Europe Asthma Treatment Market Size & Forecast:
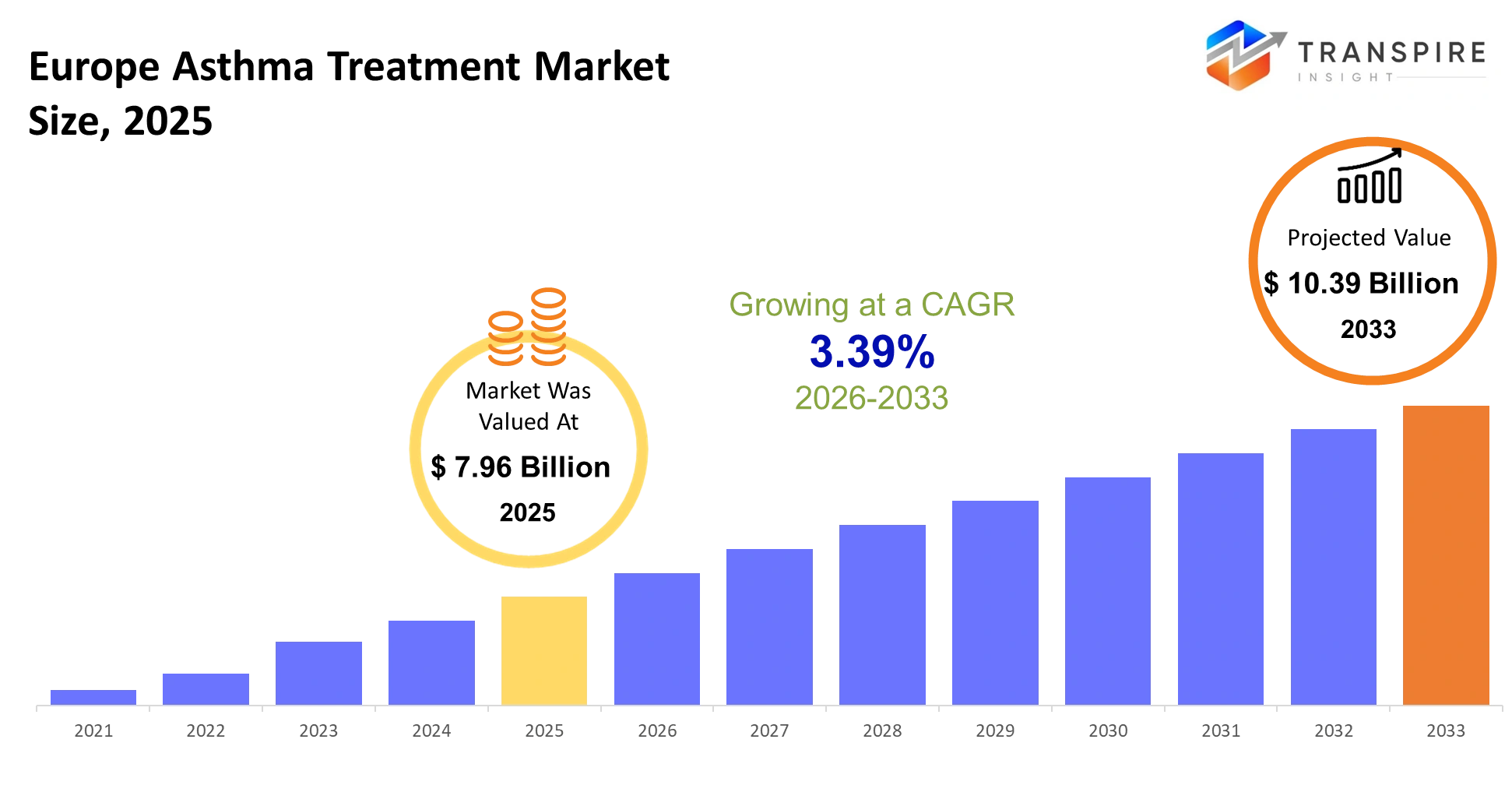
- Europe Asthma Treatment Market Size 2025: USD 7.96 Billion
- Europe Asthma Treatment Market Size 2033: USD 10.39 Billion
- Europe Asthma Treatment Market CAGR: 3.39%
- Europe Asthma Treatment Market Segments: By Type (Inhalers, Oral Medications, Biologics, Others), By Application (Mild Asthma, Moderate Asthma, Severe Asthma, Others), By End-User (Hospitals, Clinics, Homecare, Others), By Drug Class (Bronchodilators, Corticosteroids, Others).
To learn more about this report, Download Free Sample Report
Europe Asthma Treatment Market Summary:
The Europe Asthma Treatment Market size is estimated at USD 7.96 Billion in 2025 and is anticipated to reach USD 10.39 Billion by 2033, growing at a CAGR of 3.39% from 2026 to 2033. The Europe asthma treatment market exists to provide patients with better management of their condition which enables them to remain in their daily activities while their respiratory inflammation stays under control and their sudden health episodes are prevented. The system provides continuous support for disease management which extends from primary healthcare facilities to hospitals and home care services while it decreases unnecessary hospital admissions that would strain the healthcare system.
In the past three to five years market trends have shifted toward biologic treatments and precision medicine which provide effective solutions for severe asthma subtypes but charge higher prices. The COVID-19 pandemic created an actual situation that drove the implementation of remote patient monitoring systems together with digital inhalers and adherence-tracking tools because patients preferred to avoid visiting healthcare facilities.
The combination of these factors has created new patterns for revenue generation. The combination of higher-value biologics and connected devices leads to increased costs for each patient that needs treatment because of better diagnostic methods and improved patient medication compliance. The company experiences growth which stems from increased therapy usage and extended time needed to manage chronic illnesses.
Key Market Insights
- The Europe Asthma Treatment Market in Western Europe which controls 60% of the market will grow from 2025 because of its advanced medical facilities and its reimbursement system.
- The industry size of Germany France and the UK reaches its peak because these countries diagnose more patients and provide access to advanced biologic treatments.
- The fastest-growing area in Eastern Europe will experience more than 6% growth until 2030 because healthcare access continues to improve.
- Inhaled corticosteroids control the market with more than 40% of the market share because they serve as the primary treatment option for asthma patients who need ongoing control.
- The second-largest market share belongs to combination inhalers because they help patients stick to their treatment plans while showing effective results in clinical studies.
- The biologics market represents the fastest-expanding sector with annual growth forecasted to exceed 8% until 2030 because of rising demand for targeted therapies.
- The Europe asthma treatment market generates its primary revenue from chronic asthma management which accounts for about 70% of the market share because patients requireongoing treatment for their condition.
- The application of severe asthma treatment will experience its most rapid growth between 2024 and 2030 because more people will start using monoclonal antibody treatments.
- The Europe Asthma Treatment Market exists as a hospital market which will achieve around 45% market share by 2025 because hospitals provide both advanced medical treatments and specialized patient services.
- The growth of homecare settings accelerates because digital inhalers and remote monitoring technologies help patients adhere to their treatment plans.
What are the Key Drivers, Restraints, and Opportunities in the Europe Asthma Treatment Market?
The primary factor that drives the European Asthma Treatment Market forward currently occurs because patients with severe and uncontrolled asthma now choose to receive biologic therapies. The medical community approved this transition after clinical studies confirmed the effectiveness of monoclonal antibodies which European treatment guidelines adopted within the past five years. The specialists who treat complex cases started to use high-cost biologics after payers began to fund these medications for patients who met certain requirements. The direct impact of biologics results in increased revenue because their price exceeds that of standard inhalers by multiple times. The market value of the drug has permanently increased because the patient base has not expanded at the same rate of market value growth.
The most significant barrier is the high cost and restricted reimbursement of advanced therapies, particularly biologics. The European healthcare systems face a structural barrier because their budgetary limits and mandatory health technology assessments prevent the entire population from receiving health services. The approval process for therapies that show clinical effectiveness experiences delays while their eligibility requirements remain restricted. The market growth gets restricted because many patients with moderate-to-severe conditions continue using cheaper treatments, which stops them from generating full revenue potential and prevents new medications from reaching the market.
The key opportunity exists because digital health tools should integrate with asthma care through smart inhalers and remote monitoring platforms. The reimbursement system for digital therapeutics in Germany and the UK provides an expansion pathway which allows for widespread adoption. The technologies enhance patient adherence while producing real-world data, which doctors use to create personalized treatment plans that expand their patient reach and develop new revenue sources.
What Has the Impact of Artificial Intelligence Been on the Europe Asthma Treatment Market?
The introduction of artificial intelligence together with advanced digital technologies has started to change asthma treatment methods throughout Europe through their use of intelligent systems that support treatment processes and monitoring operations and clinical decision-making activities. The AI-based platforms which use connected inhalers to monitor patient progress now perform automatic monitoring through their ability to detect how patients use the device and when they take their medication and which medicines they follow. The systems transmit current patient information to clinicians who use it to modify treatment plans from a distance while decreasing the need for in-person consultations.
Environmental monitoring together with patient intelligence and behavioral patterns studies enables machine learning models to predict asthma attacks. The predictive algorithms of the system enable identification of high-risk patients by analyzing air quality and pollen data together with historical symptom patterns. The system enables a shift from reactive treatment methods to preventive care which results in better patient outcomes and decreased emergency room visits.
The technologies increase treatment compliance through operational improvements which result in 20 to 30 percent better results. The technologies provide healthcare systems with tools to manage their resources while achieving better results in long-term disease management.
The main obstacle that exists to progress in healthcare systems is the problem of divided data between different healthcare networks. The combination of different data standards together with limited system connections results in decreased model precision which prohibits large-scale AI implementation across multiple European healthcare systems.
Key Market Trends
- The European treatment guidelines have adopted biologics as the primary treatment for severe asthma since 2020 while AstraZeneca and GlaxoSmithKline drugs now receive more reimbursement approvals.
- Between 2021 and 2025 payers implemented stricter requirements for high-cost therapy eligibility which forced manufacturers to provide proof of actual treatment results in order to maintain their reimbursement agreements.
- Smart inhaler usage increased by over 25% after 2022 because UK and German healthcare systems began using digital adherence tracking as part of their regular asthma treatment methods.
- Hospital-directed asthma treatment experienced a decline in usage after 2020 because telehealth systems which developed during the COVID-19 pandemic made it possible for patients to receive asthma treatment at home.
- Pharmaceutical companies shifted their research and development funding after 2021 to create monoclonal antibodies which target specific asthma phenotypes while reducing their financial support for traditional bronchodilator research.
- The European Union funding programs have helped Eastern European markets provide earlier access to advanced treatments since 2023 by enhancing healthcare facilities and diagnostic services through their funded infrastructure improvements.
- Environmental rules became more restrictive during the period from 2020 to 2024 because air quality regulations caused an increase in diagnosed asthma cases which resulted in more patients receiving treatment.
- Sanofi and other companies changed their competitive strategies through partnerships with biotechnology companies which enabled them to develop biologics more effectively while reducing product development time.
- AI-based risk prediction systems after 2022 created data-driven treatment frameworks which enable doctors to find at-risk patients sooner and create better treatments for their future medical requirements.
Europe Asthma Treatment Market Segmentation
By Type
The inhaler type proves to be the most effective solution because it establishes clinical evidence which provides fast symptom relief and doctors in primary care clinics prefer its use. The market for metered-dose and dry powder inhalers exists as the biggest market segment because these products provide users with easy-to-use solutions which cost less for their extended use. The use of oral medications exists at a lower status because they serve as supplementary treatments which doctors use to treat patients who need more than inhalation therapy.
The biologics market segment exhibits its most rapid expansion because Western Europe now provides more funding options for severe asthma phenotype identification. The market segment has expanded because of the industry's shift towards providing specialized medical solutions which deliver superior therapeutic results. Biologic development and device development become the main focus for manufacturers while investors target therapies which demonstrate exceptional clinical results. The buyers, who include healthcare systems, work to establish an equilibrium between their need for cost control and their goal of achieving better patient results.
By Application
The highest proportion of patients who need constant medical care exists within moderate asthma which represents the most extensive patient group. The market for mild asthma presents ongoing demand but results in decreased earnings because patients only need basic inhalers. The number of patients who have severe asthma has increased because medical professionals now diagnose and refer these patients to specialized treatment at a higher rate.
The segment generates excessive income because of its use of biologic treatments together with multiple therapies. The current treatment pathways now follow more distinct paths because the guidelines recommend that doctors should start more aggressive treatment for patients who do not respond to initial therapies. The growth of the upcoming period requires accurate patient diagnosis through phenotype classification which will result in precise treatment implementation. The manufacturers and providers need to match their product offerings according to different disease severity levels while developing better tools for disease diagnosis.
To learn more about this report, Download Free Sample Report
By End-User
Hospitals maintain their position as market leaders through their ability to provide specialists and advanced diagnostic tools and medical facilities that require clinical supervision for high-cost biologic treatments. Urban healthcare networks depend on clinics as their primary option for asthma management and prescription refill services.
The homecare market is expanding rapidly because patients prefer to receive treatment at home through digital inhalers and telemedicine services. The COVID-19 pandemic facilitated this transition by speeding up the adoption of decentralized healthcare delivery systems. Healthcare systems are building remote monitoring systems which will help them decrease hospital admissions and better patient compliance. The industry needs to develop digital solutions which work with existing treatment options while establishing partnerships with outpatient service providers.
By Drug Class
Corticosteroids show the highest usage rates among drug categories because they serve as essential medications for controlling airway inflammation and achieving sustained therapeutic results. Bronchodilators function as vital emergency and ongoing treatment methods which medical professionals frequently administer together with corticosteroids to achieve better patient results. Advanced drug classes demonstrate their highest growth rate through monoclonal antibodies which treat specific inflammatory pathways that appear in severe asthma cases.
The current trend shows medical professionals moving away from standard treatment methods to focus on specialized treatment according to how diseases develop. Cost and reimbursement issues serve as main obstacles which prevent some areas from achieving full accessibility. The future will see manufacturers directing their resources toward research which develops specific medications and combination treatments while payers will require proof of both long-term clinical benefits and economic savings.
What are the Key Use Cases Driving the Europe Asthma Treatment Market?
The primary application for asthma treatment proves to be long-term management of moderate asthma which results in high treatment demand through primary care networks. Physicians depend on inhaled corticosteroids together with combination inhalers to maintain patient stability while reducing their risk of experiencing asthma attacks. The use case requires ongoing treatment which leads to regular prescription patterns and persistent patient interaction with healthcare services.
The medical field now utilizes biologics to treat severe asthma cases which require treatment through hospital and specialist clinic facilities. The field of homecare management is expanding its reach through digital inhalers and telehealth platforms which help patients follow their treatment plans while minimizing their need to visit hospitals.
The new use cases demonstrate their value through AI systems which predict and prevent health issues by using environmental data and patient information to predict disease outbreaks. The implementation of early intervention models in urban areas with strict air quality standards will gradually transform existing treatment systems.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 7.96 Billion |
|
Market size value in 2026 |
USD 8.23 Billion |
|
Revenue forecast in 2033 |
USD 10.39 Billion |
|
Growth rate |
CAGR of 3.39% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain and Rest of Europe) |
|
Key company profiled |
GSK, AstraZeneca, Novartis, Sanofi, Roche, Teva, Merck, Pfizer, Boehringer Ingelheim, Amgen, Regeneron, AbbVie, Bayer, Mylan, Cipla |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Inhalers, Oral Medications, Biologics, Others), By Application (Mild Asthma, Moderate Asthma, Severe Asthma, Others), By End-User (Hospitals, Clinics, Homecare, Others), By Drug Class (Bronchodilators, Corticosteroids, Others) |
Which Regions are Driving the Europe Asthma Treatment Market Growth?
The reimbursement systems of Western Europe demonstrate their effectiveness because they enable Germany France and the UK to enforce their clinical treatment regulations for advanced biologic therapies. Public healthcare systems provide access to advanced therapies which enable doctors to prescribe expensive treatments that follow established medical coverage procedures. The healthcare system achieves its main goal through its extensive network of pulmonologists and diagnostic labs which work together with digital health infrastructure to achieve continuous patient care and medical procedure improvement. The integrated system enables healthcare providers to implement new treatments for their patients while maintaining their dominant position in the area.
Southern European countries develop their economies through stable economic conditions which drive their government policies to adopt European Union regulations without implementing any groundbreaking changes. The budget-limited systems of Italy and Spain enable doctors to choose between combined inhalers and biologics through their continuous development of these two treatments. The government-led healthcare reforms create a system which maintains treatment accessibility during both economic stability and economic downturns. The region maintains market stability through its dependable revenue streams which result from its gradual introduction of high-end medical treatments.
The fastest-growing area in Eastern Europe has experienced growth because of recent healthcare investments which improved advanced asthma treatment access since 2022. The diagnostic capabilities and specialist availability in Poland and Romania have received improvements from EU funding and national modernization programs. The faster therapy approvals and reimbursement processes for new treatments emerged because of the regulatory system's alignment with Western European standards. The existing momentum provides manufacturers who intend to serve underserved populations with powerful market entry options while establishing the area as a primary development territory until 2033.
Who are the Key Players in the Europe Asthma Treatment Market and How Do They Compete?
The Europe Asthma Treatment Market shows moderate competition because several major pharmaceutical companies control important market sections while smaller biotech companies create new solutions. The current competition mode which exists between companies has evolved from manufacturers producing high volumes of inhalers to companies using advanced technologies for their biologic and targeted therapy products. Companies that already exist in the market use product lifecycle management together with new product approvals to protect their market share while new companies enter the market to serve specific severe asthma treatment requirements. Public healthcare systems create pricing pressure which forces companies to demonstrate product value through clinical results and actual usage data instead of relying on their market presence
AstraZeneca develops new biologic therapies which treat severe asthma by creating drugs that block particular inflammatory pathways. The method enables doctors to identify particular patients who will respond better to treatment which results in improved clinical outcomes that allow higher pricing. GlaxoSmithKline uses two strategies to create its market position because its strong inhaler collection together with its biologic development programs enables the company to reach both mainstream customers and premium market segments. The company uses its established doctor partnerships together with its device development capabilities to create products that help customers maintain their treatment schedules.
Sanofi develops its immunology treatment portfolio through partnerships that help its biologic research and regulatory approval processes. Novartis invests in future monoclonal antibodies development while it investigates digital solutions for remote patient observation through its research-based approach. Boehringer Ingelheim develops its inhalation therapy business by creating new devices and improving its drug delivery systems.
Company List
- GSK
- AstraZeneca
- Novartis
- Sanofi
- Roche
- Teva
- Merck
- Pfizer
- Boehringer Ingelheim
- Amgen
- Regeneron
- AbbVie
- Bayer
- Mylan
- Cipla
Recent Development News
In April 2026, AstraZeneca’s Breztri Wins New Asthma Approval, Expanding Respiratory Portfolio: AstraZeneca secured a major regulatory milestone for Breztri Aerosphere, strengthening its presence in the global asthma treatment segment. The approval is expected to expand the company’s respiratory market reach across Europe and support long-term biologics and inhaler growth strategies.
Source: https://www.fiercepharma.com
In April 2026, GSK Reports Strong Q1 2026 Growth Driven by Respiratory Medicines: GSK’s first-quarter 2026 financial results highlighted strong momentum in its respiratory and asthma portfolio. Investors closely tracked the company’s progress in long-acting asthma biologics as GSK continues expanding its respiratory pipeline in Europe.
Source: https://www.reuters.com
What Strategic Insights Define the Future of the Europe Asthma Treatment Market?
The Europe Asthma Treatment Market is moving toward a precision-care model which allocates value to specific biologics and companion diagnostics and systems that track patient adherence through digital technology. Payer pressure requires funding only for therapies which demonstrate positive patient outcomes while clinical guidelines recommend treatments based on specific disease phenotypes. The revenue growth of the next 5 to 7 years will depend on therapy usage and clinical results instead of patient intake numbers.
The market faces a hidden danger because it relies on a small number of biologic treatments which may lead to price investigations and reimbursement restrictions if financial constraints become more severe. The development of real-world data platforms can create new market opportunities when combined with treatment protocols especially in Eastern Europe which has developing digital health systems and better regulatory compliance.
The participants need to focus their strategic efforts on developing integrated care models which combine therapeutic treatment with digital health monitoring and data analysis for clinical outcomes because this approach allows them to present their products as affordable treatment options instead of single medication solutions.
Europe Asthma Treatment Market Report Segmentation
By Type
- Inhalers
- Oral Medications
- Biologics
- Others
By Application
- Mild Asthma
- Moderate Asthma
- Severe Asthma
- Others
By End-User
- Hospitals
- Clinics
- Homecare
- Others
By Drug Class
- Bronchodilators
- Corticosteroids
- Others
Frequently Asked Questions
Find quick answers to common questions.
The approximate Europe Asthma Treatment Market size for the market will be USD 10.39 Billion in 2033.
The key segments of the Europe Asthma Treatment Market are By Type (Inhalers, Oral Medications, Biologics, Others), By Application (Mild Asthma, Moderate Asthma, Severe Asthma, Others), By End-User (Hospitals, Clinics, Homecare, Others), By Drug Class (Bronchodilators, Corticosteroids, Others).
Major players in the Europe Asthma Treatment Market are GSK, AstraZeneca, Novartis, Sanofi, Roche, Teva, Merck, Pfizer, Boehringer Ingelheim, Amgen, Regeneron, AbbVie, Bayer, Mylan, Cipla.
The current market size of the Europe Asthma Treatment Market is USD 7.96 Billion in 2025.
The Europe Asthma Treatment Market CAGR is 3.39%.
- GSK
- AstraZeneca
- Novartis
- Sanofi
- Roche
- Teva
- Merck
- Pfizer
- Boehringer Ingelheim
- Amgen
- Regeneron
- AbbVie
- Bayer
- Mylan
- Cipla
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033