North America Genetic Toxicology Testing Market Size & Forecast:
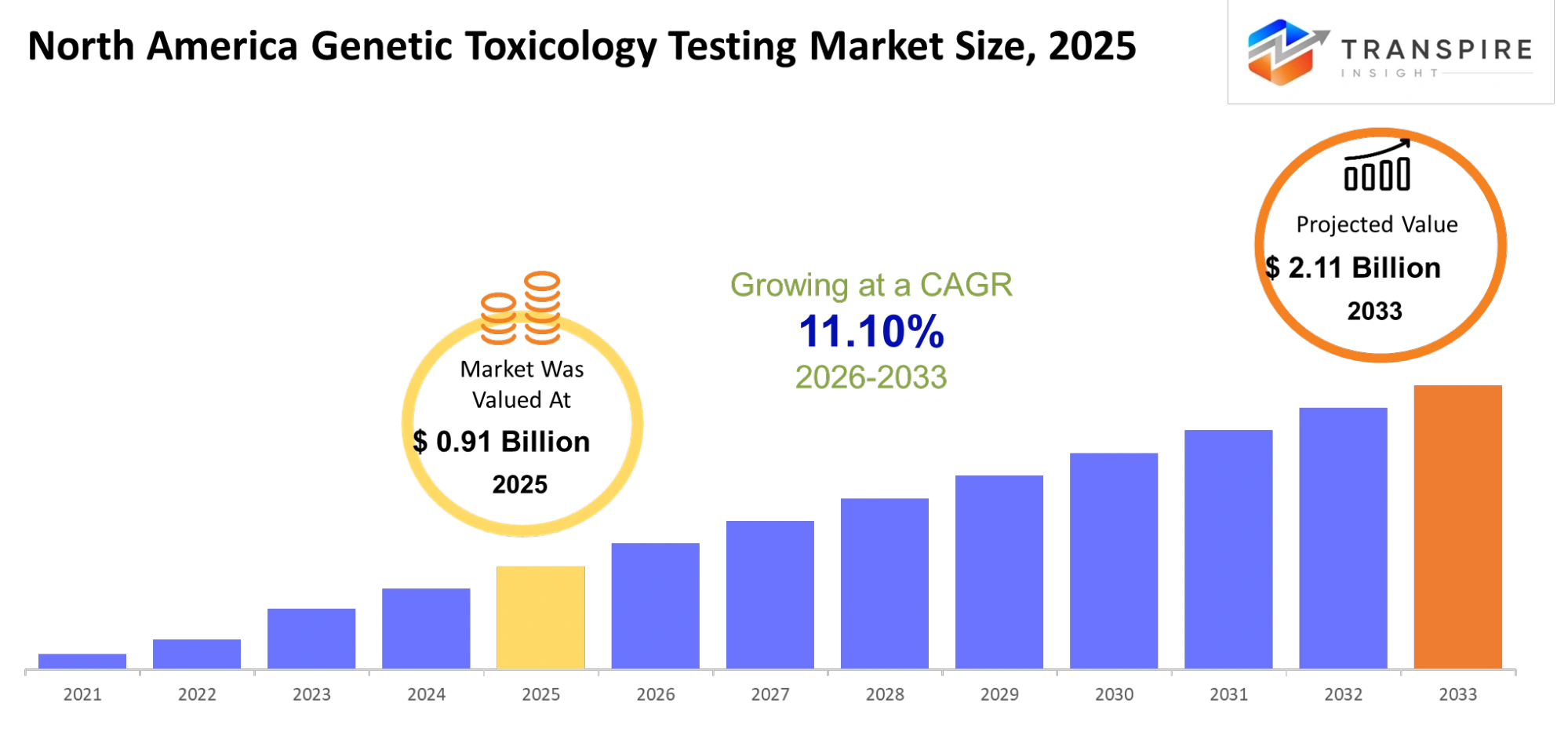
- North America Genetic Toxicology Testing Market Size 2025: USD 0.91 Billion
- North America Genetic Toxicology Testing Market Size 2033: USD 2.11 Billion
- North America Genetic Toxicology Testing Market CAGR: 11.10%
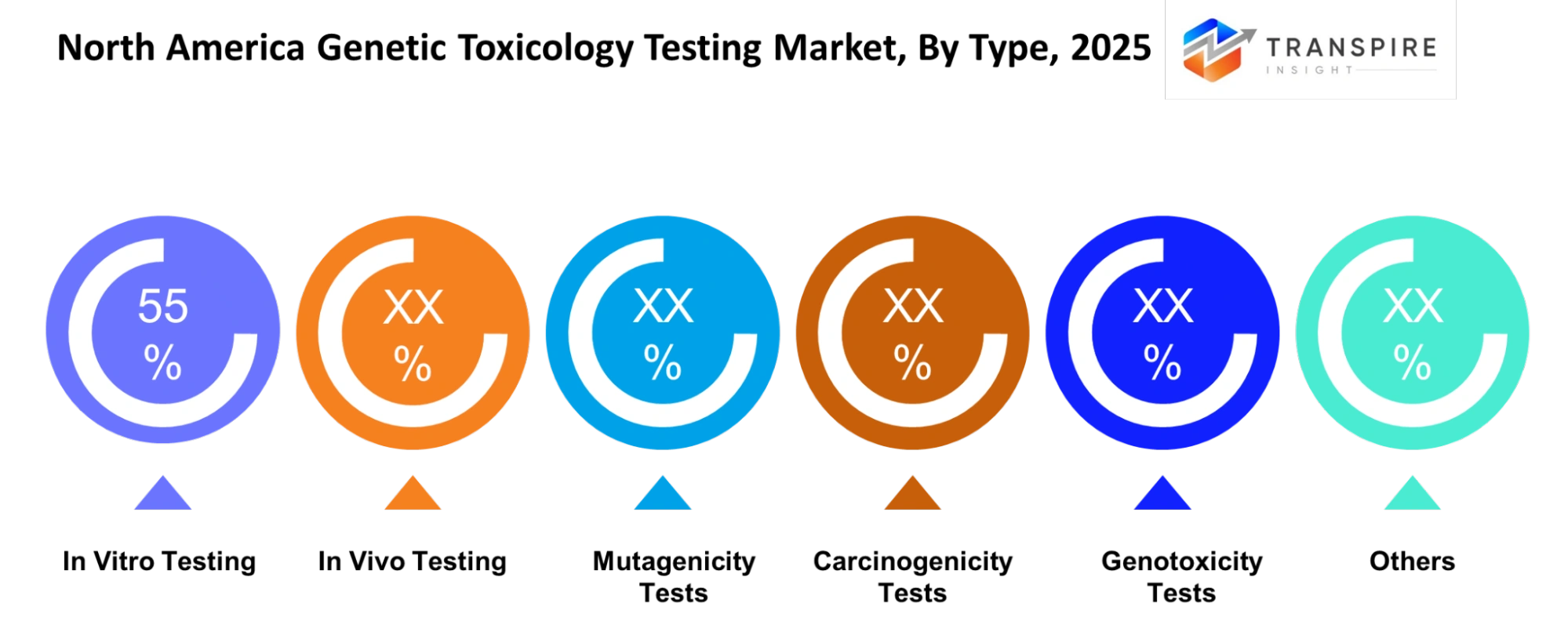
- North America Genetic Toxicology Testing Market Segments: By Type (In Vitro Testing, In Vivo Testing, Mutagenicity Tests, Carcinogenicity Tests, Genotoxicity Tests, Others); By Application (Drug Development, Chemicals Testing, Food Safety, Cosmetics, Environmental Testing, Others); By End-User (Pharma, Biotech, CROs, Research Institutes, Govt, Others); By Technology (PCR, Sequencing, Microarray, AI-based Analysis, Others)
To learn more about this report, Download Free Sample Report
North America Genetic Toxicology Testing Market Summary:
The North America Genetic Toxicology Testing Market size is estimated at USD 0.91 Billion in 2025 and is anticipated to reach USD 2.11 Billion by 2033, growing at a CAGR of 11.10% from 2026 to 2033.
The North American genetic toxicology testing market operates under two main forces which include changing regulations and growing accuracy in assessing drug safety for pharmaceuticals and chemicals and biological products. Laboratory methods and data interpretation techniques have changed because consumers now understand drug safety better and because regulatory authorities have established more difficult approval processes.
The development of sequencing technologies and in vitro testing methods has improved testing precision for all testing methods.Upcoming testing systems will use two main technologies which include AI-based analytics and high-throughput screening to meet new regulatory requirements. Laboratories in the region will expand their research capabilities and operational systems through their need to produce toxicology results.
What Has the Impact of Artificial Intelligence Been on the North America Genetic Toxicology Testing Market?
Artificial intelligence systems make quick advancements which change how North American genetic toxicity tests are conducted because they provide research and diagnostic teams with better decision-making tools. The North American genetic toxicology testing market uses artificial intelligence technology to enhance data analysis and market research because it enables better processing of extensive genomic information. The North American genetic toxicology testing market uses artificial intelligence to improve its predictive analytics capabilities while it uses market trend analysis to help stakeholders understand upcoming changes in demand. Organizations achieve better performance through machine learning systems and intelligent automation which decrease testing errors and deliver dependable results with quicker testing processes.
The North American genetic toxicology testing market improves its operations through artificial intelligence automation which makes laboratory work easier and better supply chain management. Companies achieve cost savings and faster delivery times, while improving their ability to manage resources through predictive analytics systems. Testing organizations should use machine learning systems to develop new testing methods, which will enable them to create customized toxicology assessment procedures. The North American genetic toxicology testing market gains a competitive edge through artificial intelligence which defines market movements and drives digital evolution throughout the industry.
Key Market Trends & Insights:
- The United States dominates the north america genetic toxicology testing market with an estimated 65–70% share due to advanced biotech infrastructure and high R&D spending.
- Canada is the fastest-growing region in the north america genetic toxicology testing market, driven by expanding clinical research and regulatory modernization during 2024–2030.
- The north america genetic toxicology testing market shows emerging demand from Mexico because of increasing pharmaceutical manufacturing and outsourcing trends.
- The north america genetic toxicology testing market is dominated by the services segment which holds more than 40% market share because of increasing outsourcing to CROs and specialized labs.
- The north america genetic toxicology testing market's second-largest segment consists of assay kits and consumables which receive support from ongoing demand for testing.
- The north america genetic toxicology testing market sees its main testing activities dedicated to pharmaceutical drug safety testing which fulfills Food and Drug Administration requirements.
- The north america genetic toxicology testing market experiences its fastest growth through oncology and biotechnology research which benefits from the development of precision medicine.
- The pharmaceutical and biotechnology sector controls most of the north america genetic toxicology testing market because companies require testing for their preclinical development processes.
- The north america genetic toxicology testing market experiences its highest growth through contract research organizations because these organizations increasingly use outsourcing methods.
- The north america genetic toxicology testing market sees Thermo Fisher Scientific, Charles River Laboratories, Labcorp, Eurofins Scientific, SGS, and WuXi AppTec operate as its main industry players.
North America Genetic Toxicology Testing Market Segmentation
By Type:
The evaluation of genetic damage in pharmaceutical and chemical development activities uses three testing methods: in vitro testing, in vivo testing, mutagenicity tests, carcinogenicity tests, genotoxicity tests, and other methods. The three testing methods which testing approaches use help identify hazards that companies need to assess before they can obtain regulatory approval to operate their businesses in various sectors.
Public demand for safety assessments before product launch drives companies to implement new toxicology testing methods. The combination of industry standards and regulatory requirements drives organizations to evaluate genetic risks which results in decreased chemical and drug development project failures and increased pipeline efficiency.
To learn more about this report, Download Free Sample Report
By Application :
The research process uses drug development applications to assess safety through their testing which occurs during both preclinical and clinical testing phases. The industrial sector uses chemical testing to meet its regulatory requirements while food safety and cosmetics testing procedures protect consumer health. Environmental testing assesses the danger of pollutants and dangerous materials to various ecosystems throughout the environment.
The rising need for regulatory compliance drives organizations to conduct toxicology assessments for different uses. Increased safety requirements for consumer goods together with environmental monitoring initiatives, lead to wider use of genetic testing for risk assessment and quality control purposes.
By End-User :
The pharmaceutical industry together with biotechnology companies serve as primary customers because they maintain extensive drug development programs. The testing requirements of contract research organizations need outsourced support whereas research institutes dedicate their efforts to developing new scientific methods. Government organizations create safety rules and perform monitoring tasks which cover health protection and environmental preservation efforts.
The growth of outsourcing services enables contract research organizations to extend their toxicology testing operations. The pharmaceutical industry together with biotechnology companies adopt new technologies because they receive strong research funding and regulatory requirements while government safety programs develop testing standards and evaluation methods based on compliance.
By Technology :
The combination of PCR technology with sequencing methods and microarray systems enables researchers to conduct extensive genetic research. The AI-based analysis system enables researchers to understand data better while predicting toxicology results from complicated datasets.
The development of new computational tools enables scientists to conduct more accurate toxicology assessments. The combination of automated systems with molecular testing methods increases operational effectiveness while decreasing human errors and enabling quicker safety evaluation decisions in pharmaceutical and chemical testing processes.
What are the Main Challenges for the North America Genetic Toxicology Testing Market Growth?
The north america genetic toxicology testing market encounters multiple technical and operational obstacles which prevent it from achieving optimal scalability and operational performance. Testing accuracy and turnaround time continue to experience negative impacts from complex assay protocols and genomic data interpretation variations and high-throughput screening technology limitations. The north america genetic toxicology testing market experiences workflow optimization delays because of integration problems between outdated laboratory systems and modern digital systems which result in data handling and analytical work inefficiencies.
The north america genetic toxicology testing market encounters major obstacles from strict regulatory requirements which include GMP compliance and changing FDA regulations throughout its manufacturing and commercialization activities. Market participants face increased validation requirements and higher operational expenses because of these regulations. The north america genetic toxicology testing market faces substantial growth obstacles because high production expenses for advanced reagents and assay kits and AI-enabled platforms prevent companies from achieving large-scale market entry. The need for specialized raw materials together with supply chain interruptions creates additional obstacles that prevent successful commercialization.
The north america genetic toxicology testing market suffers from adoption and infrastructure problems which stem from the existing bioinformatics and toxicogenomics workforce shortage. Advanced automation systems and funding support remain inaccessible to smaller laboratories and emerging research facilities which leads to a slower pace of technology adoption. The north america genetic toxicology testing market experiences restricted growth because of adoption challenges which prevent equal development across different regions and create a technological divide between advanced and emerging institutions
Country Insights
The increasing demand for safety assessments in pharmaceuticals and biotechnology and chemical industries will drive companies to adopt new safety assessment methods. The FDA and EPA will establish more stringent testing protocols through their regulatory frameworks. The expansion of drug discovery pipelines will increase the application of genotoxicity screening during development and preclinical evaluation because further process validation requires these tests.
The development of new in vitro testing methods together with computational toxicology platforms will improve the efficiency of assessing genetic toxicity. The high-throughput screening systems will enable faster and more precise testing processes. The rising demand for animal-free testing methods will lead to the use of contemporary toxicology solutions in pharmaceutical and chemical safety assessments and their compliance with regulatory requirements.
The market capacity will increase through the growth of contract research organizations and specialized toxicology laboratories. Pharmaceutical companies now test their products through third-party organizations which creates a need for these testing services. The testing reliability will improve while testing turnaround time will decrease through the industry's adoption of advanced laboratory infrastructure and automation systems which leads to growth in toxicology assessment services.
Recent Development News
In March 2026, Scendea appointed Hilary Fong as Principal Consultant to strengthen global regulatory and toxicology advisory services. The leadership addition supports expanded consulting capabilities in genetic toxicology testing and compliance across North America and international markets.Source https://www.scendea.com/
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 0.91 Billion |
|
Market size value in 2026 |
USD 1.01 Billion |
|
Revenue forecast in 2033 |
USD 2.11 Billion |
|
Growth rate |
CAGR of 11.10% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
North America (Canada, The United States, and Mexico) |
|
Key company profiled |
Charles River Laboratories, Eurofins Scientific, SGS, Intertek, Thermo Fisher, Labcorp, Covance, IQVIA, WuXi AppTec, Envigo, Toxikon, Creative Bioarray, GenScript, BioReliance, Agilent |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (In Vitro Testing, In Vivo Testing, Mutagenicity Tests, Carcinogenicity Tests, Genotoxicity Tests, Others); By Application (Drug Development, Chemicals Testing, Food Safety, Cosmetics, Environmental Testing, Others); By End-User (Pharma, Biotech, CROs, Research Institutes, Govt, Others); By Technology (PCR, Sequencing, Microarray, AI-based Analysis, Others) |
How Can New Companies Establish a Strong Foothold in the North America Genetic Toxicology Testing Market?
New companies entering the north america genetic toxicology testing market can establish a strong foothold by focusing on niche specialization and technology-driven differentiation. The north america genetic toxicology testing market experiences increasing demand for AI-powered toxicity prediction systems which work together with high-throughput screening methods and cost-effective genomic testing solutions. Startups that target underserved segments such as rare disease toxicology or predictive in-silico testing can gain faster adoption by addressing specific industry pain points in drug safety and regulatory compliance.
The north american genetic toxicology testing market needs strategic partnerships to enhance their market presence. The new market entrants establish partnerships with pharmaceutical companies and contract research organizations and academic institutions to gain access to advanced datasets and validation networks. The North American market expansion is demonstrated through Inotiv and Toxys which are two emerging firms that use machine learning-based toxicology platforms to enhance their research capabilities while reducing operational costs according to market trends in the north american genetic toxicology testing industry.
The north america genetic toxicology testing market derives its main competitive edge from technological innovation which includes artificial intelligence and automation and cloud-based bioinformatics platforms. New companies can create distinct market positions through their development of integrated solutions which combine predictive analytics with regulatory reporting tools.Startups establish their market position through healthcare and pharmaceutical R&D efficiency because it helps them gain market credibility while enabling faster business expansion in the n.
Key North America Genetic Toxicology Testing Market Company Insights
The North America Genetic Toxicology Testing Market experiences growth because pharmaceutical and biotechnology and chemical industries require safety assessments. Genetic toxicity testing methods receive higher usage because regulatory bodies in the United States and Canada impose strict testing requirements. The market will show continuous growth because drug development pipelines keep expanding.
The North America Genetic Toxicology Testing Market sees expansion because new in vitro testing technologies eliminate the requirement for animal experimentation. Laboratories and research organizations are moving to genetic screening methods which provide both faster results and higher accuracy. The new system enables businesses to identify DNA damage at earlier points which results in safer product development throughout various sectors.
The North America Genetic Toxicology Testing Market experiences growth because life sciences research and clinical trial funding are increasing. Pharmaceutical companies are focusing on safer drug candidates which increases their need for genotoxicity testing services. The demand for reliable testing solutions will grow because consumers want to know about chemical safety in their products.
North America Genetic Toxicology Testing Market uses competitive strategies that involve laboratory network expansion and high-throughput screening system implementation. Service providers aim to reduce testing time while improving accuracy. Research institutions and contract research organizations partnership enables businesses to deliver their services more effectively while using multiple testing methods.
Company List
- Charles River Laboratories
- Eurofins Scientific
- SGS
- Intertek
- Thermo Fisher
- Labcorp
- Covance
- IQVIA
- WuXi AppTec
- Envigo
- Toxikon
- Creative Bioarray
- GenScript
- BioReliance
- Agilent
What are the Key Use-Cases Driving the Growth of North America Genetic Toxicology Testing Market?
The north american genetic toxicology testing market experiences its primary growth through pharmaceutical drug safety assessment which pharmaceutical companies utilize to conduct genotoxicity tests during their initial drug research. The north america genetic toxicology testing market exists because regulatory bodies like the FDA require preclinical testing that evaluates DNA damage and mutation risk and carcinogenic potential. The implementation of toxicology testing in drug development processes helps pharmaceutical companies minimize late-stage drug failures while achieving efficient pipelines and developing safer medical treatments.
The north america genetic toxicology testing market receives substantial support from chemical manufacturing and cosmetic and agrochemical industries which need to comply with strict workplace safety regulations and environmental protection standards. Companies use genetic toxicity assays to evaluate long-term exposure risks and product safety before commercialization. The north america genetic toxicology testing market receives support from industrial manufacturing industries which require raw material and chemical compound risk assessments to comply with current regulatory and environmental requirements.
The north america genetic toxicology testing market experiences new transformations through emerging AI-driven toxicogenomics features and predictive modeling use cases. Biotech companies and contract research organizations (CROs) adopt machine learning solutions to predict genetic toxicity results, which decreases their need for extensive laboratory testing. Testing facilities can grow their business operations through the new method which leads to lower expenses and quicker decision-making processes that assist continuous growth in pharmaceutical and chemical and life sciences markets.
North America Genetic Toxicology Testing Market Report Segmentation
By Type
- In Vitro Testing
- In Vivo Testing
- Mutagenicity Tests
- Carcinogenicity Tests
- Genotoxicity Tests
By Application
- Drug Development
- Chemicals Testing
- Food Safety
- Cosmetics
- Environmental Testing
By End-User
- Pharma
- Biotech
- CROs
- Research Institutes
- Govt
By Technology
- PCR
- Sequencing
- Microarray
- AI-based Analysis
Frequently Asked Questions
Find quick answers to common questions.
The approximate North America Genetic Toxicology Testing Market size for the market will be USD 2.11 Billion in 2033.
Key segments for the North America Genetic Toxicology Testing Market are By Type (In Vitro Testing, In Vivo Testing, Mutagenicity Tests, Carcinogenicity Tests, Genotoxicity Tests, Others); By Application (Drug Development, Chemicals Testing, Food Safety, Cosmetics, Environmental Testing, Others); By End-User (Pharma, Biotech, CROs, Research Institutes, Govt, Others); By Technology (PCR, Sequencing, Microarray, AI-based Analysis, Others).
Major North America Genetic Toxicology Testing Market players are Charles River Laboratories, Eurofins Scientific, SGS, Intertek, Thermo Fisher, Labcorp, Covance, IQVIA, WuXi AppTec, Envigo, Toxikon, Creative Bioarray, GenScript, BioReliance, Agilent.
The North America Genetic Toxicology Testing Market size is USD 0.91 Billion in 2025.
The North America Genetic Toxicology Testing Market CAGR is 11.10%.
- Charles River Laboratories
- Eurofins Scientific
- SGS
- Intertek
- Thermo Fisher
- Labcorp
- Covance
- IQVIA
- WuXi AppTec
- Envigo
- Toxikon
- Creative Bioarray
- GenScript
- BioReliance
- Agilent
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)