to complete their work more quickly which helps organizations achieve better research results.
What are the Key Drivers, Restraints, and Opportunities in the France Pre-clinical Scientific Research Medical Device Market?
The primary driver of this market is the rapid expansion of complex drug pipelines which includes both biologics and personalized medicine. The shift occurred because genomics research made progress and regulators began supporting precision medicine development. Pharmaceutical companies need advanced preclinical tools to assess their targets because these tools will help them decrease their clinical failure rates which creates a need for advanced imaging and analytical systems.
The high capital expense together with the complex process required to establish advanced preclinical systems represents the most important limitation. The devices need special facilities together with trained staff members who can work for extended periods to complete their validation processes. The research laboratories of smaller organizations together with the new biotech startups face a structural obstacle which prevents them from using this technology while making it impossible to generate revenue in the mid-market segment.
The key opportunity exists through using AI-based simulation tools together with virtual testing systems. Researchers can now use digital twin technology together with in-silico modeling to create virtual simulations of biological reactions which they can test before conducting actual experiments. France's increasing funding for biotech innovation centers establishes a solid base which enables these technologies to expand successfully while creating efficient research processes.
What Has the Impact of Artificial Intelligence Been on the France Pre-clinical Scientific Research Medical Device Market?
Artificial intelligence develops new pre-clinical scientific research devices through its data collection and analysis methods which scientists use to conduct their initial testing. Laboratories use artificial intelligence to establish automated systems that handle repetitive tasks like image analysis and compound screening and animal study behavioral observation. The system decreases the need for human control while it maintains uniformity throughout all research activities.
Researchers use machine learning platforms to perform predictive studies which enable them to determine drug toxicity and drug effectiveness and biological interactions before they start clinical testing. The systems analyze extensive imaging and analytical data to detect patterns which remain undetectable through human examination. The research teams complete their validation processes at a faster rate which results in decreased expenses that occur during the final project stages.
AI technology enhances laboratory operations by boosting productivity and decreasing equipment downtime through its operational capabilities. The automated systems create a framework for ongoing testing which operates while the predictive maintenance system guarantees equipment functions at its highest capacity.
The system of artificial intelligence implementation suffers from two main obstacles which include expensive system integration costs and the requirement for extensive top-quality training data. Research institutions encounter difficulties when they try to standardize their data formats which creates obstacles that prevent them from achieving efficiency improvements through wider data-sharing practices.
Key Market Trends
- After 2021, AI-powered imaging systems became popular because they enabled preclinical research teams to finish their work 40% faster through better analysis methods.
- From 2022 until 2025, pharmaceutical companies increased their spending on automated laboratory systems to solve trial delays and enhance study reproducibility.
- Research in biologics required deeper cellular and molecular imaging which created a greater need for high-resolution imaging technologies.
- After 2023, biotech startups experienced rapid growth which created a need for analytical tools that could scale with their operations and stay within budget.
- The number of research partnerships between academic institutions and business organizations increased, which helped to speed up the process of developing and testing new devices.
- The introduction of advanced simulation and in-silico technologies became necessary for organizations because regulations required them to decrease their use of animal testing.
- The emergence of cloud-based data platforms established a major trend which enabled researchers to work together in real time while managing their research data from a single location.
- The equipment manufacturing industry adopted modular device design because it enabled manufacturers to create products that could be upgraded and combined with artificial intelligence technology.
- The pandemic caused supply chain disruptions which prompted manufacturing companies in France to adopt localized production methods by 2024.
- Companies started using partnerships and acquisitions as their main competitive approach because these methods helped them build stronger technology systems and expand their market reach.
France Pre-clinical Scientific Research Medical Device Market Segmentation
By Type
The type-based segmentation system functions as a main driver which determines how people choose between various devices because imaging systems and analytical systems together make up the highest selection percentage since they produce actual data samples which undergo validation processes. The two categories maintain their leading market position because their systems require exact scientific visualization together with advanced molecular assessment tools. The need for scientists who require their experiments to achieve reproducible results at high definition keeps driving market demand.
The segment experiences growth because laboratories adopt automated systems which enable high-throughput operations through their integrated systems that enhance precision by minimizing operator requirements. The industry will advance toward developing small devices which offer multiple functions because these devices will provide imaging together with analytical and monitoring features.
By Application
The research field drives equipment requirements through its application-based segmentation, which shows that drug discovery activities have the highest equipment need because pharmaceutical and biologic companies keep developing new products. The toxicology and disease research fields receive support from both regulatory mandates and the growing need to study chronic and rare diseases. Preclinical trials function as ongoing validation steps because they need equipment to perform at dependable levels throughout the testing process.
The growth of this market segment receives support from targeted therapies and precision medicine, which require scientists to investigate biological systems at more fundamental levels. The upcoming forecast period will create new application fields which will prompt device manufacturers to develop research tools designed for specific scientific workflows while investors concentrate on business sectors with high potential for expansion.
To learn more about this report, Download Free Sample Report
By End-User
The pharmaceutical sector leads end-user segmentation because its companies spend heavily on research and development while their established testing facilities support research operations. Research institutes and academic institutions maintain a stable presence, contributing to foundational studies and early-stage innovation. Biotech firms are emerging rapidly, supported by venture funding and niche research pipelines.
The industry-academic partnerships of this segment drive growth because they speed up both technology adoption and knowledge sharing processes. Future trends indicate rising demand from smaller, innovation-driven organizations, prompting suppliers to offer flexible pricing models and modular systems to capture a broader customer base.
By Technology
The segmentation through technology demonstrates how artificial intelligence and advanced imaging systems which use AI bring new benefits by improving data accuracy and shortening processing times. The existing system of traditional technologies remains active in various cost-sensitive areas because digital solutions are still developing.
Research workflows require faster data interpretation and predictive capabilities which drive adoption in this segment. The upcoming years will bring changes to device functions because artificial intelligence will combine with imaging systems and analytical platforms to create new requirements which companies must fulfill through software development and digital base expansion.
What are the Key Use Cases Driving the France Pre-clinical Scientific Research Medical Device Market?
Drug discovery remains the core use case driving adoption, as pharmaceutical companies rely on pre-clinical devices to validate drug targets and assess efficacy before human trials. The combination of high-resolution imaging and analytical systems enables scientists to observe cellular responses with extreme accuracy which leads to reduced trial failures during late stages and improved research efficiency.
Biotech firms and research institutes are using toxicology studies and disease research as their growing operational base. The European Union requires complete safety assessments through regulatory frameworks which increases demand for tracking and testing equipment. The increasing focus on testing environments for specialized preclinical assessments results from the rising need to treat uncommon and long-lasting medical conditions.
The field of in-silico modeling and AI-assisted simulation now represents an emerging area which seeks to decrease animal testing requirements through its development. The combination of advanced digital platforms with personalized medicine research enables scientists to create new testing methods which match genetic profiles with therapeutic solutions.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 0.63 Billion |
|
Market size value in 2026 |
USD 0.68 Billion |
|
Revenue forecast in 2033 |
USD 1.17 Billion |
|
Growth rate |
CAGR of 8.06% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
France |
|
Key company profiled |
Thermo Fisher, GE Healthcare, Siemens Healthineers, Agilent, Danaher, Bio-Rad, PerkinElmer, Bruker, Waters Corporation, Shimadzu, Zeiss, Olympus, Leica Microsystems, Sartorius, Abbott. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Imaging Devices, Analytical Instruments, Monitoring Devices, Others); By Application (Drug Discovery, Toxicology Studies, Disease Research, Preclinical Trials, Others); By End-User (Research Institutes, Pharma Companies, Biotech Firms, Academic Institutions, Others); By Technology (AI-enabled Devices, Advanced Imaging, Others). |
Which Regions are Driving the France Pre-clinical Scientific Research Medical Device Market Growth?
The market activity in Northern France reaches its highest point because the region contains numerous pharmaceutical headquarters and contract research organizations and government-backed innovation centers. Biomedical R&D receives dedicated tax benefits which dental research facilities need to develop cutting-edge preclinical equipment. The existing laboratory facilities together with their trained researchers and research staff members prove to be important assets that help the organization maintain its dominant market position. The ecosystem enables efficient movement from initial discovery phases toward final validation processes which leads to continuous demand for systems that operate at peak performance.
Île-de-France serves as the second key region which relies more on research conducted by academic and institutional organizations than on industrial research capacity. The region depends on public funding programs and long-term research initiatives which provide financial support for its growth instead of relying on private sector expansion. The system establishes permanent stability which guarantees that scientific equipment will be obtained during all economic conditions. The strong ties between universities and hospitals and biotech companies enable the ongoing use of preclinical research methods.
Southern France exhibits its highest growth rate because biotech startups have increased their activities since 2022 while regional funding programs have emerged. The growth of innovation centers in Marseille and Toulouse has created a need for research equipment which offers flexible usage and affordable pricing. Startups in research fields can now enter the market more easily because they receive better funding options and improved infrastructure support. Device manufacturers who want to develop scalable products between 2026 and 2033 will find excellent business opportunities.
Who are the Key Players in the France Pre-clinical Scientific Research Medical Device Market and How Do They Compete?
The market maintains moderate consolidation because global life sciences and instrumentation companies maintain control over most market share through their technological expertise and extensive distribution systems. The main competition between businesses depends on their ability to develop better imaging quality and more accurate analytical methods and digital platform integration. Established companies maintain their market leadership through ongoing product development while smaller companies create specialized AI solutions for specific markets. The competitive landscape emerges through two main factors which include business size and technological advancements.
Thermo Fisher establishes its competitive edge by developing complete laboratory systems that integrate testing equipment with data management solutions, which help large pharmaceutical businesses achieve better operational efficiency. Danaher uses its portfolio-focused approach to create specialized research tools which its subsidiaries distribute to multiple research phases, thus enabling effective product sales across different markets. Agilent specializes in molecular analysis with high precision to provide laboratories with reproducible and precise measurement results.
Bruker uses its advanced spectroscopy systems and high-resolution imaging technology to create a unique identification system that helps scientists conduct in-depth biological research at the microscopic level. Siemens Healthineers extends its capabilities beyond imaging through AI integration and digital health platform development to create research environments that operate through interconnected systems and utilize data. The companies enhance their market power through research institute partnerships and strategic acquisitions and their entry into new innovation centers.
Company List
- Thermo Fisher
- GE Healthcare
- Siemens Healthineers
- Agilent
- Danaher
- Bio-Rad
- PerkinElmer
- Bruker
- Waters Corporation
- Shimadzu
- Zeiss
- Olympus
- Leica Microsystems
- Sartorius
- Abbott
Recent Development News
In March 2026, Thermo Fisher Scientific announced the launch of an advanced AI-integrated imaging platform. The system improves preclinical data accuracy and accelerates drug validation timelines.
Source: https://www.thermofisher.com
In January 2026, Siemens Healthineers entered a partnership with a European biotech consortium. The collaboration focuses on integrating AI tools into preclinical imaging workflows to enhance research efficiency.
Source: https://www.siemens-healthineers.com
What Strategic Insights Define the Future of the France Pre-clinical Scientific Research Medical Device Market?
The market trend advances toward complete research ecosystems which utilize data for their operations through combined hardware, software, and analytical systems. The need to decrease drug development hazards together with shortening drug approval processes drives investment into technologies which will enable automated systems and forecasting capabilities. Laboratories lack standardized data formats which create hidden risks because this issue prevents laboratories from sharing data and using advanced analytical tools effectively.
The combination of physical experiments with in-silico simulations through hybrid research models creates new research opportunities because regulatory agencies investigate alternatives to animal testing. The new approach enables organizations to achieve substantial cost reductions while they speed up their initial product testing process. Organizations should focus on developing platforms which enable system compatibility to meet future data requirements because these platforms will help them expand their business and maintain their competitive advantage in technology markets.
France Pre-clinical Scientific Research Medical Device Market Report Segmentation
By Type
- Imaging Devices
- Analytical Instruments
- Monitoring Devices
- Others
By Application
- Drug Discovery
- Toxicology Studies
- Disease Research
- Preclinical Trials
- Others
By End-User
- Research Institutes
- Pharma Companies
- Biotech Firms
- Academic Institutions
- Others
By Technology
- AI-enabled Devices
- Advanced Imaging
- Others
Frequently Asked Questions
Find quick answers to common questions.
The confirmed 2033 market size figure in USD 1.17 Billion.
Key segments for the France Pre-clinical Scientific Research Medical Device Market are By Type (Imaging Devices, Analytical Instruments, Monitoring Devices, Others); By Application (Drug Discovery, Toxicology Studies, Disease Research, Preclinical Trials, Others); By End-User (Research Institutes, Pharma Companies, Biotech Firms, Academic Institutions, Others); By Technology (AI-enabled Devices, Advanced Imaging, Others).
Major France Pre-clinical Scientific Research Medical Device Market players are Thermo Fisher, GE Healthcare, Siemens Healthineers, Agilent, Danaher, Bio-Rad, PerkinElmer, Bruker, Waters Corporation, Shimadzu, Zeiss, Olympus, Leica Microsystems, Sartorius, Abbott .
The France Pre-clinical Scientific Research Medical Device Market size is USD 0.63 Billion in 2025.
The France Pre-clinical Scientific Research Medical Device Market CAGR is 8.06% from 2026 to 2033.
- Thermo Fisher
- GE Healthcare
- Siemens Healthineers
- Agilent
- Danaher
- Bio-Rad
- PerkinElmer
- Bruker
- Waters Corporation
- Shimadzu
- Zeiss
- Olympus
- Leica Microsystems
- Sartorius
- Abbott
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)









