Europe Live Biotherapeutic Products and Microbiome CDMO Market Size & Forecast:
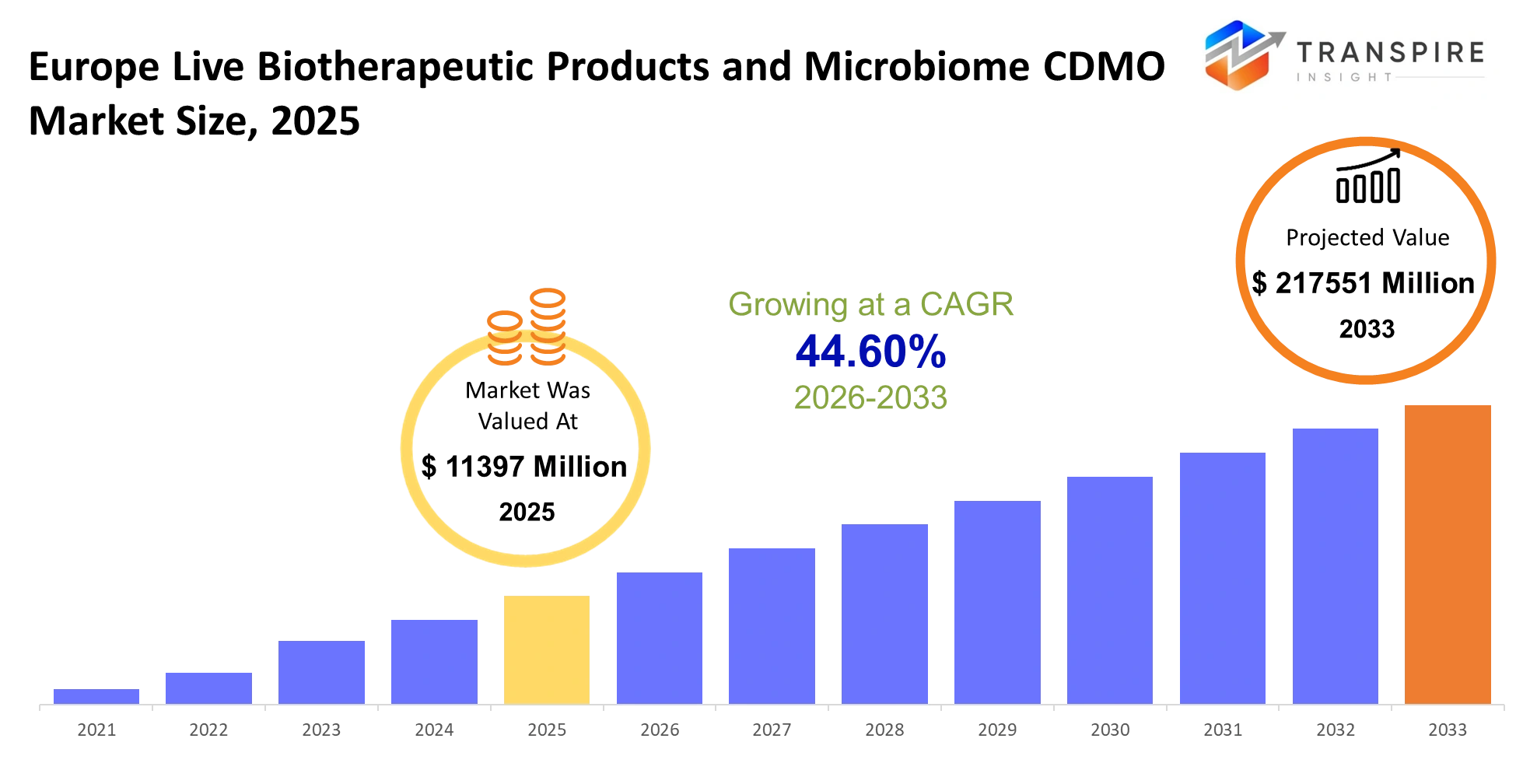
- Europe Live Biotherapeutic Products and Microbiome CDMO Market Size 2025: USD 11397 Million
- Europe Live Biotherapeutic Products and Microbiome CDMO Market Size 2033: USD 217551 Million
- Europe Live Biotherapeutic Products and Microbiome CDMO Market CAGR: 44.60%
- Europe Live Biotherapeutic Products and Microbiome CDMO Market Segments: By Type (Microbiome-based Products, Probiotic-based Products, Engineered Microbes, Others); By Application (Gut Health, Oncology, Infectious Diseases, Metabolic Disorders, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, CROs, Others); By Service (Manufacturing, Development, Analytical Testing, Regulatory Services, Others).
To learn more about this report, Download Free Sample Report
Europe Live Biotherapeutic Products and Microbiome CDMO Market Summary
The Europe Live Biotherapeutic Products and Microbiome CDMO Market was valued at USD 11397 Million in 2025. It is forecast to reach USD 217551 Million by 2033. That is a CAGR of 44.60% over the period.
The Europe Live Biotherapeutic Products and Microbiome CDMO market provides support to biotech and pharmaceutical companies by transforming microbiome research into developable treatments through their services which include GMP manufacturing and process development and analytical validation and clinical supply. The system establishes connections between initial laboratory discoveries of gut health and oncology and infectious disease research to production processes which comply with European quality requirements for controlled manufacturing. The market has developed during the last three years from academic fermentation facilities to industrial CDMO systems which establish standardized manufacturing processes through their use of automated systems and predetermined quality controls. European companies chose to outsource their manufacturing operations because COVID-19 supply chain problems and new EU regulatory requirements needed them to produce goods closer to their clinical testing locations. The combination of these factors enabled organizations to decrease production delays while maintaining their testing schedule and increasing their partnership with specialized CDMO companies which resulted in faster revenue growth for established service providers.
Key Market Insights
- The Live Biotherapeutic Products and Microbiome CDMO Market in Europe is controlled by Western Europe which holds 42% market share during 2024 because the region possesses advanced biotech infrastructure and complete regulatory systems.
- The countries of Germany and France and the Netherlands lead in both clinical pipeline development and the growth of their GMP manufacturing capabilities.
- The Eastern European region will experience its highest growth rate during the period from 2024 to 2030 because of affordable CDMO services and the rising demand for clinical trial outsourcing.
- The Microbiome CDMO services segment leads the market with a 55% share in 2024 because companies heavily outsource their fermentation and formulation tasks.
- Live biotherapeutic product development services represent the second-largest segment with increasing early-stage R&D collaboration.
- The segment of analytical testing and quality assurance services experiences the fastest growth rate because of the stricter EU GMP compliance requirements that need to be followed.
- Gastrointestinal disorder therapeutics dominate with nearly 48% share in 2024, reflecting strong clinical focus on gut microbiome modulation.
- The fastest-growing applications for oncology and immunotherapy use microbiome technology to improve drug response and immune system control.
- The Europe Live Biotherapeutic Products and Microbiome CDMO Market is primarily controlled by pharmaceutical and biotechnology companies which hold a 60% market share because they outsource most of their pipeline development work.
- The competitive advantage of organizations now relies on their capacity to implement technology that includes AI systems for strain optimization and automated biomanufacturing.
What are the Key Drivers, Restraints, and Opportunities in the Europe Live Biotherapeutic Products and Microbiome CDMO Market?
Driver:
The Europe Live Biotherapeutic Products and Microbiome CDMO Market experiences its main growth because companies now prefer to outsource their complicated microbiome production work to dedicated CDMOs. The European Union Good Manufacturing Practice standards became stricter which caused companies to stop their in-house anaerobic fermentation operations because they lacked sufficient technical expertise. Biotech companies have started to depend more on CDMO partners because they need help transforming their initial microbiome research results into market-ready clinical products. The contract manufacturing business experienced growth because companies with proven GMP facilities and full production capabilities brought their live biotherapeutic products to market faster through contract manufacturing agreements.
Restraint:
The absence of uniform regulatory procedures which govern live biotherapeutic product classification and potency assessment throughout Europe serves as a fundamental structural limitation. The regulatory process moves slowly because microbiome therapies consist of different biological components which depend on specific strains which makes it difficult to meet the approval requirements. The extended development process results in two negative effects which include delaying clinical use and decreasing immediate income generation for both CDMOs and therapy developers. The existing complexity requires time to dissolve because EU health authorities must work together with multiple agencies to achieve harmonization.
Opportunity:
The combination of advanced synthetic microbiome engineering and modular CDMO facilities creates a major opportunity in the current market. Countries like France and Denmark have developed scalable anaerobic bioreactors which now support quicker clinical batch production operations. The use of AI-powered strain selection systems by CDMOs enables them to decrease their product development periods while establishing Europe as the main production center for advanced microbiome therapeutics.
What Has the Impact of Artificial Intelligence Been on the Europe Live Biotherapeutic Products and Microbiome CDMO Market?
Advanced digital technologies and artificial intelligence technologies enable real-time monitoring and operational control systems to transform scrubber performance systems and exhaust gas cleaning technology. AI-driven automation operates modern marine emission control systems by controlling scrubber water flow and pH balance and exhaust treatment cycles without human operators needing to monitor these processes. The system reduces manual calibration errors while delivering better compliance results with IMO sulfur regulations which show better performance in European shipping corridors that experience high traffic.
The organization uses machine learning models for predictive maintenance operations which analyze pump and valve and corrosion-prone component sensor data to identify potential equipment failures before they happen. The operators experience operational enhancements which produce 10 to 15 percent declines in maintenance downtime and maintain better fuel efficiency through improved exhaust backpressure control. Emissions forecasting tools help voyage planning by providing compliance cost estimates for different fuel and route scenarios which enhance operational budgeting precision.
A structural barrier prevents full adoption of the system. Many AI systems face challenges when working with incomplete or noisy maritime data because they need to process actual ocean conditions which include extreme weather and equipment failure and intermittent connection problems. The high costs required to retrofit existing ships with new systems constrain their widespread adoption. Digital optimization continues to enhance compliance reliability and decrease operational expenses throughout the entire operational life of European fleets despite existing challenges.
Key Market Trends
- The regulatory environment establishes new requirements which compel manufacturers to implement standardized validation procedures for microbial strains between 2022 and 2025.
- The surge in clinical-grade microbiome demand from 2023 onward led CDMOs such as Lonza and Biose Industrie to expand their anaerobic manufacturing capabilities.
- Academic microbiome research at the early stage converted into industry partnerships which improved translational research processes and shortened commercial development times since 2022.
- The market began to prefer specific bacterial strains used in live biotherapeutic products which treat cancer and immune system disorders after 2021.
- European biotech funding increasingly prioritized microbiome platforms with venture investments which originated from CDMO-linked startups growing two times between 2022 and 2025.
- European production facilities became the primary source for supply chains which reduced their need for US microbial fermentation plants after COVID-19 operational interruptions.
- Leading CDMOs implemented automated systems with digital bioprocess monitoring technologies to substitute traditional manual quality assessment methods between 2023 and 2026.
- Sartorius AG and Evonik Industries expanded their partnership network to create scalable microbiome production systems while their competitive behavior transformed into joint business strategies.
Europe Live Biotherapeutic Products and Microbiome CDMO Market Segmentation
By Type:
The European market for Live Biotherapeutic Products and Microbiome Contract Development Manufacturing Organizations currently sees its most successful product category through Microbiome-based products which scientists have developed for gut and immune therapies. Pharmaceutical companies develop their product pipelines to focus on these products because they provide more precise results than standard biologics this creates ongoing customer needs for CDMO services. The current market leadership of this company originates from its successful initial product launch and its increasing number of clinical studies that reach advanced testing stages throughout Europe.
Engineered microbes represent the fastest-growing type segment, driven by advancements in synthetic biology and precision gene editing. Engineered strains provide targeted treatment capabilities which differ from probiotic-based products because they treat cancer and metabolic disorders that conventional methods cannot handle. The growth of next-generation microbial engineering platforms receives support through increased venture funding for their development.
The future will see engineered microbial systems and hybrid systems achieve greater dominance when regulatory bodies establish clear guidelines for manufacturing systems that achieve industrial production volumes. The CDMO providers will start allocating their resources to develop advanced bioprocess services which require their expertise to handle complex microbial strains.

To learn more about this report, Download Free Sample Report
By Application:
The Europe Live Biotherapeutic Products and Microbiome CDMO Market shows dominance of gut health applications because microbiome-gut axis therapies have received strong clinical validation. European populations experience high gastrointestinal disorder rates which create continuous market demand while preexisting clinical trial systems boost product approval processes within this market.
The quickest growth in oncology applications occurs because research shows how microbiome factors affect immunotherapy results. Pharmaceutical companies are increasingly integrating microbial modulation into cancer treatment pipelines which creates strong demand for CDMO-supported clinical manufacturing and analytical services.
Research will shift toward oncology and metabolic disorders because upcoming studies will explore these areas. The transition shows how microbiome-based treatments will expand their use from treating gut disorders to managing entire body diseases.
By End-User:
The European Live Biotherapeutic Products and Microbiome CDMO Market has its largest demand share which pharmaceutical companies control because they outsource all their manufacturing and clinical supply work. The companies use CDMOs because they want to decrease their capital expenses while speeding up the process of bringing microbiome therapies to market.
Biotech firms represent the fastest-growing end-user group, driven by rising early-stage innovation in microbial therapeutics. Emerging biotech clusters in Europe need CDMO partners because their organizations lack sufficient capacity to produce regulatory-grade products while scaling up their operations.
Research institutes and biotech firms will increase their adoption of future technologies because funding for research will expand and collaboration networks will become more effective. CDMO dependency will deepen as product complexity rises and regulatory expectations tighten across European markets.
By Service:
The Europe Live Biotherapeutic Products and Microbiome CDMO Market maintains its manufacturing services as the dominant force because GMP-grade microbial fermentation and scale-up processes require advanced technical expertise. Sterile anaerobic production needs create high entry obstacles which lead companies to choose established CDMO operators for their services.
The fastest growth in development services occurs because companies want to optimize their processes while receiving support for their strain engineering needs. Biotech companies demand specialized CDMO services to achieve shorter development timelines and better success rates in clinical transitions.
The future of growth will focus on integrated service models which combine manufacturing and development and regulatory support. CDMOs that provide complete end-to-end services will obtain better control over their pricing and will secure long-term customer agreements throughout Europe.
What are the Key Use Cases Driving the Europe Live Biotherapeutic Products and Microbiome CDMO Market?
The European market for Live Biotherapeutic Products and Microbiome CDMO services relies primarily on gut health therapies because European populations experience high rates of gastrointestinal disorders. Pharmaceutical companies prioritize these applications because clinical validation of microbiome-gut interactions has progressed faster here than in other therapeutic areas, creating a stable pipeline for outsourced GMP manufacturing.
Biotech firms and academic research institutes show rapid growth in their use of oncology and infectious disease applications. Cancer immunotherapy integration with microbiome modulation has created new trial pipelines, while post-antibiotic infection management programs are increasing reliance on CDMO-supported clinical batch production and analytical testing services.
Metabolic disorder treatments and neurological applications represent emerging use cases with strong long-term potential. Early studies have established links between gut microbiota and diabetes as well as neuroinflammation, which has created interest among venture-backed biotech companies to pursue development in this area. This development indicates that the Europe Live Biotherapeutic Products and Microbiome CDMO Market will expand beyond its current therapeutic limitations.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 11397 Million |
|
Market size value in 2026 |
USD 16457 Million |
|
Revenue forecast in 2033 |
USD 217551 Million |
|
Growth rate |
CAGR of 44.60% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain, Rest of Europe) |
|
Key company profiled |
Lonza, Catalent, WuXi AppTec, Samsung Biologics, Recipharm, Evotec, Charles River, Boehringer Ingelheim, Thermo Fisher, Fujifilm Diosynth, PCI Pharma, Delpharm, Siegfried, Cambrex, Almac Group. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Microbiome-based Products, Probiotic-based Products, Engineered Microbes, Others); By Application (Gut Health, Oncology, Infectious Diseases, Metabolic Disorders, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, CROs, Others); By Service (Manufacturing, Development, Analytical Testing, Regulatory Services, Others). |
Which Regions are Driving the Europe Live Biotherapeutic Products and Microbiome CDMO Market Growth?
The Europe Live Biotherapeutic Products and Microbiome CDMO Market finds its leading position in Western Europe because this region possesses advanced biotechnology infrastructure while it adheres to EMA regulatory standards. Germany France and the Netherlands contain numerous GMP-certified CDMO facilities which enable fast development of microbiome therapies through their clinical testing services. The pharmaceutical industry together with academic and industrial partnerships maintains this dominance by supplying their early-stage research results to commercial development processes. The strict quality standards enforcement system pushes innovators to select Western European manufacturing centers which helps these regions maintain their industry leadership position.
The northern European region provides dependable support through its three main countries Denmark Sweden and Finland. The region develops through public research funding and sober advanced therapeutic regulatory processes whereas Western Europe expands through business growth. The combination of government-supported life sciences initiatives and dependable healthcare systems creates an environment which attracts biotech investors for extended periods. The scientific innovation shipowners in this region focus on making gradual clinical improvements which leads to slow yet consistent growth in microbiome manufacturing requirements.
Eastern and Southern Europe constitute the fastest growing area in Europe because Poland Italy and Spain have recently invested in their biotechnology parks and clinical research centers. The EU modernization programs together with reduced operational expenses have created an environment where CDMO outsourcing now operates outside its former Western European base. Biotech startups which partner with universities through their early-stage operations have driven up requirements for manufacturing assistance.The current changes demonstrate that investors who make their investments between 2026 and 2033 will discover two types of valuable resources which include cost-efficient production centers and developing clinical systems that can expand their operations.
Who are the Key Players in the Europe Live Biotherapeutic Products and Microbiome CDMO Market and How Do They Compete?
The European Live Biotherapeutic Products and Microbiome CDMO Market shows moderate consolidation which results in a few specialized CDMOs maintaining control over advanced GMP fermentation operations. The existing companies maintain their market position through their specialized knowledge of regulations and their operational manufacturing systems and their existing agreements with pharmaceutical companies while the new companies face obstacles from the expensive startup costs and the strict regulations that exist in the European Union. Clients choose to work with their service providers based on technological abilities and operational reliability because they consider contamination protection and genetic material stability and clinical-grade product reproducibility to be more crucial than financial advantages.
Through its complete biologics manufacturing system Lonza Group establishes stronger market presence by utilizing its extensive GMP production facilities and modern microbial manufacturing technologies. The company can establish enduring partnerships with pharmaceutical companies because it provides services for both preliminary product development and full-scale commercial manufacturing operations. Sartorius AG establishes its market presence through unique technological solutions which deliver bioprocessing systems and digital monitoring solutions that help CDMO partners achieve better fermentation results and decreased batch production inconsistencies. The equipment-based approach enables European manufacturers to achieve better operational integration within their production processes.
Biose Industrie and Evonik Industries pursue niche specialization strategies in microbial fermentation and strain-specific production systems. Biose Industrie differentiates itself from competitors through its special live biotherapeutic GMP facilities which operate in France to provide biotech companies with speedy clinical batch testing services. Evonik establishes its business operations through microbial strain development and its partnerships with biotech startups which enable it to develop advanced microbiome therapies.The companies which work together with their collaboration networks, expand their operational capacity, which strengthens the competitive contract development and manufacturing organization ecosystem of Europe.
Company List
- Lonza
- Catalent
- WuXi AppTec
- Samsung Biologics
- Recipharm
- Evotec
- Charles River
- Boehringer Ingelheim
- Thermo Fisher
- Fujifilm Diosynth
- PCI Pharma
- Delpharm
- Siegfried
- Cambrex
- Almac Group
Recent Development News
In October 2025, Lonza announced continued expansion of its CDMO capacity with strong integration of its Vacaville biologics site in the United States. The company confirmed new long-term commercial supply contracts for integrated biologics and bioconjugates manufacturing, strengthening its global CDMO leadership in complex biologics and microbial platforms.
Source: https://www.lonza.com
In January 2025, Boehringer Ingelheim expanded its biologics and oncology pipeline through a licensing agreement for advanced antibody-drug conjugate technology. The move strengthens its long-term biotherapeutic development capabilities and indirectly supports microbiome-linked oncology research collaborations across European innovation hubs.
Source: https://www.boehringer-ingelheim.com
What Strategic Insights Define the Future of the Europe Live Biotherapeutic Products and Microbiome CDMO Market?
The Europe Live Biotherapeutic Products and Microbiome CDMO Market is changing its business operations to adopt complex biomanufacturing systems which enable CDMOs to become permanent partners for clinical development. The market shift is happening because companies now develop specialized therapeutic pipelines which require specific strains and because European Union regulations now mandate complete production systems instead of fragmented outsourcing methods.
Approval for new live biotherapeutics will face delays because of existing regulatory differences among European agencies which create obstacles for achieving consistent approval standards. The process of establishing potency standards and strain classification systems will create delays which will affect commercialization and force developers to work in compliant centers which will raise the risk of system failures.
The development of GMP micro-facilities which use decentralized modular systems to produce clinical batches has created an opportunity for Eastern European markets which are now investing in their infrastructure. The flexible units provide biotechnology companies with two advantages because they enable faster trial responses while reducing supply chain delays. Market participants should build scalable manufacturing networks which use digital technology and build partnerships with local biotech clusters to gain advantages in market competition.
Europe Live Biotherapeutic Products and Microbiome CDMO Market Report Segmentation
By Type
- Microbiome-based Products
- Probiotic-based Products
- Engineered Microbes
- Others
By Application
- Gut Health
- Oncology
- Infectious Diseases
- Metabolic Disorders
- Others
By End-User
- Pharma Companies
- Biotech Firms
- Research Institutes
- CROs
- Others
By Service
- Manufacturing
- Development
- Analytical Testing
- Regulatory Services
- Others
Frequently Asked Questions
Find quick answers to common questions.
The Europe Live Biotherapeutic Products and Microbiome CDMO Market size is USD 217551 Million in 2033.
Key segments for the Europe Live Biotherapeutic Products and Microbiome CDMO Market are By Type (Microbiome-based Products, Probiotic-based Products, Engineered Microbes, Others); By Application (Gut Health, Oncology, Infectious Diseases, Metabolic Disorders, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, CROs, Others); By Service (Manufacturing, Development, Analytical Testing, Regulatory Services, Others).
Major Europe Live Biotherapeutic Products and Microbiome CDMO Market players are Lonza, Catalent, WuXi AppTec, Samsung Biologics, Recipharm, Evotec, Charles River, Boehringer Ingelheim, Thermo Fisher, Fujifilm Diosynth, PCI Pharma, Delpharm, Siegfried, Cambrex, Almac Group.
The Europe Live Biotherapeutic Products and Microbiome CDMO Market size is USD 11397 Million in 2025.
The Europe Live Biotherapeutic Products and Microbiome CDMO Market CAGR is 44.60% from 2026 to 2033.
- Lonza
- Catalent
- WuXi AppTec
- Samsung Biologics
- Recipharm
- Evotec
- Charles River
- Boehringer Ingelheim
- Thermo Fisher
- Fujifilm Diosynth
- PCI Pharma
- Delpharm
- Siegfried
- Cambrex
- Almac Group
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


