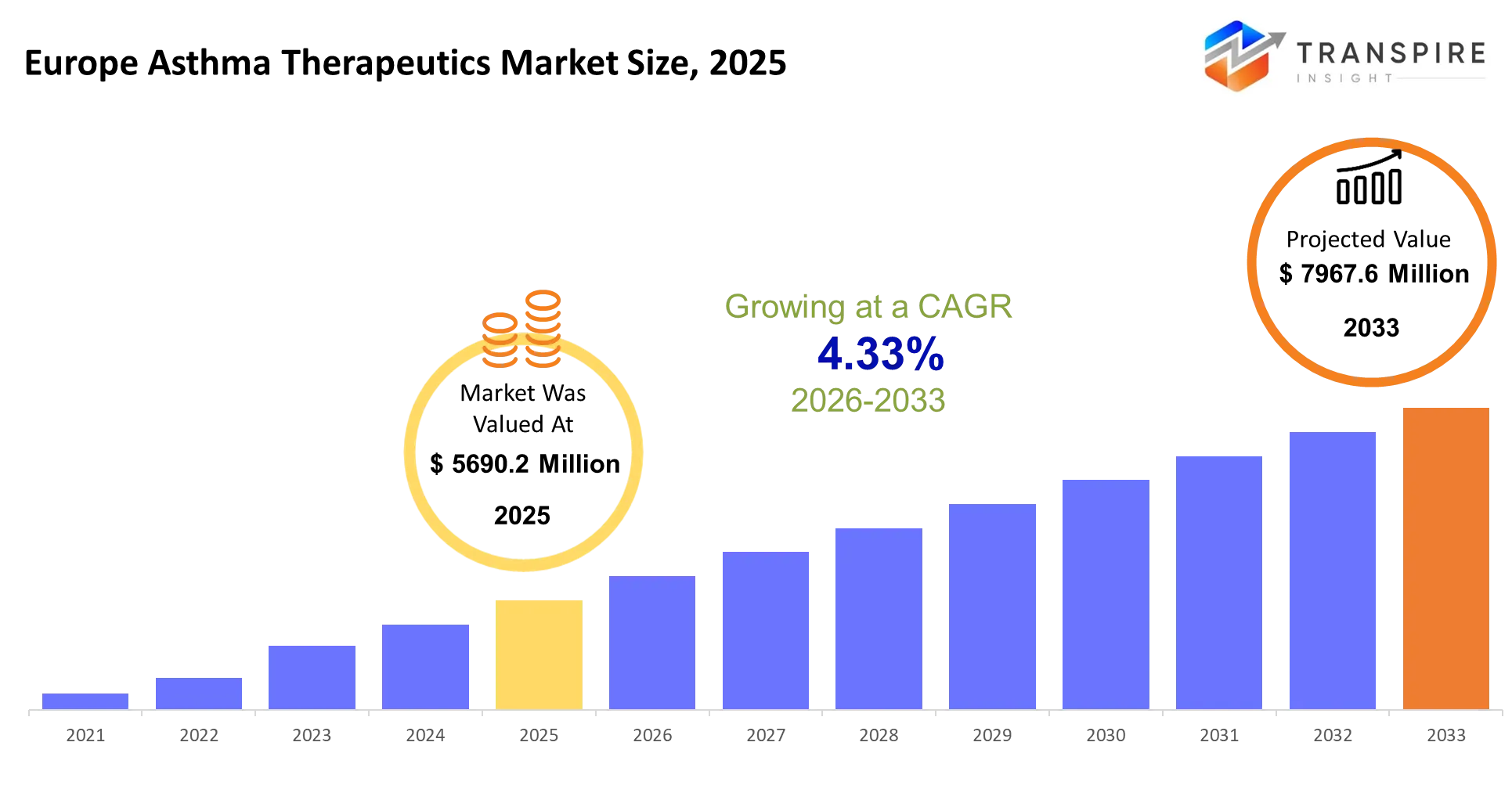
Europe Asthma Therapeutics Market Size & Forecast:
- Europe Asthma Therapeutics Market Size 2025: USD 5690.2 Million
- Europe Asthma Therapeutics Market Size 2033: USD 7967.6 Million
- Europe Asthma Therapeutics Market CAGR: 4.33%
- Europe Asthma Therapeutics Market Segments: By Type (Biologics, Bronchodilators, Corticosteroids, Others), By Application (Severe Asthma, Chronic Asthma, Others), By End-User (Hospitals, Clinics, Homecare, Others), By Distribution (Hospital Pharmacies, Retail Pharmacies, Others).
To learn more about this report, Download Free Sample Report
Europe Asthma Therapeutics Market Summary:
The Europe Asthma Therapeutics Market size is estimated at USD 5690.2 Million in 2025 and is anticipated to reach USD 7967.6 Million by 2033, growing at a CAGR of 4.33% from 2026 to 2033. The Europe Asthma Therapeutics Market functions at the frontline of respiratory care, enabling patients to maintain daily productivity by preventing flare-ups and reducing emergency interventions. The therapies effectively prevent hospital admissions while assisting health systems to forecast their chronic disease maintenance needs.
The market has undergone a structural shift during the past 3-5 years, which now focuses on biologics and personalized treatment pathways while moving away from traditional inhalers toward precise immunotherapy solutions. The current transition exists because both diagnostic accuracy has improved and payers now accept reimbursement for expensive treatments which decrease future health complications. The COVID-19 pandemic created the primary reason which demonstrated that respiratory patients face multiple health risks while fulfilling their commitment to preventive care and remote assessment procedures.
The market expansion process begins with enhanced targeting methods, which produce superior results. The superior results validate high-cost solutions, which receive additional reimbursement, and that leads to increased market access for severe asthma treatments.
Key Market Insights
- Western Europe occupies the leading position in the Europe Asthma Therapeutics Market because its advanced healthcare infrastructure enables the region to maintain 45% of the market share which operates at 2025.
- The Northern Europe region experiences the quickest growth during the forecast period because government-sponsored respiratory care initiatives exist and the region has already started using biologic treatments.
- Inhaled corticosteroids maintain their leading position because they account for more than 40% of the market which medical professionals use as their primary maintenance treatment throughout Europe.
- The second-largest market share belongs to combination inhalers because they help patients stick to their treatment plans while delivering dual-action therapeutic benefits.
- Biologics represent the fastest-growing segment which will achieve more than 12% CAGR growth until 2030 because they provide treatment for severe asthma through targeted therapies.
- Chronic asthma management controls approximately 65% of the total market revenue because patients require long-term treatment for their condition.
- The fastest-growing application in asthma treatment involves severe asthma treatment which has gained traction because healthcare facilities now use monoclonal antibody therapies.
- Hospitals control approximately 50% of the market because they provide clients access to specialized medical services and advanced biologic treatments.
- The homecare segment experiences the fastest growth rate because European countries adopt digital inhalers and remote patient monitoring systems.
What are the Key Drivers, Restraints, and Opportunities in the Europe Asthma Therapeutics Market?
The Europe Asthma Therapeutics Market experiences its main growth driver through the increasing adoption of biologic treatments for severe asthma cases. The development started because clinical studies confirmed that monoclonal antibodies could block specific inflammatory pathways, which resulted in larger European healthcare systems approving reimbursement for the treatment after 2020. The health insurers started to accept higher initial costs for treatment because they understood that biologic medicines would decrease hospital stays and prevent future health problems. The system resulted in higher earnings from each patient because it improved treatment compliance among patients with severe asthma, which raised the total market value.
The expensive nature of biologic therapies together with their narrow eligibility requirements creates a fundamental structural obstacle that prevents access to these treatments. The need for biomarker-based diagnosis together with specialist oversight requirements for these treatments creates an access limitation that affects only specific patients. The public healthcare systems in Southern and Eastern European regions cannot handle widespread biologic therapy implementation unless they establish strict financial management procedures. The restriction leads to treatment delays which also prevent revenue generation in mid-income markets because their diagnostic systems and infrastructure development remain inconsistent.
The digital therapeutics market together with smart inhaler systems enables organizations to pursue new growth opportunities that drive their business expansion. The connected inhalers used in Germany and the UK enable users to monitor their inhaler usage together with their current treatment progress. The tools create a twofold benefit by allowing medical professionals to monitor patient health while they access telehealth services, which boosts their ability to identify health issues early and control their treatment progress.
What Has the Impact of Artificial Intelligence Been on the Europe Asthma Therapeutics Market?
The European asthma healthcare system undergoes transformation through artificial intelligence and advanced digital technologies which enhance therapy monitoring and optimization and personalized treatment approaches. The AI-based platforms now perform automated treatment adherence monitoring through their connected inhalers which track both dosage timing and frequency of usage to enable doctors to monitor patients' behavior from a distance while making prescription changes without requiring patients to visit their offices. The automated system streamlines follow-up processes which leads to better treatment compliance and results in improved clinical results.
The medical field uses machine learning models to forecast asthma attacks by studying real-time patient data that includes inhaler usage patterns and environmental exposure and past health records. The predictive systems create opportunities for early intervention which decreases emergency hospital visits and maintains stable expenses for long-term medical treatment. The digital platforms identify the best treatment options for patients through their biomarker data which helps them find the most effective biologic or inhaler-based medicines.
These technologies boost treatment rates and cut emergency medical expenses while they enhance operational efficiency throughout all healthcare facilities. European healthcare systems face a major obstacle because their data infrastructure remains divided into separate components. The inconsistent data integration process together with privacy regulations creates obstacles for AI systems to operate at full capacity which decreases model precision and prevents digital solutions from becoming widely used.
Key Market Trends
- The use of biologics medications has increased since 2020 from their original restricted applications to now extending their impact through targeted treatments to more than 30 percent of severe asthma patients who need treatment.
- After 2021 officials in Germany and the UK altered their reimbursement systems to support outcome-based pricing models for expensive asthma biologics.
- The usage of smart inhalers experienced a major boost after the COVID-19 pandemic because digital tracking systems for patients showed a 20 percent better adherence rate.
- AstraZeneca and GlaxoSmithKline shifted their R&D funding towards monoclonal antibody development and biomarker-based therapeutic research since 2019.
- From 2021 until 2024 regulators approved combination inhalers which feature improved drug delivery systems because they help patients who suffer from chronic conditions to stick to their treatment plans.
- Since 2022 Eastern European countries have been steadily increasing access to respiratory care because their governments have allocated more resources for respiratory health facilities and diagnostic equipment.
- The use of telehealth for asthma management increased dramatically after 2020 because it allowed patients to have remote medical consultations while decreasing their need to visit hospitals for non-urgent medical issues in Western European countries.
- After 2021 real-world evidence studies became commonly used because they affected doctors' prescribing patterns while demonstrating the effectiveness of personalized asthma treatment methods.
- Manufacturers changed their supply chain approaches by establishing local production facilities after the pandemic to decrease their need for imported active pharmaceutical ingredients.
Europe Asthma Therapeutics Market Segmentation
By Type
The primary care system depends on corticosteroids and bronchodilators because these two medication types have existed in clinical practice for a long time and they remain affordable. Physicians depend on inhaled corticosteroids as their primary treatment option which results in these medications becoming the most commonly used asthma maintenance therapy. Bronchodilators maintain a strong market position because they provide patients with fast relief from symptoms which is particularly valuable during emergency situations. Biologics show their most rapid growth pattern because they deliver specific treatment for severe asthma pathways while Western Europe expands its reimbursement system.
Biologic treatments experience market growth because doctors can now identify diseases at a higher accuracy rate and they understand personalized medicine treatment options better. Lower-income regions face challenges because people there cannot access healthcare services which become unaffordable for them. The future will develop into a market system which has two components because traditional inhalers will continue to create sales volume while biologics will generate new value which prompts manufacturers to develop affordable products through their research activities.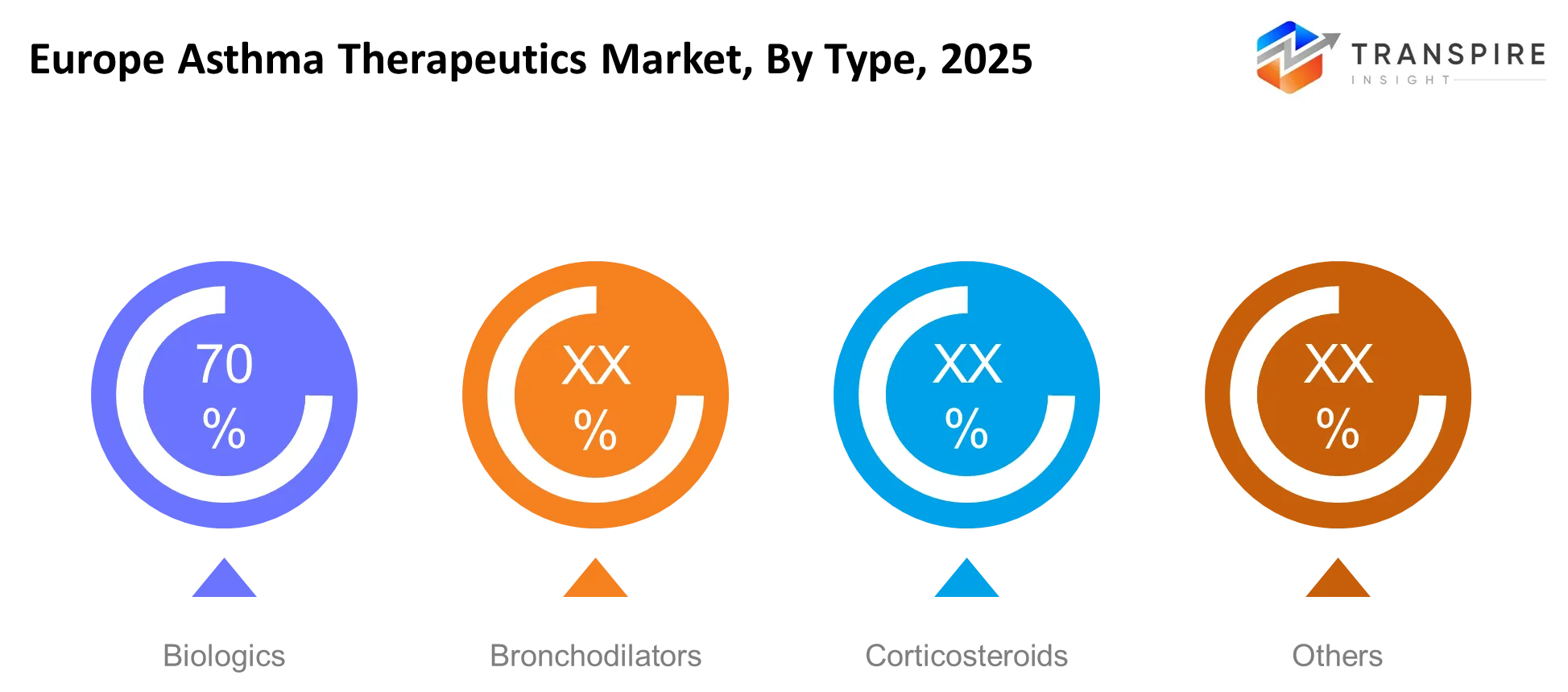
To learn more about this report, Download Free Sample Report
By Application
The most common treatment method for chronic asthma requires ongoing monitoring because it serves the needs of the largest patient group who require continuous medical care. The segment generates consistent income because patients follow their treatment plans and doctors issue repeat medication orders. The severe asthma treatment market has a small customer base but it is experiencing rapid growth because more people start using biologic treatments and specialized treatment methods. Medical professionals can better identify patients with different disease levels because they now use biomarker tests to distinguish between moderate and severe conditions.
The treatment of intermittent asthma only provides minimal value for which people access medical services because patients only need treatment during their asthma episodes. Healthcare systems will direct their resources to handle severe asthma cases because they want to decrease their expenses associated with patient hospitalizations. The current trend leads drug manufacturers to focus their resources on developing treatment options that deliver high success rates while medical practitioners concentrate their efforts on detecting patients who have a high probability of developing serious health conditions.
By End-User
The end-user hospitals maintain their leadership position because they possess advanced therapies and expert specialists and the facilities needed to deliver and monitor biologic treatments. The hospitals treat the highest number of patients because severe asthma requires both observation and diagnostic assistance. Clinics function as crucial treatment centers for patients with mild to moderate conditions because they deliver healthcare services that are both affordable and easily reachable.
The homecare market is experiencing rapid growth because digital inhalers and remote monitoring tools have entered the market while patients increasingly choose to manage their health at home. The healthcare systems started to decrease hospital admissions for non-urgent medical needs after the pandemic period. The success of homecare programs requires patients to receive training and healthcare facilities to establish digital service networks. The future will bring a slow transition of asthma care from hospitals to home-based treatment except for patients with severe medical needs.
By Distribution
The majority of hospital pharmacies distribute their medications because they serve patients who need inpatient treatment and they require special controls to dispense biologic medicines. Retail pharmacies distribute maintenance drugs and inhalers through their extensive network which enables customers to buy products multiple times. Online and alternative channels are becoming more popular because digital health adoption makes it easier for people to fulfill their prescriptions.
The distribution channels have expanded because new patient behaviors have emerged which show that e-pharmacies serve as the primary resource for chronic disease treatment. The existing regulations and reimbursement frameworks shape how distribution channels operate especially when it comes to expensive medical treatments. The upcoming distribution patterns will establish a combination system where hospital pharmacies manage complex medical treatments while retail and online platforms extend access to ongoing treatment options which will allow combined supply chain management methods.
What are the Key Use Cases Driving the Europe Asthma Therapeutics Market?
The primary purpose of the system depends on its ability to provide continuous treatment needed by patients who receive primary and outpatient medical services throughout Europe. Patients who use inhaled corticosteroids and bronchodilators on a regular basis achieve symptom stabilization while their risk of dangerous health complications decreases, which leads to fewer hospitalizations and decreased long-term medical expenses. The application creates its highest demand because it needs to handle large groups of patients who require daily medical care and prescription renewals.
The medical field needs more use cases which would allow hospitals and specialist clinics to treat severe asthma cases through biologic treatments. These therapies target specific inflammatory pathways and are used for patients who do not respond to standard inhalers. Homecare-based monitoring systems are beginning to gain popularity because digital inhalers enable healthcare providers to monitor patient adherence and conduct remote patient consultations.
The healthcare system needs two new use cases, which combine AI-based predictive care with environmental data to create better treatment outcomes. Western European countries operate their initial programs which use real-time air quality data to modify therapy approaches, providing the opportunity for proactive control of asthma symptoms.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 5690.2 Million |
|
Market size value in 2026 |
USD 5923.3 Million |
|
Revenue forecast in 2033 |
USD 7967.6 Million |
|
Growth rate |
CAGR of 4.33% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain and Rest of Europe) |
|
Key company profiled |
GSK, AstraZeneca, Novartis, Sanofi, Roche, Teva, Merck, Pfizer, Boehringer Ingelheim, Amgen, Regeneron, AbbVie, Bayer, Mylan, Cipla |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Biologics, Bronchodilators, Corticosteroids, Others), By Application (Severe Asthma, Chronic Asthma, Others), By End-User (Hospitals, Clinics, Homecare, Others), By Distribution (Hospital Pharmacies, Retail Pharmacies, Others) |
Which Regions are Driving the Europe Asthma Therapeutics Market Growth?
The market is currently dominated by Western Europe because of its efficient public healthcare systems and its early use of biologics and its established reimbursement processes. The countries of Germany and the UK and France establish specific treatment routes which promote precision medicine and expert-driven treatment. Advanced diagnostic systems enable precise patient classification which enables healthcare providers to implement specific treatments throughout their operations. The region maintains its pharmaceutical industry leadership through its established research and development capabilities which drive ongoing product development.
Northern Europe establishes its special identity through its combination of stable systems and preventive healthcare methods which enhance its healthcare services beyond organizational size. The countries of Sweden and Denmark focus on implementing early treatment methods and digital health solutions and developing strategies for long-term management of chronic illnesses. The government provides continuous funding while the healthcare system establishes networks to deliver asthma treatment which results in predictable medicine usage throughout the year. The region generates consistent revenue growth because patients maintain their treatment plans and healthcare providers implement new treatments throughout the entire population.
Eastern Europe has become the region that grows the most rapidly because of new healthcare funding and better access to contemporary medical treatments. Since 2022 multiple nations have increased their financial support for respiratory medicine while they developed better diagnostic technologies which enable faster and more precise medical intervention. The gradual process of adding biologics to reimbursement lists has begun to provide treatment access for patients who previously lacked coverage. The period between 2026 and 2033 will create multiple market entry opportunities because the region develops better infrastructure while government policies provide more support.
Who are the Key Players in the Europe Asthma Therapeutics Market and How Do They Compete?
The Europe Asthma Therapeutics Market shows moderate consolidation through its competitive landscape because multinational pharmaceutical firms control most valuable biologics segments while generic manufacturers battle for market share in inhaler products that generate high sales volumes. Companies now compete through technological advancements and unique clinical attributes instead of solely using pricing strategies. The existing companies maintain their market position through exclusive medications that they protect with patents and their established relationships with payers while new companies concentrate their efforts on developing biosimilars and integrating digital health solutions. The shift toward personalized medicine has raised the barrier to entry, as success now depends on biomarker expertise, clinical data, and regulatory alignment across European markets.
AstraZeneca establishes its market presence through biologics research which leads to drug development for eosinophilic asthma that provides specific treatment results which decrease the number of severe asthma attacks. The company establishes its unique position through its comprehensive clinical trial results which enable it to connect with specialized medical networks throughout Europe. GlaxoSmithKline maintains its competitive edge through product development of respiratory solutions which combine inhaler treatments with modern drug delivery technologies. The company achieves its long-term financial success through ongoing development of advanced inhalers and its strategies for managing product lifecycles.
Novartis uses precision medicine to match asthma treatments with biomarker-based diagnostic tests, which enables better treatment results because of improved patient selection. Sanofi develops its business through strategic partnerships which help the company build its immunology knowledge while creating new biologics access points throughout European healthcare systems. The companies achieve market advantage by integrating treatment effectiveness with their methods for engaging payers because this combination secures funding and establishes long-term use in high-value patient groups.
Company List
- GSK
- AstraZeneca
- Novartis
- Sanofi
- Roche
- Teva
- Merck
- Pfizer
- Boehringer Ingelheim
- Amgen
- Regeneron
- AbbVie
- Bayer
- Mylan
- Cipla
Recent Development News
In April 2026, 7UP Enters Energy Drinks Category with New Product Launch: A major shift in the U.S. beverage landscape as 7UP expands into energy drinks through a strategic collaboration. This move reflects increasing crossover between soda and energy drink segments to capture younger consumers.
Source: https://www.allrecipes.com
In April 2026, PepsiCo Reports Strong Q1 2026 Performance with Beverage Strategy Shift: PepsiCo strengthened its beverage portfolio through pricing strategy, product refresh, and healthier offerings. The company highlighted ongoing adjustments to remain competitive amid rising costs and changing consumer demand.
Source: https://www.reuters.com
What Strategic Insights Define the Future of the Europe Asthma Therapeutics Market?
The Europe Asthma Therapeutics Market is progressing toward treatment delivery through precision medicine that uses digital technology to provide respiratory care solutions which use biomarker testing and real-world data and ongoing patient observation to make treatment choices. The system changes because payers demand decreased expenses from ongoing hospital stays together with medical professionals needing better treatment options which deliver specific results. Over the next 5–7 years, severe asthma management will create value through biologic treatments and digital adherence systems rather than through traditional inhaler volume growth.
European health systems require high-cost biologics as reimbursement policies become stricter through ongoing budget constraints. The mid-income regions will experience access restrictions because cost-containment policies become stricter which will decrease adoption of products with proven clinical efficacy. Two Western European research hospitals currently pilot an opportunity that enables treatment algorithms to utilize both environmental data and genomic information for patient diagnosis. This method will establish new patient groups while enhancing treatment options for advanced medical procedures.
Market participants should develop hybrid product lines which integrate biologics and digital monitoring systems to achieve reimbursement stability and maintain their clinical value throughout time.
Europe Asthma Therapeutics Market Report Segmentation
By Type
- Biologics
- Bronchodilators
- Corticosteroids
- Others
By Application
- Severe Asthma
- Chronic Asthma
- Others
By End-User
- Hospitals
- Clinics
- Homecare
- Others
By Distribution
- Hospital Pharmacies
- Retail Pharmacies
- Others
Frequently Asked Questions
Find quick answers to common questions.
The Approximate Europe Asthma Therapeutics Market size for the Market will be USD 7967.6 Million in 2033.
The key Segments of the Europe Asthma Therapeutics Market are By Type (Biologics, Bronchodilators, Corticosteroids, Others), By Application (Severe Asthma, Chronic Asthma, Others), By End-User (Hospitals, Clinics, Homecare, Others), By Distribution (Hospital Pharmacies, Retail Pharmacies, Others).
Major Players in the Europe Asthma Therapeutics Market are GSK, AstraZeneca, Novartis, Sanofi, Roche, Teva, Merck, Pfizer, Boehringer Ingelheim, Amgen, Regeneron, AbbVie, Bayer, Mylan, Cipla.
The Current Market size of the Europe Asthma Therapeutics Market is USD 5690.2 Million in 2025.
The Europe Asthma Therapeutics Market CAGR is 4.33%.
- GSK
- AstraZeneca
- Novartis
- Sanofi
- Roche
- Teva
- Merck
- Pfizer
- Boehringer Ingelheim
- Amgen
- Regeneron
- AbbVie
- Bayer
- Mylan
- Cipla
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


