Europe Cancer Therapeutics and Biotherapeutics Market Size & Forecast:
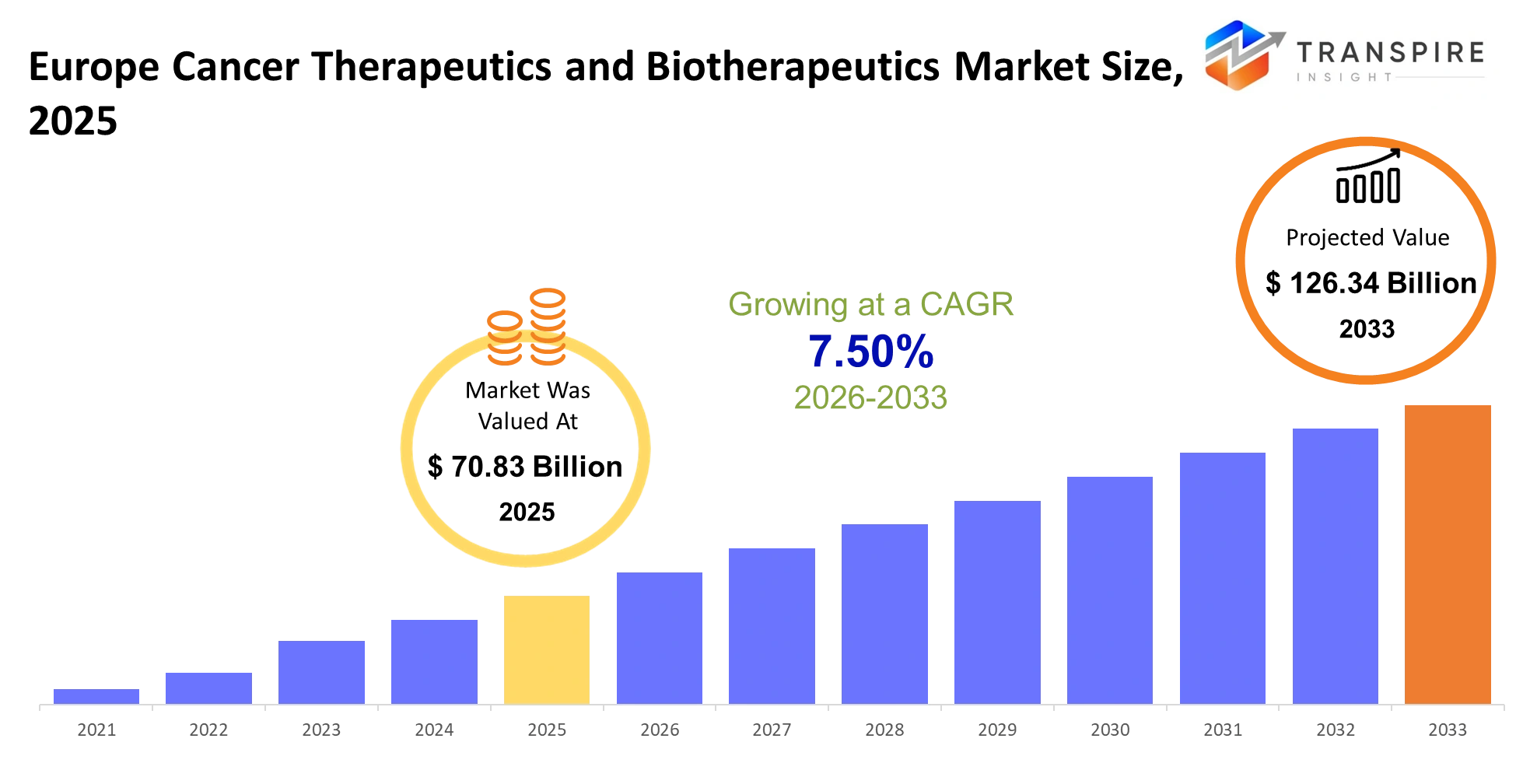
- Europe Cancer Therapeutics and Biotherapeutics Market Size 2025: USD 70.83 Billion
- Europe Cancer Therapeutics and Biotherapeutics Market Size 2033: USD 126.34 Billion
- Europe Cancer Therapeutics and Biotherapeutics Market CAGR: 7.50%
- Europe Cancer Therapeutics and Biotherapeutics Market Segments: By Type (Chemotherapy, Immunotherapy, Targeted Therapy, Hormonal Therapy, Others), By Application (Breast Cancer, Lung Cancer, Colorectal Cancer, Blood Cancer, Others), By End-User (Hospitals, Cancer Research Centers, Clinics, Others), By Drug Type (Biologics, Small Molecules, Others).
To learn more about this report, Download Free Sample Report
Europe Cancer Therapeutics and Biotherapeutics Market Summary:
The Europe Cancer Therapeutics and Biotherapeutics Market size is estimated at USD 70.83 Billion in 2025 and is anticipated to reach USD 126.34 Billion by 2033, growing at a CAGR of 7.50% from 2026 to 2033. The Europe Cancer Therapeutics and Biotherapeutics Market delivers treatments that directly intervene in tumor growth. The treatments enable doctors to treat patients. The hospital oncology units require his solution because it provides targeted drugs and monoclonal antibodies and cell-based therapies which researchers now develop for specific genetic profiles.
The market has transformed during the last three to five years because precision oncology has become the standard treatment method which matches specific therapies to patient biomarkers instead of using general cancer treatments. The transition has gained momentum because regulators now approve advanced therapies and companion diagnostics through expedited approval processes. The healthcare system became overloaded with post-pandemic patients which forced systems to adopt more efficient treatments that produced better results, leading to increased usage of high-efficacy biologics.
The company experiences growth because its longer treatment duration therapies create higher treatment costs per patient, which results in increased revenue from its specialized products that drive business innovations.
Key Market Insights
- Western Europe leads the European Cancer Therapeutics and Biotherapeutics Market because it holds approximately 65% of market share in 2025 which Germany France and the UK control.
- The healthcare system in Eastern Europe will experience its most rapid development until 2030 because of new healthcare facilities and better access to cancer treatment.
- Monoclonal antibodies and immunotherapies which demonstrate superior clinical success rates drive the biotherapeutics market which will reach over 55% market share in 2025.
- The chemotherapy market segment ranks as the second biggest because it provides affordable treatment options that doctors continue to use in combination with other therapies.
- The cell and gene therapy market represents its fastest growth period because new treatment solutions will drive market expansion at double-digit growth rates until 2030.
- The market for lung and breast cancer applications reached 40% market share in 2025 because of the high cancer incidence rates and the use of targeted therapies.
- The development of CAR-T therapies and the creation of precision biologics have made hematological cancers the most rapidly expanding application segment.
- Hospitals make up the largest share of end-user demand, which will reach 60% in 2025 because they have advanced oncology systems and their teams work together to deliver healthcare.
- Specialty cancer centers experience their most rapid growth because they combine personalized treatment approaches with clinical trial research.
What are the Key Drivers, Restraints, and Opportunities in the Europe Cancer Therapeutics and Biotherapeutics Market?
The Europe Cancer Therapeutics and Biotherapeutics Market experiences its strongest growth through precision oncology which functions as its main driving force. The European Medicines Agency and other regulatory agencies have established accelerated approval pathways that enable faster market entry for therapies which depend on biomarkers. The development of genomic sequencing and companion diagnostics technology enables doctors to select treatments based on the specific genetic mutations present in patients. Pharmaceutical companies prefer to develop high-efficiency biologics that come with substantial costs because this approach enables them to boost their income from each patient while achieving better clinical results. Hospitals are adopting these therapies at an increasing pace because they help physicians select relevant treatments more effectively, which leads to faster treatment results and helps hospitals use their resources better.
The most important fundamental obstacle for healthcare systems exists in their struggle to cope with pricing pressures and reimbursement restrictions. The total cost of CAR-T treatments and other advanced therapies often exceeds €300,000 which creates financial challenges for public healthcare systems. The situation becomes complicated because Europe depends on centralized pricing negotiations and health technology assessments which cause delays in obtaining approval. Patient access gets postponed because of this situation which leads to different countries adopting the innovation at different rates. The situation results in suppressed market revenue as Southern and Eastern European countries demonstrate weak clinical development capacities.
The development of cell and gene therapy infrastructure creates a significant opportunity for business growth. Germany and other nations are establishing local production facilities and medical treatment centers to decrease their operational challenges and time required for processing. The therapies will shift from their current limited applications to wider use in oncology when production volumes increase and expenses decrease.
What Has the Impact of Artificial Intelligence Been on the Europe Cancer Therapeutics and Biotherapeutics Market?
The oncology treatment system in Europe experiences transformation through the combined power of artificial intelligence and modern digital technologies. AI platforms in drug development use genomic data analysis and real-world evidence assessment to automate target identification and clinical trial design which enables researchers to complete their first research phase 30 percent faster. Roche and Novartis use machine learning systems to enhance their clinical trial recruitment process by connecting patients to trials that match their specific biomarkers which leads to better recruitment results and shorter wait times.
The development of predictive capabilities enables more accurate treatment delivery. AI models predict how patients will react to immunotherapy by combining imaging data with pathology results and genomic information which helps doctors to choose treatments that have the highest chance of success. The process helps to decrease treatment cycles that do not lead to positive results while it also reduces total treatment expenses and enhances patient survival rates. The use of digital twins and AI process control systems in manufacturing operations helps to maintain consistent biotherapeutic production which results in higher batch success rates and decreased product loss.
Hospitals implement AI-driven decision support systems to optimize their oncology operations which results in faster diagnostic results and more precise treatment planning. European healthcare systems face a main obstacle because their health data remains divided into multiple separate systems. The current data sharing restrictions together with existing standardization issues create training difficulties for models which results in restricted system development and delays in achieving complete AI-based oncology transformation.
Key Market Trends
- Biomarker-based treatment methods have taken over from standard chemotherapy treatments since 2021 because more than half of European oncology drugs who were launched after 2021 now target specific genetic mutations.
- The pharmaceutical industry accelerated its product development process because regulatory agencies developed new approval mechanisms which enabled faster market entry of advanced therapies following 2020.
- Between 2020 and 2025 hospitals acquired high-cost biologics because they wanted to achieve better patient outcomes through their purchasing methods which focused on treatment results instead of purchasing drugs based on their quantities.
- After 2022, more cancer patients received immunotherapy because checkpoint inhibitors became standard treatment for various cancer types which replaced older treatment methods.
- AstraZeneca and Bristol Myers Squibb increased their research spending on combination therapies which resulted in better treatment outcomes but made medical procedures more difficult and expensive.
- The Western markets used to control advanced treatment distribution until 2023 when Eastern European countries started building their oncology treatment infrastructure.
- The number of cell and gene therapy programs increased after 2021 because clinical research showed growing interest in developing personalized therapies which require only one treatment session.
- European health insurers established tighter reimbursement requirements after 2022 which included more detailed health technology assessments that resulted in delayed market entry for high-priced treatment options.
- The healthcare system integrated digital health solutions which enabled doctors to make faster treatment decisions while using AI-based diagnostic tools and treatment planning software.
- The manufacturing sector changed its operational methods since 2021 by using localized production methods to protect against supply chain problems while maintaining reliable access to biologics.
Europe Cancer Therapeutics and Biotherapeutics Market Segmentation
By Type
The Type of treatment immunotherapy demonstrates its status as the leading treatment method because it delivers strong clinical results and maintains its effectiveness across different tumor types. The development of precise molecular interventions through genomic profiling advancements brings doctors closer to targeted therapy as their next treatment option. Chemotherapy functions as a fundamental treatment method because it remains effective when combined with other treatments which need to control expenses and treat various patient groups.
Essential hormonal therapy treats all hormone-sensitive cancers which include breast cancer and prostate cancer. Medical professionals now use combination therapies which unite immunotherapy with targeted drugs to achieve superior treatment results which causes the market to grow. The future of medicine will move towards personalized treatment approaches which will require manufacturers to develop biomarker-based pipelines while healthcare providers learn to navigate more advanced medication distribution systems.

To learn more about this report, Download Free Sample Report
By Application
The breast cancer and lung cancer segments lead the market because both diseases show high patient rates and researchers keep developing new targeted and biologic treatments. The blood cancer market develops rapidly because researchers discovered new cell therapies which include CAR-T and monoclonal antibodies. Colorectal cancer maintains a stable market share because screening programs and new treatment methods provide incremental advancements.
The medical field shows different treatment patterns because lung and blood cancers both use precision therapies but colorectal cancer treatment follows established medical procedures. The market will grow through two factors which include improved access to advanced medical treatments and the combination of diagnostic tests with treatment development. This development path offers business opportunities for companies that use disease-specific biomarkers to direct their drug research efforts.
By End-User
The market is controlled by hospitals because they possess sophisticated facilities and multiple cancer treatment teams and their capacity to deliver high-cost biological medications and intravenous treatment services. Cancer research centers function as critical facilities that create new treatment methods while executing clinical research studies. Clinics and smaller care settings are expanding their service capacity although their growth remains restricted by their current infrastructure and their funding limitations.
Outpatient oncology services are expanding because their operational costs have decreased while their treatment process has become more convenient for patients. Healthcare providers will receive additional financing for specialized medical facilities and digital health systems which will enable them to deliver complex treatments outside traditional hospital environments.
By Drug Type
The market for biologic drugs is currently the largest segment because these drugs show greater treatment success and precise disease targeting power. Patients continue to prefer small molecules because they offer easy oral consumption and more affordable price points and their production methods have reached established standard levels. The two segments maintain their current distribution because they provide different levels of patient treatment access and medical results.
Biologics production increases because of new developments in monoclonal antibodies and cell-based therapies while the industry sees ongoing development of small molecules through their next-generation targeted inhibitors. The upcoming period will experience a transition towards hybrid treatment methods which combine biologics with small molecules, compelling manufacturers to enhance their production capacity and develop new pricing models.
What are the Key Use Cases Driving the Europe Cancer Therapeutics and Biotherapeutics Market?
The main reason hospitals adopt targeted therapies for breast and lung cancers which occur frequently in patients. Hospitals give priority to biomarker-based biologics and immunotherapies because these treatments lead to better survival rates while decreasing the need for unnecessary medical procedures. The clinical process efficiency results in continuous demand growth which increases the amount spent on each patient.
New treatment methods are developing for both hematological cancers and combination therapy treatments. Cancer research centers together with specialty oncology clinics have begun using CAR-T therapies and monoclonal antibodies to treat leukemia and lymphoma. The combination of clinical trial research work and modern facility access creates an environment that encourages early implementation of this technology.
The new use cases of personalized neoantigen therapies and AI-guided treatment selection present solutions for advanced cancer treatment. Advanced oncology centers have begun implementing these methods because genomic data will enable them to create individualized cancer treatment plans at least until the forecasted period ends.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 70.83 Billion |
|
Market size value in 2026 |
USD 76.14 Billion |
|
Revenue forecast in 2033 |
USD 126.34 Billion |
|
Growth rate |
CAGR of 7.50% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain and Rest of Europe) |
|
Key company profiled |
Roche, Novartis, Pfizer, Merck, Bristol Myers Squibb, AstraZeneca, Sanofi, GSK, Amgen, Eli Lilly, Johnson & Johnson, AbbVie, Bayer, Takeda, Regeneron. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Chemotherapy, Immunotherapy, Targeted Therapy, Hormonal Therapy, Others), By Application (Breast Cancer, Lung Cancer, Colorectal Cancer, Blood Cancer, Others), By End-User (Hospitals, Cancer Research Centers, Clinics, Others), By Drug Type (Biologics, Small Molecules, Others). |
Which Regions are Driving the Europe Cancer Therapeutics and Biotherapeutics Market Growth?
European countries maintain their market leadership because they implement precision oncology systems through official government policies. The United Kingdom France and Germany use established reimbursement systems to facilitate quick access to valuable biological products. The network of academic medical centers and research facilities and biotechnology companies creates an environment that supports ongoing discovery and medical research. The scientific community uses this system to transform research breakthroughs into market-ready medical treatments which strengthen the area's business advantage.
Northern European countries maintain their position as a secure secondary market because they maintain steady funding for healthcare services while implementing their regulatory systems in an organized manner. Swedish and Danish markets focus on value-based healthcare systems which create a gradual yet controlled process for new medical technologies to enter their markets. The centralized healthcare systems of Western European countries combined with their long-term financial management approaches create a predictable growth pattern that differs from the more aggressive expansion of Western European markets. The region's economic stability enables it to become a dependable source of ongoing revenue generation.
Recent improvements in healthcare facilities and greater availability of oncology services have made Eastern Europe the fastest expanding area. Since 2022 Poland and Romania have boosted their public spending while creating regulatory systems that match European Union standards. The new regulations permit hospitals to distribute biologics faster which results in patients getting innovative medical treatments more rapidly. The region provides investors with expansion opportunities which will last until 2026-2033 because current adoption rates continue to decrease.
Who are the Key Players in the Europe Cancer Therapeutics and Biotherapeutics Market and How Do They Compete?
The Europe Cancer Therapeutics and Biotherapeutics Market experiences moderate competition because global pharmaceutical companies control most valuable biologics yet smaller biotech companies develop new technologies. The market players maintain their market positions by using their extensive product development capabilities and their knowledge of regulatory processes and their established connections with payers. The industry now competes through clinical effectiveness and treatment precision which organizations use to assess their products by matching outcomes information with biomarker data. Organizations that prove they offer better survival rates and safer treatment options will achieve faster reimbursement processes and gain more trust from doctors.
Roche uses its combined diagnostic and therapeutic solutions to develop treatment methods based on biomarkers which result in better clinical results and enable the company to charge higher prices. Novartis develops its business through specialized manufacturing networks which enable it to create advanced therapy platforms especially CAR-T. AstraZeneca develops combination treatments in oncology which involve targeted medications and immunotherapies to provide extended treatment benefits for various cancer forms.
Sanofi creates competitive advantage through its collaboration with biotech companies which helps it develop biologics faster while it establishes its presence in European markets. Bristol Myers Squibb uses its strong immuno-oncology portfolio to grow through strategic acquisitions which increase its pipeline capacity and clinical trial capabilities to strengthen its competitive advantages in high-growth markets.
Company List
- Roche
- Novartis
- Pfizer
- Merck
- Bristol Myers Squibb
- AstraZeneca
- Sanofi
- GSK
- Amgen
- Eli Lilly
- Johnson & Johnson
- AbbVie
- Bayer
- Takeda
- Regeneron
Recent Development News
In April 2026, AstraZeneca boosts UK oncology-driven investment after pharma pricing deal: AstraZeneca announced a major £300 million UK investment, reinforcing its oncology and biotherapeutics R&D footprint in Europe. The move follows a new US–UK pharmaceutical pricing agreement that supports increased access to innovative medicines. Cancer drugs remain the company’s primary revenue driver, with oncology contributing nearly half of total sales.
Source: https://www.reuters.com
In May 2026, Big pharma accelerates Europe-linked oncology biotech acquisitions amid patent cliff: European oncology-biotech-linked deal activity surged as global pharmaceutical firms accelerated biotech acquisitions in cancer therapeutics, especially in immunotherapy and CAR-T platforms. The wave is driven by upcoming patent expiries and the need to rebuild oncology pipelines across Europe and the US.
Source: https://www.reuters.com
What Strategic Insights Define the Future of the Europe Cancer Therapeutics and Biotherapeutics Market?
The Europe Cancer Therapeutics and Biotherapeutics Market is structurally moving toward highly individualized, data-driven treatment models, which use real-time patient biological data to create personalized treatment solutions that clinicians will implement instead of using standard treatment methods. The combination of genomic sequencing and AI-based diagnostic tools along with next-generation biologics is changing the methods used to assess value in oncology treatment.
The process of manufacturing advanced therapies poses a hidden danger because companies need to rely on specific production centers. The production facilities for cell and gene therapies face capacity limitations because only a few specialized centers exist, which will create delivery delays and treatment access problems that emerge from strongest clinical requirement.
The development of decentralized manufacturing and point-of-care production systems in Central and Eastern Europe constitutes a new business opportunity because these regions experience modernization of their infrastructure. Companies that establish their manufacturing facilities and diagnostic systems at an early stage will achieve operational benefits because they will enhance patient contact while decreasing their expenses and manufacturing time.
Europe Cancer Therapeutics and Biotherapeutics Market Report Segmentation
By Type
- Chemotherapy
- Immunotherapy
- Targeted Therapy
- Hormonal Therapy
- Others
By Application
- Breast Cancer
- Lung Cancer
- Colorectal Cancer
- Blood Cancer
- Others
By End-User
- Hospitals
- Cancer Research Centers
- Clinics
- Others
By Drug Type
- Biologics
- Small Molecules
- Others
Frequently Asked Questions
Find quick answers to common questions.
The approximate Europe Cancer Therapeutics and Biotherapeutics Market size for the market will be USD 126.34 Billion in 2033.
The key segments of the Europe Cancer Therapeutics and Biotherapeutics Market are By Type (Chemotherapy, Immunotherapy, Targeted Therapy, Hormonal Therapy, Others), By Application (Breast Cancer, Lung Cancer, Colorectal Cancer, Blood Cancer, Others), By End-User (Hospitals, Cancer Research Centers, Clinics, Others), By Drug Type (Biologics, Small Molecules, Others).
Major players in the Europe Cancer Therapeutics and Biotherapeutics Market are Roche, Novartis, Pfizer, Merck, Bristol Myers Squibb, AstraZeneca, Sanofi, GSK, Amgen, Eli Lilly, Johnson & Johnson, AbbVie, Bayer, Takeda, Regeneron.
The current market size of the Europe Cancer Therapeutics and Biotherapeutics Market is USD 70.83 Billion in 2025.
The Europe Cancer Therapeutics and Biotherapeutics Market CAGR is 7.50%.
- Roche
- Novartis
- Pfizer
- Merck
- Bristol Myers Squibb
- AstraZeneca
- Sanofi
- GSK
- Amgen
- Eli Lilly
- Johnson & Johnson
- AbbVie
- Bayer
- Takeda
- Regeneron
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


