Europe Orthopedic Biomaterials Market Size & Forecast:
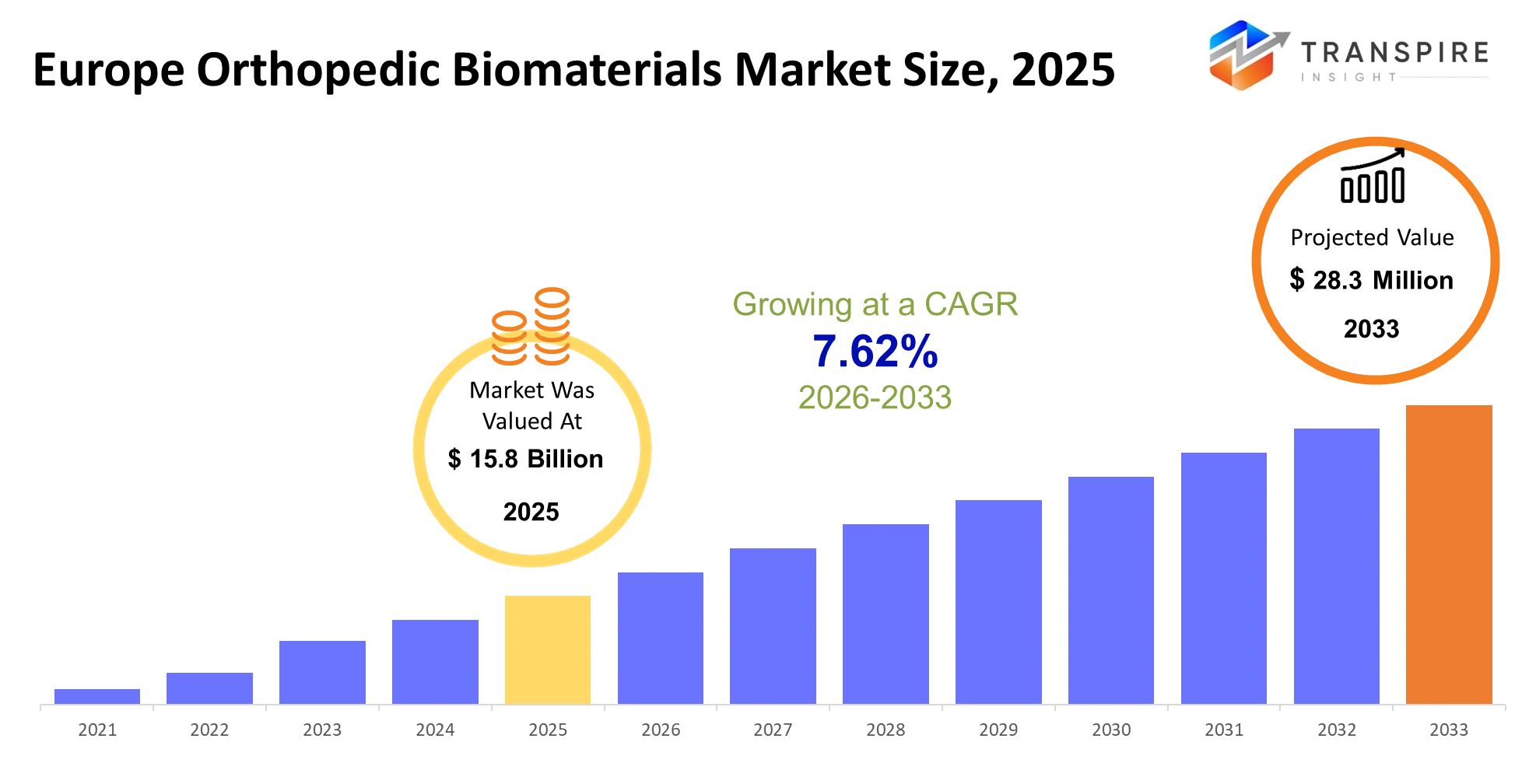
- Europe Orthopedic Biomaterials Market Size 2025: USD 15.8 Billion
- Europe Orthopedic Biomaterials Market Size 2033: USD 28.3 Million
- Europe Orthopedic Biomaterials Market CAGR: 7.62%
- Europe Orthopedic Biomaterials Market Segments: By Type (Metallic, Polymeric, Ceramic), By Application (Joint Replacement, Dental), By End-User (Hospitals)
To learn more about this report, Download Free Sample Report
Europe Orthopedic Biomaterials Market Summary:
Europe Orthopedic Biomaterials Market size was estimated at USD 15.8 Billion 2025 and is anticipated to reach USD 28.3 Million by 2033, growing at a CAGR of 7.62% from 2026 to 2033.
The European Orthopedic Biomaterials Market will lead to better medical device development which will establish enhanced treatment options for bone and joint diseases across Europe. The hospitals and specialty clinics will depend more on advanced materials which replicate natural tissue behavior to achieve better patient recovery results. The demand for medical services will change because aging patients require implants with longer lifespans and procedures that need less recovery time while they want to achieve quicker recovery and experience fewer surgical complications.
The research-based study will develop bioactive ceramics and polymer blends and surface-engineered metals which will improve material durability and human tissue integration. The European Union will establish stricter safety and traceability regulations which manufacturers must follow to implement cleaner production methods and conduct more thorough clinical testing. Sustainability concerns will shape material sourcing decisions which will promote the use of recyclable materials and ethically sourced products that meet the new expectations of both providers and patients.
Key Market Trends & Insights:
- The increasing number of elderly people in European countries has resulted in a greater number of osteoarthritis cases and bone degeneration disorders. Hospitals now implement cutting-edge biomaterials to achieve better surgical results and extended operational lifespan. The market for European orthopedic biomaterials continues to experience steady demand because customers prefer long-lasting implants which will become more popular because of advanced surgical methods.
- Ongoing research activities have developed new ceramic and polymer and composite materials which exhibit better strength and compatibility properties. Manufacturers aim to decrease product rejection rates while they work toward faster healing solutions. The development of bioactive coatings together with surface modification strategies has become popular because they enable better human tissue integration and thus become more widely accepted in orthopedic medical practices.
- European medical institutions now adopt treatment methods that minimize both patient recovery times and necessary hospital durations. Small incision biomaterials which enable accurate placement have become essential in modern medical practice. Healthcare institutions will increasingly demand lightweight flexible materials because they enhance patient comfort and enable quicker recovery times.
- European nations maintain consistent funding levels for both healthcare infrastructure development and advanced medical treatment establishment. The funding support for orthopedic research and innovation activities has enabled the development of new products. Modern surgical equipment together with experienced professionals enables healthcare facilities to implement biomaterials across their public and private medical services.
- European Union regulations establish strict quality requirements that guarantee the safety and operational effectiveness of orthopedic materials. The existing regulatory systems permit medical advancements to occur while safeguarding patient health. The European Orthopedic Biomaterials Market has experienced a shift because manufacturers now prioritize sustainable production methods and recyclable materials as their main focus for future growth.
Europe Orthopedic Biomaterials Market Segmentation
By Type :
- Metallic: The Europe Orthopedic Biomaterials Market depends on metallic materials which provide essential strength together with long-lasting durability. Common usage includes load-bearing implants such as hip and knee replacements. The orthopedic field in Europe can use titanium and stainless steel materials because these metals provide protection against corrosion while sustaining their performance throughout extended periods of operation.
- Polymeric: Polymeric biomaterials continue to gain attention in the Europe Orthopedic Biomaterials Market because of flexibility and lightweight properties. The field of medicine uses these materials for two main purposes which include soft tissue repair and the creation of cushioning components found in implants. Biocompatibility and ease of shaping support wider adoption, especially where reduced stiffness and improved comfort remain important considerations in orthopedic treatments.
- Ceramic: The Europe Orthopedic Biomaterials Market shows continuous growth for ceramic biomaterials because these materials possess outstanding wear resistance and they match human tissue compatibility. The material finds its most common application on joint surfaces that require smooth movement for proper functioning. The combination of low friction and high hardness extends the lifespan of implants, while the reduced chances of allergic reactions increase their demand for use in specialized orthopedic procedures.
To learn more about this report, Download Free Sample Report
By Application :
- Joint Replacement: Joint replacement represents a major segment within the Europe Orthopedic Biomaterials Market which experiences growth because of increasing arthritis cases and the aging population. Biomaterials function as essential components which help restore movement and decrease discomfort. The selection of advanced materials guarantees product strength through their permanent properties which enhance surgical success rates and result in enduring patient contentment.
- Dental: Dental applications provide continuous support to the Europe Orthopedic Biomaterials Market through increasing public knowledge about oral health and dental cosmetic treatments. Biomaterials function as essential materials for implants and crowns and bone grafting applications. The treatment options which dental professionals provide through their strong bone tissue compatibility and aesthetic benefits create reliable and long-lasting results..
By End-User :
- Hospitals: The Europe Orthopedic Biomaterials Market shows hospitals as its main customer base because they operate advanced surgical facilities and employ highly trained medical staff. The steady stream of patients creates a continuous need for orthopedic surgery procedures. The combination of contemporary equipment and established safety protocols enables hospitals to use biomaterials safely during their regular operations and complex medical procedures.
Country Insights
- Germany will lead due to strong healthcare infrastructure and high surgical volume. Demand for advanced orthopedic implants will remain steady because established hospitals and research centers provide reliable support. The joint replacement and trauma procedures need biomaterials which will achieve widespread usage because precision and high-quality materials will meet their requirements. The United Kingdom will experience steady growth because public healthcare funding will increase and orthopedic case numbers will rise. The combination of cost-effective and durable biomaterials will determine which products organizations choose to buy. The rising number of patients waiting for surgery will create a need for implant materials which provide both effectiveness and long-lasting performance.
- France will maintain steady demand because it conducts extensive medical research and develops new biomaterials. Hospitals will select materials which shorten recovery duration while enhancing patient comfort. The government will assist healthcare progress through its efforts to promote contemporary orthopedic treatments.
- Population growth and rising orthopedic surgeries will drive moderate expansion in Italy. There will be an increased need for biomaterials which help patients recover faster while minimizing medical problems. The regional healthcare disparities will affect how people adopt new practices but the total usage throughout the area will keep rising. The development of the healthcare system and the expanding knowledge of contemporary treatment methods will lead to incremental progress for Spain. The need for orthopedic biomaterials will increase as hospitals enhance their surgical operations. The clinical material selection process will be determined by materials that can deliver cost-effective solutions for clinical requirements.
Recent Development News
EU invests €403 million to boost medical device innovation
The European Union approved €403 million in funding to support companies developing advanced medical technologies, including biomaterials used in orthopedic implants. The funding will speed up development work on next-generation implants which contain bioresorbable materials and smart prosthetics technology throughout Europe.
Johnson & Johnson to spin off orthopedic business
Johnson & Johnson announced plans to separate its orthopedic division (DePuy Synthes), which includes implant technologies and biomaterials. The organization plans to concentrate its funding resources on research and development activities which will focus on creating new biomaterials and surgical technologies.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 15.8 Billion |
|
Market size value in 2026 |
USD 16.92 Billion |
|
Revenue forecast in 2033 |
USD 28.3 Million |
|
Growth rate |
CAGR of 7.62% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain, Rest of Europe) |
|
Key company profiled |
Zimmer Biomet, Stryker, Johnson & Johnson, Smith & Nephew, Medtronic, NuVasive, Globus Medical, Arthrex, Wright Medical, Integra LifeSciences, B. Braun, Exactech, Orthofix, Conmed, Aesculap |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Metallic, Polymeric, Ceramic), By Application (Joint Replacement, Dental), By End-User (Hospitals) |
Key Europe Orthopedic Biomaterials Company Insights
- The Europe Orthopedic Biomaterials Market shows steady growth which relies on increasing joint replacement procedures and the growing elderly population and ongoing advancements in material science. Demand remains strong for biomaterials which include ceramics and polymers and metal-based solutions that support bone repair and implant stability. Healthcare systems across Europe continue to adopt advanced biomaterials that improve recovery time and reduce complications. The regional regulatory frameworks establish safety requirements together with quality standards which enhance product trustworthiness. Research activities also play a key role because universities and medical institutions conduct studies to help manufacturers achieve better performance and longer-lasting products. The market expansion between patients and surgeons occurs because they learn about modern orthopedic solutions which results in hospitals and specialized clinics adopting these treatments.
- The Key Europe Orthopedic Biomaterials Company Insights reveal a competitive market environment which develops through three main drivers: innovation, partnerships and product development. The major companies develop materials which enable better human body compatibility while sustaining their strength and durability. The companies will continue to invest in research and development because they need to develop solutions which meet orthopedic requirements for spinal injuries and trauma treatment. The testing and development of new materials will benefit from strategic partnerships with healthcare providers who will evaluate their performance before they undergo extensive implementation. Market participants establish distribution networks throughout European countries to provide better access to their products for a larger customer base. The European Orthopedic Biomaterials Market requires companies to maintain product quality and develop effective pricing methods while following strict regulatory guidelines which helps them achieve sustainable growth and market presence.
Europe Orthopedic Biomaterials Market Companies:
- Zimmer Biomet
- Stryker
- Johnson & Johnson
- Smith & Nephew
- Medtronic
- NuVasive
- Globus Medical
- Arthrex
- Wright Medical
- Integra LifeSciences
- B. Braun
- Exactech
- Orthofix
- Conmed
- Aesculap
Europe Orthopedic Biomaterials Market Segmentation
By Type
- Metallic
- Polymeric
- Ceramic
By Application
- Joint Replacement
- Dental
By End-User
- Hospitals
Frequently Asked Questions
Find quick answers to common questions.
The Approximate Europe Orthopaedic Biomaterials Market size will be USD 28.3 Million in 2033.
Key Segments for the Europe Orthopedic Biomaterials Market are By Type (Metallic, Polymeric, Ceramic), By Application (Joint Replacement, Dental), By End-User (Hospitals).
Major Europe Orthopedic Biomaterials Market Players are Zimmer Biomet, Stryker, Johnson & Johnson, Smith & Nephew, Medtronic, NuVasive, Globus Medical, Arthrex, Wright Medical, Integra LifeSciences, B. Braun, Exactech, Orthofix, Conmed, Aesculap.
The Current Europe Orthopedic Biomaterials Market size is USD 15.8 Billion in 2025.
The Europe Orthopedic Biomaterials Market CAGR is 7.62%.
- Zimmer Biomet
- Stryker
- Johnson & Johnson
- Smith & Nephew
- Medtronic
- NuVasive
- Globus Medical
- Arthrex
- Wright Medical
- Integra LifeSciences
- B. Braun
- Exactech
- Orthofix
- Conmed
- Aesculap
Recently Published Reports
-
May 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


