France Gene Therapy Market Size & Forecast:
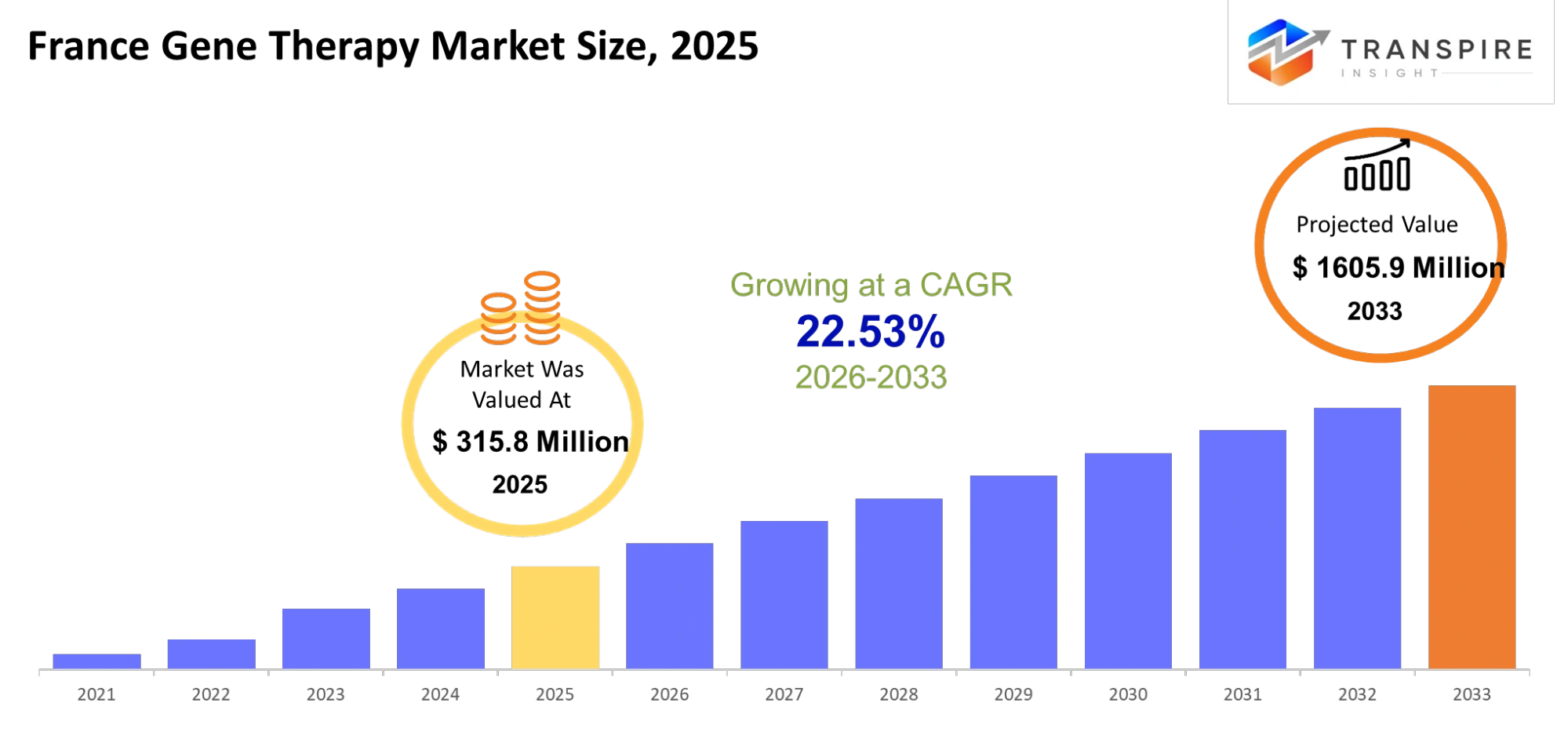
- France Gene Therapy Market Size 2025: USD 315.8 Million
- France Gene Therapy Market Size 2033: USD 1605.9 Million
- France Gene Therapy Market CAGR: 22.53%
- France Gene Therapy Market Segments: By Type (In Vivo Therapy, Ex Vivo Therapy, Viral Vectors, Non-viral Vectors, Others); By Application (Cancer, Rare Diseases, Genetic Disorders, Others); By End-User (Hospitals, Research Institutes, Others); By Delivery (AAV, Lentivirus, Retrovirus, Others)
To learn more about this report, Download Free Sample Report
France Gene Therapy Market Summary:
The France Gene Therapy Market size is estimated at USD 315.8 Million in 2025 and is anticipated to reach USD 1605.9 Million by 2033, growing at a CAGR of 22.53% from 2026 to 2033.
The France gene therapy market will develop through advanced biotechnology together with precision medicine because its healthcare system supports innovation while enabling patient access. The past few years will show a shift in demand toward treatments which precisely target medical conditions because patients and doctors prefer permanent solutions over temporary relief of symptoms. The French regulatory system will keep changing its approval processes, which will enable faster access to innovative therapies while ensuring patient safety. The development of vector design and manufacturing technologies will improve both treatment delivery methods and the dependability of results. Public understanding of genetic therapy together with ethical discussions will determine how people accept these treatments, which will require open communication between parties and trustworthy systems to establish which treatments will receive financial support and become standard medical practice.
What Has the Impact of Artificial Intelligence Been on the France Gene Therapy Market?
The France gene therapy market experiences a rapid transformation through artificial intelligence, which enables researchers to use data analytics for their therapy development work. The France gene therapy market uses AI to enhance market research capabilities by examining clinical data, patient registry data and genomic data at a speed and accuracy which exceeds traditional research methods.
The france gene therapy market benefits from smart automation and machine learning technologies which boost operational efficiency beyond their analytical functions. Advanced algorithms streamline production workflows which reduce errors during vector development and enable scalable manufacturing that will become essential for personalized therapies. AI-driven supply chain optimization further reduces costs by improving inventory planning and minimizing waste and ensuring timely delivery of sensitive biologics. The France gene therapy market will experience enhanced innovation processes which will establish unique competitive advantages for businesses that incorporate artificial intelligence into their fundamental business operations.
Key Market Trends & Insights:
- The France gene therapy market in 2025 sees Northern France as its leading region which controls 38% of market share because of its healthcare facilities and research centers.
- The Southern France region establishes itself as the fastest expanding area which will experience more than 20% CAGR growth until 2030 because of increasing clinical trial operations.
- The market for viral vector-based therapies leads with a 55% market share because these treatments show strong effectiveness against rare genetic disorders and oncology diseases.
- Non-viral gene delivery systems hold the second-largest share because these systems provide better safety through reduced immunogenicity hazards.
- The CRISPR-based therapy market identifies itself as the fastest expanding segment which expects to experience strong growth from 2026 until 2032 because its precision-editing features.
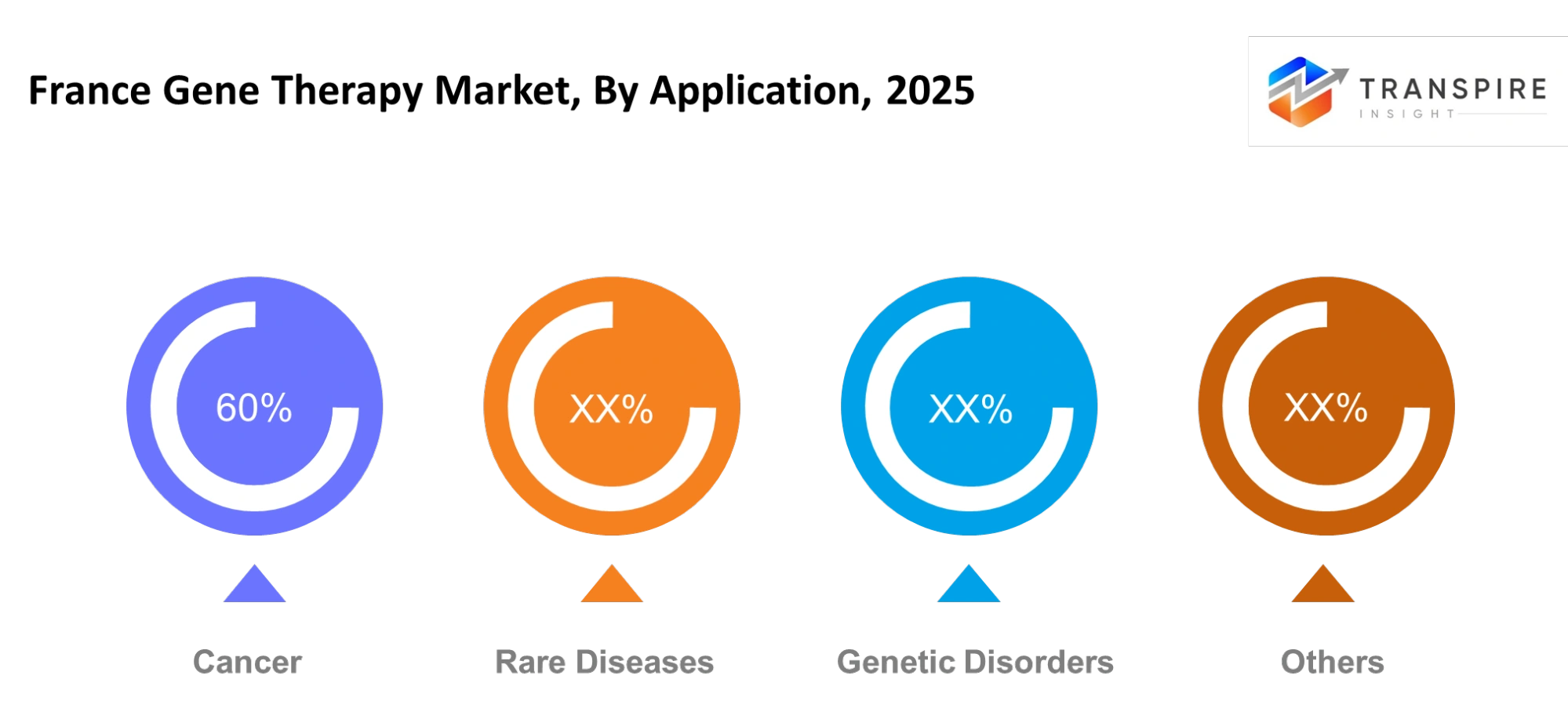
- The France gene therapy market shows more than 45% market share for oncology applications because there exists a strong need for targeted cancer solutions.
- The application segment for rare genetic disorders shows the fastest growth because of increasing orphan drug approvals and development of patient-specific therapies.
- The market shows leading hospitals and specialty clinics which hold almost 50% market share because they have access to advanced treatment facilities and qualified medical staff.
- Research institutes display rapid expansion because funding increases and gene-based therapy collaboration projects continue to rise.
- The company achieves competitive advantages through European market expansion which supports its production capabilities while securing future business development.
France Gene Therapy Market Segmentation
By Type:
The study divides its segments into five categories which include In Vivo Therapy and Ex Vivo Therapy and Viral Vectors and Non-viral Vectors and other methods. In vivo therapy will deliver genetic material directly into the body while ex vivo therapy will change cells outside the body before they receive reinfusion treatment. The field will continue to use viral vectors because they provide efficient results while non-viral vectors will attract increasing interest because they offer safer options. The development of new solutions will continue through technological advancements.
By Application:
The growing number of cancer cases, combined with the need for specific treatment methods, will make cancer treatment the main focus of medical work. The pharmaceutical industry will experience rapid development in rare diseases because new treatments will fulfill existing treatment gaps. Early diagnosis together with precision tools will create advantages for genetic disorders. The study will identify new therapeutic applications through research which will gradually develop additional applications.
To learn more about this report, Download Free Sample Report
By End-User:
The study needs to determine which end-users will use its results, which include hospitals and research institutes and various other organizations. Hospitals will lead adoption because their personnel possesses training and hospitals maintain infrastructure which enables them to deliver complex therapies. Research institutes will drive forward both technological development and clinical research activities. Specialty centers and other end-user organizations will help patients by making treatment more accessible and they will extend their medical services throughout various regions to multiple patient groups.
By Delivery:
AAV delivery will become the preferred option because it offers both safety and precise gene transfer capabilities. Lentivirus will enable permanent gene expression during extended therapeutic periods. The use of retrovirus remains applicable in particular medical treatments although it has certain restrictions. The research will examine better delivery techniques which will increase treatment accuracy and decrease treatment dangers while boosting complete treatment success.
What are the Main Challenges for the France Gene Therapy Market Growth?
The French gene therapy market faces two main obstacles which stem from both technical issues and operational difficulties as they try to achieve stable product results between different patient groups. The treatment process requires two complex steps which involve delivering genes and controlling immune responses which makes it difficult to expand operations. The deployment process will experience delays because of supply chain restrictions that affect essential raw materials and cold storage system requirements, which will result in market constraints.
The France gene therapy market faces major obstacles to its manufacturing and commercialization processes because production needs to follow strict Good Manufacturing Practice (GMP) requirements which require highly controlled production conditions. The combined expenses for developing vectors, the restricted ability to produce them, and the protracted product approval process will push back the launch dates. These growth barriers will restrict smaller companies from entering the market and slow broader commercialization efforts.
The France gene therapy market faces adoption challenges which stem from two main factors: the lack of specialized infrastructure and the limited availability of trained professionals. The healthcare system will struggle to incorporate advanced therapies into standard care procedures while funding deficiencies together with reimbursement uncertainty will make treatments less available. The market limitations will decrease patient access and cause overall demand to increase at a slower rate.
The France gene therapy market faces expanding difficulties because of two main factors: competitive pressure from other treatment methods and the development of new technologies. The combination of pricing issues and ethical dilemmas together with shifting regulatory requirements will lead to business uncertainty which forces companies to modify their growth strategies in order to achieve sustainable development.
Regional Insights
The France gene therapy market will continue to develop, supported by national healthcare policies and the rising focus on advanced treatment methods. Research funding together with clinical trial expansion will boost innovative development capabilities. The market will advance through three essential factors which involve determining treatment expenses, ensuring patient accessibility, and establishing clear rules for therapy administration to various patient categories.
Northern regions will lead due to strong research infrastructure, established biotechnology hubs, and access to skilled professionals. Central regions will show stable development supported by hospital networks and clinical adoption. Southern regions will experience gradual growth driven by increasing investments, improved healthcare facilities, and expanding participation in clinical research programs.
Recent Development News
In April 2026, Roche launched a late-stage clinical study for its gene therapy Elevidys following feedback from the European regulator, aiming to address prior safety concerns and support future approval in Europe. https://www.reuters.com
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 315.8 Million |
|
Market size value in 2026 |
USD 387.3 Million |
|
Revenue forecast in 2033 |
USD 1605.9 Million |
|
Growth rate |
CAGR of 22.53% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
France |
|
Key company profiled |
Novartis, Gilead, Roche, Pfizer, Bluebird Bio, Spark Therapeutics, Biogen, Sanofi, Bristol Myers Squibb, AstraZeneca, Orchard Therapeutics, CRISPR Therapeutics, Editas Medicine, Intellia Therapeutics, Sarepta |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (In Vivo Therapy, Ex Vivo Therapy, Viral Vectors, Non-viral Vectors, Others); By Application (Cancer, Rare Diseases, Genetic Disorders, Others); By End-User (Hospitals, Research Institutes, Others); By Delivery (AAV, Lentivirus, Retrovirus, Others) |
How Can New Companies Establish a Strong Foothold in the France Gene Therapy Market?
New entrants aiming to establish market leadership in the france gene therapy industry must develop their unique value propositions through specific product development initiatives. Companies should concentrate their efforts on specialized markets which include rare genetic disorders and unmet oncology requirements because these areas present higher demand and lower competitive threats. Organizations achieve better development results through their implementation of data-backed research methods which study regulatory requirements from the beginning of their projects.
The france gene therapy market requires partnerships as essential elements to help companies enter the market. The collaboration with academic institutions together with contract manufacturing organizations and established pharmaceutical companies will enable access to vital resources which include both expertise and financial support. GenSight Biologics and Cellectis demonstrate how companies can establish a competitive edge in the market through innovative strategies which center on their proprietary platform and specialized treatment development.
The france gene therapy market will measure success through technology differentiation which includes advanced vector design and scalable manufacturing and AI-driven development processes. Companies which develop solutions for critical production challenges which include high production expenses and limited production capabilities and supply chain problems will achieve market leadership. New companies will use their innovative capabilities together with strategic partnerships and operational effectiveness to acquire market share while building their long-lasting development capacity.
Key France Gene Therapy Market Company Insights
The France gene therapy market will experience continuous development because companies are dedicated to creating new treatments which provide specific therapies with better patient results. The market will develop through research support and clinical advancements while pricing methods and regulatory frameworks will determine how products will be used. The market participants will try to achieve two goals which require them to provide less expensive treatment options while maintaining treatment standards.
The leading companies will enhance their market positions through product development and strategic partnerships and the expansion of their manufacturing facilities. Established players will invest in advanced technologies, while smaller firms will focus on niche innovation. The competitive market will see greater competition from new companies who bring unique products, which they will promote through research and pricing and partner collaborative methods.
Company List
- Novartis
- Gilead
- Roche
- Pfizer
- Bluebird Bio
- Spark Therapeutics
- Biogen
- Sanofi
- Bristol Myers Squibb
- AstraZeneca
- Orchard Therapeutics
- CRISPR Therapeutics
- Editas Medicine
- Intellia Therapeutics
- Sarepta
What are the Key Use-Cases Driving the Growth of the France Gene Therapy Market?
The France gene therapy market expansion proceeds through key use cases which identify oncology as its main driving force. Researchers utilize advanced gene therapies to develop treatments that target specific cancer mutations which deliver better patient outcomes through reduced side effects and improved survival rates when compared to standard medical treatment. The need for hospitals and research facilities to adopt new technologies creates a direct link between growing clinical evidence and market expansion while establishing permanent customer needs.
The France gene therapy market development receives a significant boost through the treatment of rare diseases. Gene therapies provide patients with either one-time treatment or limited-dose therapy for conditions that used to need permanent medical attention. The new treatment methods enable providers to achieve better patient results while decreasing total healthcare expenditures thus making their services more appealing to both health systems and payers.
The France gene therapy market expansion receives support from genetic disorder treatment programs. The combination of early disease detection with specific gene correction methods leads to better treatment outcomes. Researchers are investigating treatment applications for inherited blood disorders and neurological conditions through their current clinical studies and the development of new delivery methods.
The France gene therapy market benefits from manufacturing process innovations which bring two advantages to the biotechnology industry through improved production capacity and operational effectiveness. The combination of automation with AI-based manufacturing processes delivers two advantages to companies through better product quality and lower operational expenses. The market will expand through these real-world applications which will boost both adoption rates and innovation activities while also creating new business opportunities.
France Gene Therapy Market Report Segmentation
By Type
- In Vivo Therapy
- Ex Vivo Therapy
- Viral Vectors
- Non-viral Vectors
- Others
By Application
- Cancer
- Rare Diseases
- Genetic Disorders
- Others
By End-User
- Hospitals
- Research Institutes
- Others
By Delivery
- AAV
- Lentivirus
- Retrovirus
- Others
Frequently Asked Questions
Find quick answers to common questions.
The approximate France Gene Therapy Market size for the market will be USD 1605.9 Million in 2033.
Key segments for the France Gene Therapy Market are By Type (In Vivo Therapy, Ex Vivo Therapy, Viral Vectors, Non-viral Vectors, Others); By Application (Cancer, Rare Diseases, Genetic Disorders, Others); By End-User (Hospitals, Research Institutes, Others); By Delivery (AAV, Lentivirus, Retrovirus, Others).
Major France Gene Therapy Market players are Novartis, Gilead, Roche, Pfizer, Bluebird Bio, Spark Therapeutics, Biogen, Sanofi, Bristol Myers Squibb, AstraZeneca, Orchard Therapeutics, CRISPR Therapeutics, Editas Medicine, Intellia Therapeutics, Sarepta.
The France Gene Therapy Market size is USD 315.8 Million in 2025.
The France Gene Therapy Market CAGR is 22.53%.
- Novartis
- Gilead
- Roche
- Pfizer
- Bluebird Bio
- Spark Therapeutics
- Biogen
- Sanofi
- Bristol Myers Squibb
- AstraZeneca
- Orchard Therapeutics
- CRISPR Therapeutics
- Editas Medicine
- Intellia Therapeutics
- Sarepta
Recently Published Reports
-
Apr 2026
Cord Blood Banking Services Market
Cord Blood Banking Services Market Size, Share & Analysis Report By Type of Service (Collection, Processing, Storage), By Bank Type (Private Banks, Public Banks, and Hybrid Banks), By Component (Cord Blood, and Cord Tissue), By End User (Hospitals, Research Institutions, Biotechnology Companies, and Others) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
ELISA Processors Market
ELISA Processors Market Size, Share & Analysis Report By Type (Optical Filter ELISA Analyzer, and Optical Grating ELISA Analyzers), By Mode (Automated ELISA Analyzers, and Semi-Automated ELISA Analyzers), By Applications (Immunology, Vaccine development, Drug Monitoring, and Others), By End Users (Biotechnology Companies, Pharmaceutical Companies, Contract Research Organization, and Diagnostic Laboratories) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Microscope Slide Scanner Market
Microscope Slide Scanner Market Size, Share & Analysis Report By Type (Portable Microscope Slide Scanner, and Desktop Microscope Slide Scanner), By Application (Biological Research, Medicine, Veterinary, and Other) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031
-
Apr 2026
Microwave Cookware Market
Microwave Cookware Market Size, Share & Analysis Report By Material Type (Plastic, Glass, Ceramic, Silicone, and Metal), By Application (Cooking, Reheating, and Defrosting), By End User (Household, Commercial (Restaurants, Hotels, and Cafes), and Food Service Industry), By Sales Channel (Online Retail, Offline Retail (Supermarkets, Hypermarkets, Specialty Stores), and Direct Sales (Company Outlets)) and Geography (North America, Europe, Asia-Pacific, Middle East and Africa, South and Central America), 2021 - 2031