Europe Lipid Nanoparticles Market Size & Forecast:
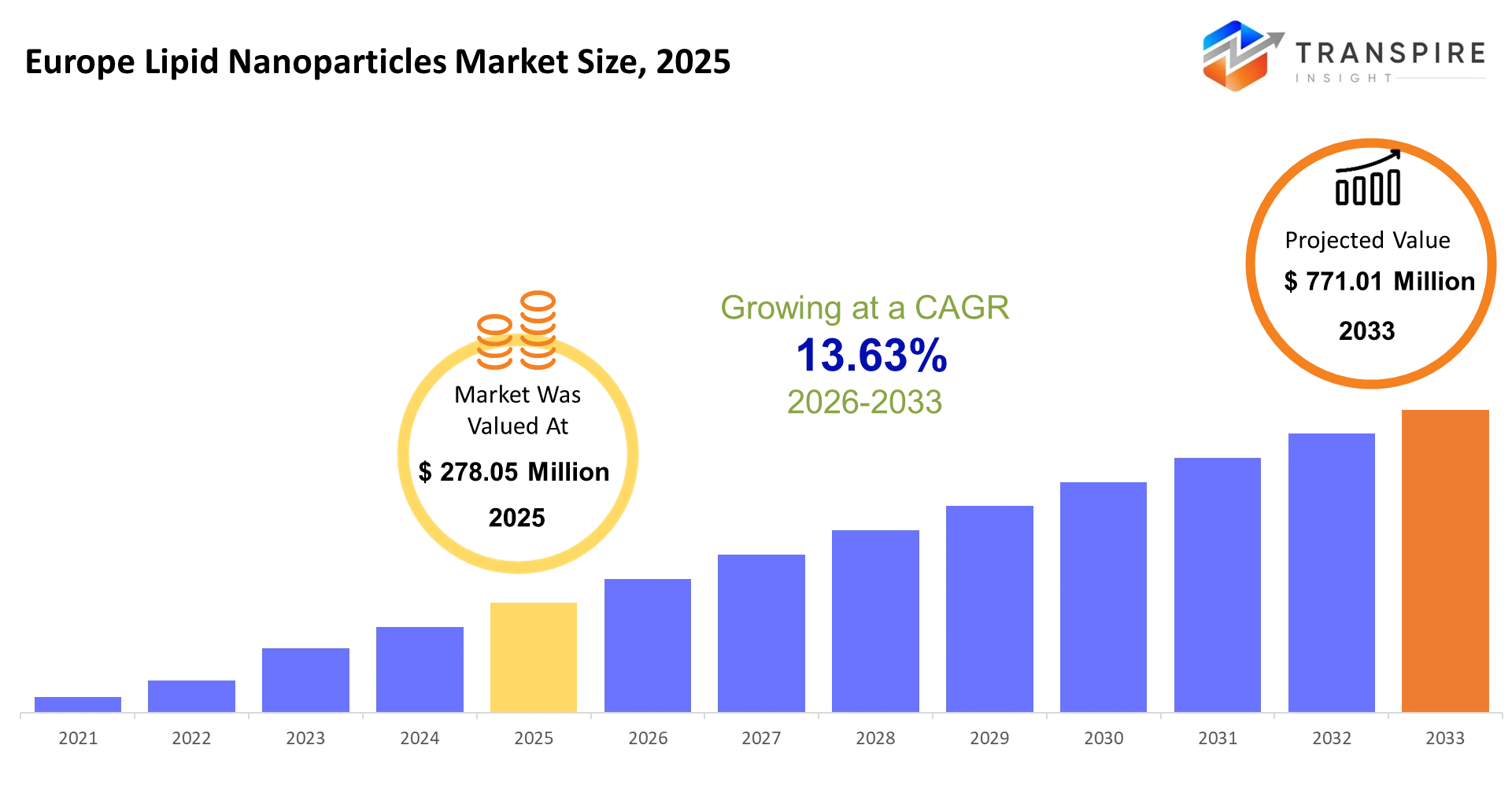
- Europe Lipid Nanoparticles Market Size 2025: USD 278.05 Million
- Europe Lipid Nanoparticles Market Size 2033: USD 771.01 Million
- Europe Lipid Nanoparticles Market CAGR: 13.63%
- Europe Lipid Nanoparticles Market Segments: By Type (Solid Lipid Nanoparticles, Nanostructured Lipid Carriers, Liposomes, Others); By Application (Drug Delivery, Vaccines, Gene Therapy, Cancer Treatment, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Hospitals, Academic Institutions, CROs, Others); By Formulation (Liquid, Powder, Suspension, Others)
To learn more about this report, Download Free Sample Report
Europe Lipid Nanoparticles Market Summary:
The Europe Lipid Nanoparticles Market size is estimated at USD 278.05 Million in 2025 and is anticipated to reach USD 771.01 Million by 2033, growing at a CAGR of 13.63% from 2026 to 2033.
The European lipid nanoparticles market in advanced drug delivery systems will experience changes because of precision medicine development through mRNA treatments and cancer drug development and research on uncommon diseases. The market will develop because doctors need specific delivery mechanisms which can decrease side effects while enhancing treatment outcomes. The European Union will enforce stricter regulations which will require higher quality and safety standards for gene therapy nanocarriers. The manufacturing industry will develop scalable and reproducible processes for lipid formulation through partnerships between biotechnology companies and public health organizations. Patients will increasingly demand quicker access to personalized treatments which will drive advancements in lipid-based delivery systems and transform treatment development and distribution methods.
What Has the Impact of Artificial Intelligence Been on the Europe Lipid Nanoparticles Market?
Artificial intelligence is currently transforming European markets for lipid nanoparticles through its ability to improve data analysis methods which companies use to make decisions and develop new products. The European lipid nanoparticles market uses AI-based analytics solutions which enable businesses to conduct comprehensive market assessments by analyzing intricate data from clinical trials and formulation testing and regulatory outcomes. Artificial intelligence in Europe lipid nanoparticles market applications will support predictive analytics which enables stakeholders to forecast demand trends and develop their production plans based on future therapeutic requirements. The new approach will enhance strategic planning efforts while decreasing investment decision-making uncertainty.
The European lipid nanoparticles market uses AI technology to enhance production efficiency through automated systems which use machine learning to improve lipid formulation operations. These systems will improve production consistency through reduced raw material consumption while decreasing product development duration. AI-based forecasting solutions will enhance inventory management and decrease transportation expenses for supply chain networks in the European lipid nanoparticles market. Companies that implement intelligent systems will create personalized drug delivery systems which enable them to stay ahead of their competitors through better adaptation to changing healthcare requirements and market shifts.
Key Market Trends & Insights:
- The europe lipid nanoparticles market is mostly controlled by Western Europe which holds approximately 45% of the market share in 2025 because of its advanced pharmaceutical facilities.
- The period between 2025 and 2030 will witness Eastern Europe developing into the most rapidly advancing region because its clinical research activities are increasing while global biotech companies expand their operations.
- Solid lipid nanoparticles dominate the market because they provide drug delivery systems with stability and scalability benefits which achieve over 40% market share in 2025.
- The second-largest market share of nanostructured lipid carriers exists because they enable better drug loading and formulation flexibility for users.
- The segment of lipid-based mRNA delivery systems will experience the highest growth rate until 2030 because vaccine development and gene therapy research are increasing.
- The europe lipid nanoparticles market shows its primary focus on oncology applications which hold about 38% of the market share because of their support for targeted cancer therapies and precision medicine development.
- The infectious disease sector will experience its highest growth rate because vaccine development resources are increasing and there is a rising need for quick-response solutions.
- Pharmaceutical and biotechnology companies hold more than 55% of the market share because they use their internal research and development capabilities to create commercial products.
- Research institutes and academic centers will experience fast growth until 2030 because funding programs and collaborative innovation approaches will support their development.
- The combination of regional development in emerging European markets and investments into operational manufacturing capacity improvements is creating better supply chain systems and allowing companies to enter new markets.
Europe Lipid Nanoparticles Market Segmentation
By Type:
Solid lipid nanoparticles will maintain their market position because of their ability to deliver pharmaceuticals through stable controlled release mechanisms. The drug delivery system will use nanostructured lipid carriers which provide superior drug loading capabilities. Liposomes will continue to be the preferred choice because of their biocompatibility while other lipid-based systems will enable specific formulations and distinct medical applications.
By Application:
Drug delivery will lead due to increasing demand for targeted and efficient treatment methods across chronic conditions. The vaccine market will grow because mRNA platforms receive ongoing development. The European lipid nanoparticles market will experience growth through gene therapy and cancer treatment while other applications support new treatment areas.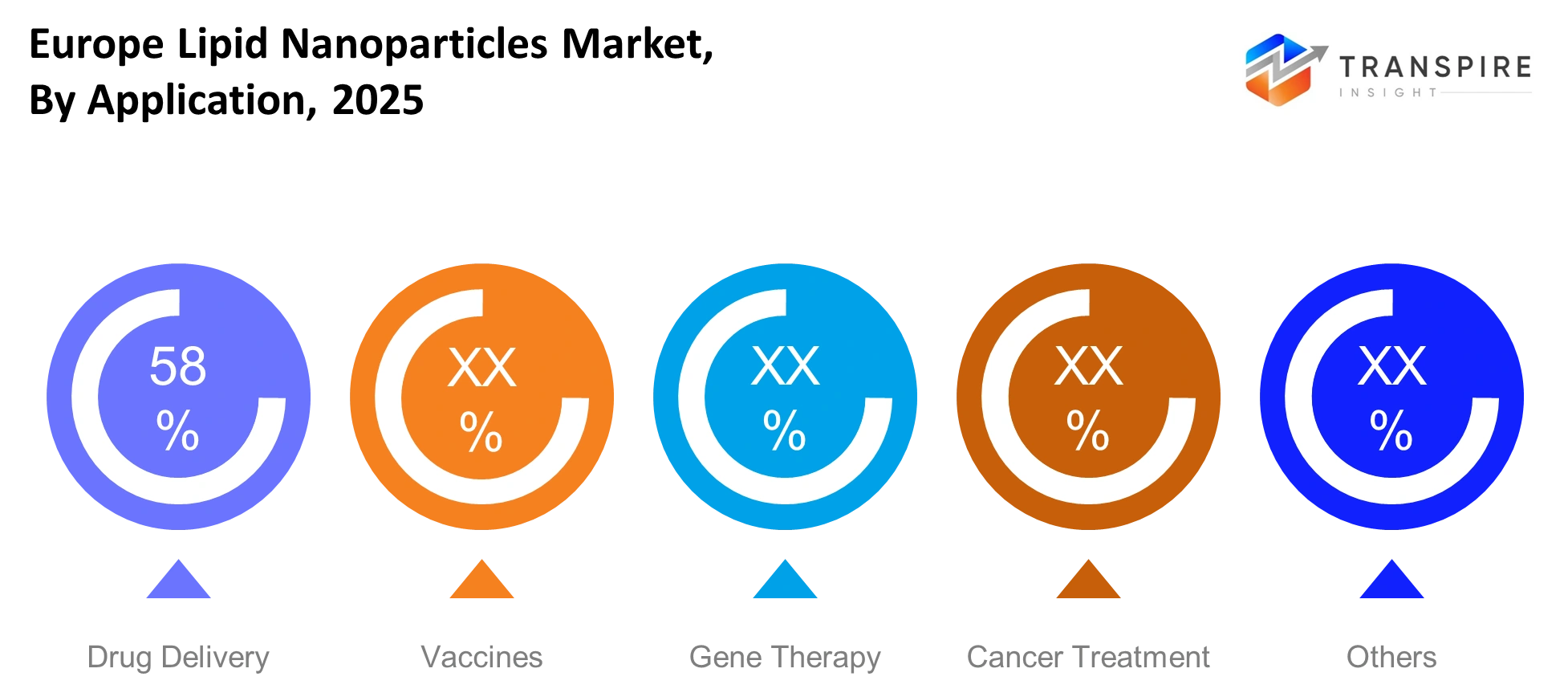
To learn more about this report, Download Free Sample Report
By End-User:
Pharma companies will dominate the market because they possess both high production capacity and strong financial backing. Biotech firms will drive innovation through specialized research. Research institutes and academic institutions will support early-stage development, while hospitals and CROs will contribute to clinical validation and trial execution.
By Formulation:
Liquid formulations will remain widely used for ease of administration and rapid absorption. The demand for powder forms will increase because they offer longer shelf life and better transportation benefits. Suspension formats will support stability in complex compounds, while other formulations will address specific delivery requirements across evolving therapeutic needs.
What are the Main Challenges for the Europe Lipid Nanoparticles Market Growth?
The European lipid nanoparticles market encounters significant technical and operational difficulties which result in reduced application performance. The stability of lipid formulations represents a primary challenge because their effectiveness depends on temperature and storage conditions. Quality control testing becomes difficult because the formulation process requires complex procedures which result in different particle sizes. Production times in the European lipid nanoparticles market face additional difficulties because supply chain disruptions for specialized lipids and raw materials have become a major production bottleneck.
The European lipid nanoparticles market faces two major obstacles which prevent companies from expanding their production capabilities. The manufacturing process demands specialized facilities which must comply with Good Manufacturing Practice regulations, resulting in increased operational expenses. The majority of businesses face difficulties when they attempt to transform their laboratory achievements into market-ready products because they need to maintain their product standards. The approval process for regulations in European countries requires extensive time, which results in product release delays while creating obstacles that hinder market entry, which needs to be solved for business expansion.
The Europe lipid nanoparticles market develops through adoption challenges which research shows to be essential to its development. The implementation process faces major obstacles because specialized facilities remain inaccessible and there exists a deficiency of trained personnel. The market development faces restrictions because research institutions experience funding problems from budget limitations and uneven resource allocation. The combination of these elements creates obstacles which prevent companies from adopting new technologies while restricting their market reach.
The European lipid nanoparticles market faces future dangers which arise from the competitive threat that comes from different drug delivery systems. Non-lipid nanocarriers and polymer-based systems now compete with existing products because they provide similar advantages to customers. The market landscape will experience shifts because pricing pressures and changing regulatory requirements will determine how customers adopt products and companies establish their market presence.
Regional Insights
Western Europe will retain its market leadership because it has strong pharmaceutical manufacturing bases and advanced research centers and its major economies including Germany and France maintain consistent regulatory alignment. The biotechnology sector and clinical trials will create continuous demand because existing infrastructure will enable successful development and marketing of lipid-based delivery systems.
Northern Europe will achieve dependable growth because of research programs supported by the government and funding for innovative projects. The drug delivery research in Sweden and Denmark will bring together academic institutions and biotech companies to work on advanced research. The ecosystem will help early-stage development while making it easier for people to access new treatment technologies.
Eastern Europe will see its healthcare system develop because its healthcare infrastructure improves and more people join clinical studies. Cost advantages and supportive policies will attract outsourcing activities from larger markets. Advanced therapies will see increased usage because awareness grows but their development will proceed at a slower pace than in Western Europe.
Recent Development News
In April 2026, Merck KGaA entered early-stage discussions to acquire Inhibrx Biosciences’ experimental cancer drug assets. The move will aim to strengthen oncology pipelines and expand advanced biologics linked to lipid-based delivery systems. https://www.reuters.com
In March 2026, Merck KGaA reported a projected decline in 2026 earnings due to currency pressure and patent expirations, while maintaining focus on biotech and laboratory technologies. The update will reflect ongoing financial adjustments influencing investment strategies in advanced drug delivery platforms. https://www.reuters.com
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 278.05 Million |
|
Market size value in 2026 |
USD 315.31 Million |
|
Revenue forecast in 2033 |
USD 771.01 Million |
|
Growth rate |
CAGR of 13.63% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain, Rest of Europe) |
|
Key company profiled |
Merck KGaA, Thermo Fisher, Evonik, Precision NanoSystems, Pfizer, Moderna, BioNTech, GSK, AstraZeneca, Sanofi, Novartis, Lonza, AbbVie, Roche, Acuitas Therapeutics |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Solid Lipid Nanoparticles, Nanostructured Lipid Carriers, Liposomes, Others); By Application (Drug Delivery, Vaccines, Gene Therapy, Cancer Treatment, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Hospitals, Academic Institutions, CROs, Others); By Formulation (Liquid, Powder, Suspension, Others) |
How Can New Companies Establish a Strong Foothold in the Europe Lipid Nanoparticles Market?
New entrants aiming to succeed in the europe lipid nanoparticles market will need to focus on clear differentiation and targeted innovation rather than broad competition. The specialized nature of demand in the European lipid nanoparticles market enables companies to achieve better market penetration when they develop products for specific medical areas which include rare diseases and personalized medicine. Emerging companies will achieve market alignment through their focus on developing solutions for problems which include formulation stability and delivery accuracy and storage capacity constraints.
The Europe lipid nanoparticles market will use innovation strategies as the main method to establish business credibility. Startups that invest in advanced formulation techniques, including AI-assisted design and scalable production methods, will position themselves ahead of traditional approaches. Focused research combined with proprietary delivery platforms has enabled companies such as NanoVation Therapeutics and Evox Therapeutics to accelerate their development processes while attracting strategic partnerships. The ability to differentiate technology through better lipid efficiency and lower toxicity will continue to be a crucial element for maintaining market competition.
Companies establishing entry into the market will benefit from strategic partnerships. The partnerships between pharmaceutical companies and research organizations and contract manufacturing organizations will enable access to vital infrastructure and regulatory knowledge and financial support. The European lipid nanoparticles market allows companies to decrease market entry risks through operational alliances which enable rapid product introduction. Companies that succeed in this competitive environment will establish better market positions through their combination of innovative practices with partnership-based growth strategies.
Key Europe Lipid Nanoparticles Market Company Insights
Pharmaceutical companies with existing manufacturing capabilities and extensive research funding will create strong competitive challenges. Market participants will focus on improving lipid formulation quality, ensuring stability, and enhancing delivery efficiency. Continuous development of proprietary technologies will support differentiation and strengthen long-term competitive positioning in the market.
Emerging biotech firms will introduce focused innovation through specialized delivery platforms and targeted therapies. Research institutions will help speed up product development through their collaborative work with researchers. Smaller companies will use strategic licensing agreements and joint ventures to access resources for production scaling and industry visibility enhancement.
Organizations will attain competitive advantage through their ability to meet regulatory requirements while controlling expenses and bringing products to market quickly. European regulations require companies to establish scalable production systems and quality assurance processes for their operations. The company will maintain its competitive advantage by expanding into new markets which lack services and offering products that match existing healthcare requirements.
Company List
- Merck KGaA
- Thermo Fisher
- Evonik
- Precision NanoSystems
- Pfizer
- Moderna
- BioNTech
- GSK
- AstraZeneca
- Sanofi
- Novartis
- Lonza
- AbbVie
- Roche
- Acuitas Therapeutics
What are the Key Use-Cases Driving the Growth of the Europe Lipid Nanoparticles Market?
The europe lipid nanoparticles market exists because advanced drug delivery applications need both precise and efficient solutions for their operations. Lipid nanoparticles serve as effective carriers which deliver sensitive biological materials like mRNA and siRNA, thus improving treatment results while decreasing adverse reactions. This application has become essential for vaccine research and targeted treatment development, which helps European companies grow their lipid nanoparticle business operations.
The european market for lipid nanoparticles uses oncology treatment as its second major application area. Lipid-based carriers enable targeted delivery of anticancer drugs, which improves treatment efficacy while protecting healthy cells from harm. Pharmaceutical companies are increasingly adopting these systems in chemotherapy and immunotherapy pipelines, which helps them achieve better success rates while gaining traction among specialized medical fields.
The europe lipid nanoparticles market is experiencing growth because gene therapy and rare disease treatment have become important market drivers. The applications need advanced lipid formulations because they require stable and efficient delivery systems which handle genetic material. The ongoing research in genetic medicine will create new treatment methods which will lead to personalized medical solutions and future treatment pathways.
The market for lipid nanoparticles in Europe is showing increased demand from research and development centers which operate beyond basic medical functions. Academic institutions and biotech firms are using lipid nanoparticles in experimental therapies and formulation testing, which creates new business possibilities for the entire life sciences industry.
Europe Lipid Nanoparticles Market Report Segmentation
By Type
- Solid Lipid Nanoparticles
- Nanostructured Lipid Carriers
- Liposomes
- Others
By Application
- Drug Delivery
- Vaccines
- Gene Therapy
- Cancer Treatment
- Others
By End-User
- Pharma Companies
- Biotech Firms
- Research Institutes
- Hospitals
- Academic Institutions
- CROs
- Others
By Formulation
- Liquid
- Powder
- Suspension
- Others
Frequently Asked Questions
Find quick answers to common questions.
The Approximate Europe Lipid Nanoparticles Market size for the Market will be USD 771.01 Million in 2033.
Key segments for the Europe Lipid Nanoparticles Market are By Type (Solid Lipid Nanoparticles, Nanostructured Lipid Carriers, Liposomes, Others); By Application (Drug Delivery, Vaccines, Gene Therapy, Cancer Treatment, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Hospitals, Academic Institutions, CROs, Others); By Formulation (Liquid, Powder, Suspension, Others)
Major Europe Lipid Nanoparticles Market Players are Merck KGaA, Thermo Fisher, Evonik, Precision NanoSystems, Pfizer, Moderna, BioNTech, GSK, AstraZeneca, Sanofi, Novartis, Lonza, AbbVie, Roche, Acuitas Therapeutics.
The Current Europe Lipid Nanoparticles Market size is USD 278.05 Million in 2025.
The Europe Lipid Nanoparticles Market CAGR is 13.63%.
- Merck KGaA
- Thermo Fisher
- Evonik
- Precision NanoSystems
- Pfizer
- Moderna
- BioNTech
- GSK
- AstraZeneca
- Sanofi
- Novartis
- Lonza
- AbbVie
- Roche
- Acuitas Therapeutics
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


