North America Pediatric Drugs Market Size & Forecast:
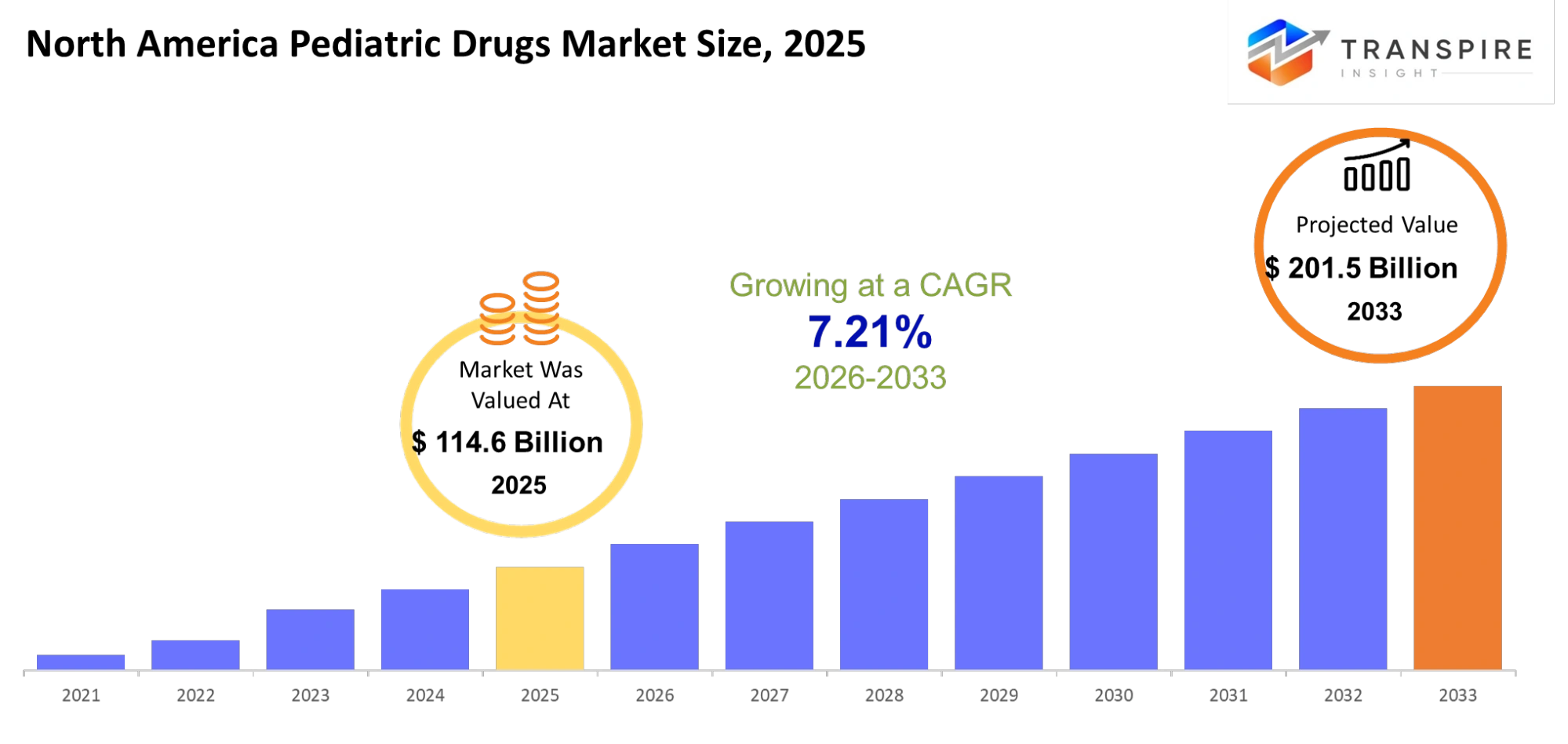
- North America Pediatric Drugs Market Size 2025: USD 114.6 Billion
- North America Pediatric Drugs Market Size 2033: USD 201.5 Billion
- North America Pediatric Drugs Market CAGR: 7.21%
- North America Pediatric Drugs Market Segments: By Type (Vaccines, Anti-infectives, CNS Drugs, Others, Respiratory, Oncology), By Route (Oral, Injectable, Topical, Others, Inhalation, Transdermal), By Application (Infections, Neurology, Respiratory, Others, Oncology, Rare Diseases), By End-User (Hospitals, Clinics, Pharmacies, Others, Homecare, Research).
To learn more about this report, Download Free Sample Report
North America Pediatric Drugs Market Summary:
The North America Pediatric Drugs Market size is estimated at USD 114.6 Billion in 2025 and is anticipated to reach USD 201.5 Billion by 2033, growing at a CAGR of 7.21% from 2026 to 2033. The North America Pediatric Drugs Market is growing steadily because people now understand the healthcare requirements of children better and because researchers create better medicines for children.
Pharmaceutical companies are developing age-appropriate medication delivery systems which include liquid forms and chewable tablets and dispersible tablets to help children better follow their treatment plans. The North America Pediatric Drugs Market is growing because more children now suffer from chronic diseases which include asthma and diabetes and infectious diseases. Government programs and regulatory incentives which support pediatric drug research create innovation opportunities in this field.
The North America Pediatric Drugs Market growth now receives support from increased demand for precise medical treatments and safe medicines developed for children. Clinical trials which test pediatric medications are now receiving funding from companies which want to improve treatment results while decreasing side effects. The increasing use of biologic drugs and specialty medications leads to better treatment outcomes for patients who have rare pediatric diseases. The North America Pediatric Drugs Market will continue to expand because healthcare facilities in the region improve and health expenditures increase.
What Has the Impact of Artificial Intelligence Been on the North America Pediatric Drugs Market?
AI technology rapidly changes the North American pediatric drugs market because it improves research capabilities and data-based decision-making processes. North America pediatric drugs market development through AI technology enables market researchers to use machine learning algorithms which process extensive clinical and demographic data to discover practical market solutions.
The use of predictive analytics helps stakeholders to forecast demand through trend identification which allows them to track changes in pediatric healthcare requirements. Artificial intelligence technology enhances strategic planning processes for organizations operating in the North American pediatric drugs market because it helps them to better understand current market developments.
AI-driven smart automation technology now provides production process improvements and supply chain operational enhancements throughout the North American pediatric drugs market. Machine learning models for manufacturing efficiency and inventory management enable cost reductions which maintain product quality and ensure timely drug distribution.
The North American pediatric drugs market employs artificial intelligence which enables doctors to create tailored treatment plans for specific medical conditions in children. The North American pediatric drugs market uses artificial intelligence to develop innovative products which help companies achieve business growth through competitive advantages in the data-driven healthcare sector.
Key Market Trends & Insights:
- The United States controls most of the North American pediatric drug market which will reach an 80% market share by 2025 because the country has developed superior healthcare systems.
- Canada represents the fastest-growing region, projected to expand at over 7% CAGR during the 2025–2030 forecast period.
- Vaccines segment holds a significant share of over 35% in the north america pediatric drugs market, driven by immunization programs and government support.
- Anti-infective drugs rank as the second-largest segment, supported by high demand for treating common pediatric infections.
- The biologics field represents the expansion segment which will increase at an 8% annual growth rate until 2030 because of its targeted therapies.
- The North American pediatric drug market shows 40% of its applications dedicated to infectious diseases because of their high occurrence rates.
- The demand for chronic disease treatments which include asthma and diabetes care shows the fastest growth rate because more pediatric cases emerge.
- The North American pediatric drugs market sees hospitals operating with more than 55% market share because they maintain special infrastructure for treating pediatric patients.
- Homecare settings represent the fastest-growing market segment because they provide convenient solutions for growing outpatient treatment needs.
North America Pediatric Drugs Market Segmentation
By Type
Vaccines serve an essential function in pediatric medicine because they stop infectious diseases through their implementation in standard immunization programs. The medical field employs anti-infective medications to treat both bacterial and viral infections which continue to affect children. CNS drugs treat neurological disorders including epilepsy and attention deficits while respiratory medications assist with asthma treatment and related conditions. The medical field uses oncology medications to treat childhood cancer while other medication categories provide both general and supportive treatment options.
Different treatment types show doctors how to treat their young patients while they consider safety needs. Medical professionals need vaccines and anti-infective medications because these two types of medical supplies protect against diseases and provide instant medical assistance. Research teams need to study CNS and oncology medications because their usage requires precise measurement and constant observation. The medical field sees increasing development of respiratory solutions because more children develop breathing problems.
To learn more about this report, Download Free Sample Report
By Route
The most common medication administration method for children medical cases is oral administration because it provides simple use and higher acceptance rates. Vaccines and emergency medications require injectable drugs to produce quick effects and maintain precise dosage control. The use of topical medications enables the treatment of skin disorders and specific areas while causing minimal body effects. Patients with asthma manage their condition through inhalation therapies which serve as their primary breathing treatment method.
Transdermal delivery provides controlled release through patches, which helps improve compliance in long-term treatments. The selection of a treatment method depends on three factors: the drug type and the patient's comfort level and the treatment urgency. Pediatric formulations are designed to improve taste, reduce discomfort, and ensure safety. The ongoing development of delivery methods will lead to improved treatment results.
By Application
The primary application field for infections exists because young children experience frequent contact with others while their immune systems remain undeveloped. Neurology studies both seizure disorders and developmental disabilities which need ongoing medical treatment and special medication. The field of respiratory research develops treatments for asthma and various other respiratory disorders which are becoming more common among children. The field of oncology studies childhood cancers which need both early discovery and immediate medical intervention for successful treatment.
The medical field is becoming more knowledgeable about rare diseases because of better diagnostic methods that have emerged. Each application area requires customized drug development processes which must follow established safety regulations. The medical field is moving toward a new treatment method which focuses on delivering healthcare services at an earlier stage. The need for targeted therapies will continue to grow across different application areas.
By End-User
The primary end-user of hospitals exists because specialized pediatric care and advanced treatment facilities are available in these healthcare institutions. Clinics provide routine care and follow-up treatments which make their services available to families. Pharmacies distribute drugs while they provide guidance to patients about how to use their medications. The demand for homecare services expands because patients require both comfortable treatment solutions and continuous therapy supervision.
Research institutions drive innovation by conducting clinical trials while they create safer pediatric drug formulations. Each end-user contributes to the overall healthcare system by ensuring access and proper use of medications. Direct patient care services make hospitals and clinics the primary providers of healthcare. Homecare and research segments will continue to grow with advancements in treatment methods and technology.
What are the Main Challenges for the North America Pediatric Drugs Market Growth?
The north america pediatric drugs market encounters multiple technical problems and operational difficulties which impede its ability to create stable drug formulations that suit different age groups. The process of creating suitable dosing solutions which also taste good and remain usable for extended periods proves to be particularly difficult when dealing with infants and young children. The need for cold-chain vaccine storage together with supply chain interruptions creates additional challenges for operational functions. The north america pediatric drugs market experiences these two main challenges which restrict market growth and create delays in product distribution.
The strict regulatory requirements, including GMP standards, create production challenges that lead to increased costs for manufacturing and commercial processes. The approval process for clinical trials involving pediatric populations requires additional safety measures which extend the duration for obtaining approval. The industry faces additional barriers because high research and development costs together with minimal financial rewards for specific drug types restrict market access. The north american pediatric drugs market experiences product launch delays because companies need additional time to develop innovative solutions.
The north america pediatric drugs market faces adoption challenges because different regions lack proper healthcare infrastructure and access to specialized pediatric services. The availability of pediatric treatments is limited because there are not enough trained professionals and people do not know about these specialized medical options. The lack of financial resources for research and development activities creates obstacles that prevent new ideas from developing. The healthcare systems experience reduced drug adoption rates because these growth barriers prevent new drugs from entering their systems.
Country Insights
The United States maintains its worldwide leadership position because of its effective healthcare systems and its advanced medical research facilities and its substantial financial investment in pediatric medical services. The market maintains steady demand because pharmaceutical companies introduce their latest treatments and maintain their strong market presence. The market for essential medical treatments will experience growth because of regulatory authorities who permit researchers to conduct studies with pediatric patients.
Canada shows stable progress because its citizens access public healthcare services while child-specific medical treatments become more widely recognized. The public dental health system and preventive medicine programs which receive government funding create ongoing demand for pediatric medications. The region will experience growth because research funding increases and people gain better access to advanced medical treatments.
Mexico is developing its healthcare system through better medical facilities and more resources dedicated to treating children. The healthcare system will expand because people become more aware of child health issues. The region will experience economic growth because essential medicines will become more affordable and accessible which supports current efforts to expand essential medicine availability.
Recent Development News
In April 2026, U.S. Government Pushes Drugmakers on Pricing Reforms: U.S. regulators expanded efforts to reduce drug prices through Medicaid negotiations, directly impacting pharmaceutical companies’ revenue strategies. This move is expected to influence pricing models for pediatric drugs and improve accessibility across healthcare systems.
Source: https://www.reuters.com
In January 2026, the U.S. FDA approves the first treatment for a rare paediatric genetic disorder: A major breakthrough in pediatric care was achieved with a new drug approval targeting a rare childhood disease. This development highlights innovation and increased focus on rare pediatric conditions in the United States.
Source: https://www.reuters.com
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 114.6 Billion |
|
Market size value in 2026 |
USD 123.8 Billion |
|
Revenue forecast in 2033 |
USD 201.5 Billion |
|
Growth rate |
CAGR of 7.21% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
North America (Canada, The United States, and Mexico) |
|
Key company profiled |
Pfizer, Johnson & Johnson, Novartis, GSK, Sanofi, Roche, Merck, AbbVie, AstraZeneca, Bristol Myers Squibb, Eli Lilly, Takeda, Bayer, Teva, Amgen |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Vaccines, Anti-infectives, CNS Drugs, Others, Respiratory, Oncology), By Route (Oral, Injectable, Topical, Others, Inhalation, Transdermal), By Application (Infections, Neurology, Respiratory, Others, Oncology, Rare Diseases), By End-User (Hospitals, Clinics, Pharmacies, Others, Homecare, Research). |
How Can New Companies Establish a Strong Foothold in the North America Pediatric Drugs Market?
New companies can establish a strong foothold in the north america pediatric drugs market by targeting niche therapeutic areas such as rare diseases, neonatal care, and pediatric oncology. The emerging players who focus on meeting unmet clinical needs can develop their unique market positioning which matches current industry trends. The research will use data-driven market research and predictive analytics to discover segments with high growth potential and areas with demand shortages. The company uses a targeted approach to enhance its market position which competes with other companies in north america pediatric drugs market.
Innovation strategies play a critical role in establishing child-friendly formulations which include flavored liquids and dissolvable tablets and low-dose biologics. The use of artificial intelligence and smart automation in drug discovery processes will speed up research development and cut expenses. Organizations that spend money on creating unique technology solutions and precision medicine will achieve better results in handling safety and efficacy issues. The advancements will enable sustainable market expansion throughout the north america pediatric drugs market.
The strategic partnerships between new market entrants and hospitals research institutes and established pharmaceutical companies will enable new companies to handle regulatory requirements while they build their distribution channels. The partnerships between organizations provide better ways to access clinical trial facilities and pediatric patient information. The startup companies Pediatric Bioscience and Sprout Pharmaceuticals demonstrate how startups can achieve initial success through dedicated innovation work and partnership development. The business models which these companies use enable them to expand their operations throughout the north america pediatric drugs market.
Key North America Pediatric Drugs Market Company Insights
Leading companies maintain their market position through ongoing product development and efforts to expand their pediatric drug portfolios. The healthcare system experiences constant demand because organizations concentrate their efforts on developing vaccines and anti-infective medicines and their specialized therapeutic products. Companies that invest heavily in research and adopt advanced technologies at an early stage will achieve sustainable growth while their treatment outcomes improve under competitive conditions.
Organizations will enhance their market standing and innovation accessibility through strategic alliances with research entities and healthcare organizations. Mergers and acquisitions enable companies to grow their business operations by entering new markets and developing capabilities in vital business areas. The use of competitive pricing methods together with better supply chain operations will enable businesses to achieve operational stability while they adapt to shifts in customer demand.
Company List
- Pfizer
- Johnson & Johnson
- Novartis
- GSK
- Sanofi
- Roche
- Merck
- AbbVie
- AstraZeneca
- Bristol Myers Squibb
- Eli Lilly
- Takeda
- Bayer
- Teva
- Amgen
What are the Key Use-Cases Driving the Growth of the North America Pediatric Drugs Market?
The north america pediatric drugs market shows growth because of increasing adoption of targeted therapies which treat chronic and infectious diseases in children. The requirement for age-specific formulations exists because asthma and diabetes and bacterial infections need specialized medications which have better safety and effectiveness. The north america pediatric drugs market experiences growth because hospitals and pediatric care centers expand their treatment methods which medical facilities implement.
The north america pediatric drugs market uses immunization and preventive care as another primary application. The government-backed vaccination programs for influenza outbreaks and measles and HPV diseases maintain steady demand because of public health initiatives and citizen awareness. Pharmaceutical companies are developing combination vaccines with better delivery systems which will improve efficiency and compliance while sustaining market growth.
Personalized medicine development creates new market opportunities for the north america pediatric drugs market especially in oncology and rare genetic disorder treatments. Advanced biologics and precision therapies enable development of custom treatment strategies which use genetic data to achieve better treatment results with fewer adverse effects. The shift toward individualized care practices results in substantial innovation possibilities with extended usage in medical settings.
The north america pediatric drugs market shows new possibilities which homecare services and digital health technologies create. The combination of remote monitoring and telehealth support with user-friendly medication solutions boosts treatment compliance and patient access. These market trends provide patients with easier access to treatment while they create healthcare solutions which can grow and spread throughout the industry.
North America Pediatric Drugs Market Report Segmentation
By Type
- Vaccines
- Anti-infectives
- CNS Drugs
- Others
- Respiratory
- Oncology
By Route
- Oral
- Injectable
- Topical
- Others
- Inhalation
- Transdermal
By Application
- Infections
- Neurology
- Respiratory
- Others
- Oncology
- Rare Diseases
By End-User
- Hospitals
- Clinics
- Pharmacies
- Others
- Homecare
- Research
Frequently Asked Questions
Find quick answers to common questions.
The approximate North America Pediatric Drugs Market size for the market will be USD 201.5 Billion in 2033.
The key segments of the North America Pediatric Drugs Market are By Type (Vaccines, Anti-infectives, CNS Drugs, Others, Respiratory, Oncology), By Route (Oral, Injectable, Topical, Others, Inhalation, Transdermal), By Application (Infections, Neurology, Respiratory, Others, Oncology, Rare Diseases), By End-User (Hospitals, Clinics, Pharmacies, Others, Homecare, Research
Major players in the North America Pediatric Drugs Market are Pfizer, Johnson & Johnson, Novartis, GSK, Sanofi, Roche, Merck, AbbVie, AstraZeneca, Bristol Myers Squibb, Eli Lilly, Takeda, Bayer, Teva, Amgen.
The current market size of the North America Pediatric Drugs Market is USD 114.6 Billion in 2025.
The North America Pediatric Drugs Market CAGR is 7.21%.
- Pfizer
- Johnson & Johnson
- Novartis
- GSK
- Sanofi
- Roche
- Merck
- AbbVie
- AstraZeneca
- Bristol Myers Squibb
- Eli Lilly
- Takeda
- Bayer
- Teva
- Amgen
Recently Published Reports
-
Apr 2026
AI in Diabetic Retinopathy Market
AI in Diabetic Retinopathy Market Size, Share & Analysis Report By Type (Screening AI Systems, Diagnostic AI Systems, and Predictive AI Models), By Application Segment (Hospitals, Ophthalmology Clinics, Diagnostic Centers, and Research and Development), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Psychometric Tests Market
Psychometric Tests Market Size, Share & Analysis Report By Type (Personality Tests, Ability/Aptitude Tests, Skill/Knowledge Tests, and Others), By Application (Talent Acquisition and Talent Management), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Rare Biological Sample Collection Market
Rare Biological Sample Collection Market Size, Share & Analysis Report By Type (Isolation Kits & Reagent, Blood Collection Tubes, and Others), By Application (Oncology, Transcriptomics, Pharmacogenomics, and Others), and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 – 2031
-
Apr 2026
Patient Lift Pendant Market
Patient Lift Pendant Market By Product Type (Wired Pendants, Wireless, Programmable), By Operation Type(Manual, Electric), By End-Users (Healthcare Professionals, Patients), By Application (Hospitals, Homecare, Rehabilitation Centers, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
Our Clients
.jpg)