Japan Phenylenketonuria (PKU) Treatment Market Size & Forecast:
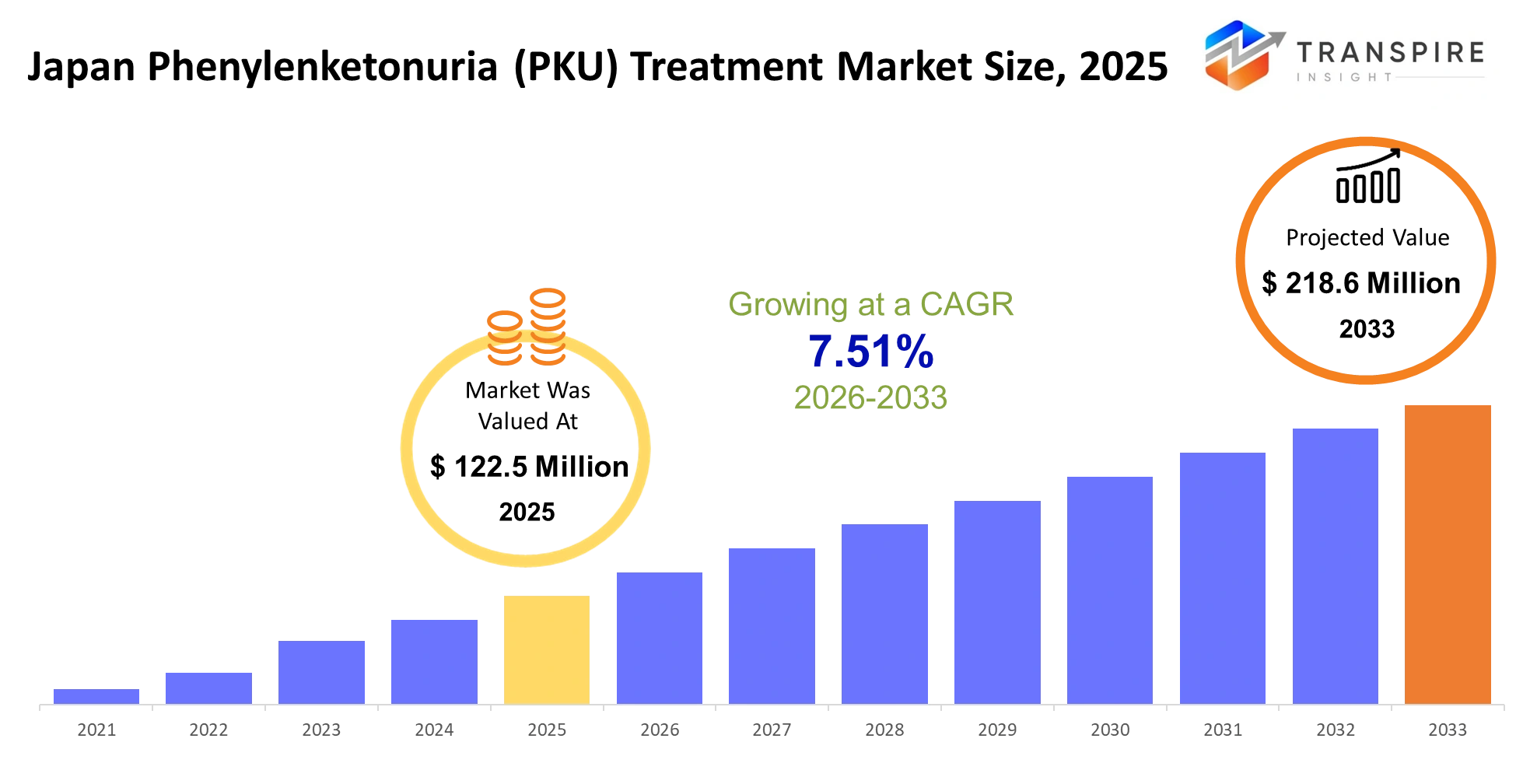
- Japan Phenylenketonuria (PKU) Treatment Market Size 2025: USD 122.5 Million
- Japan Phenylenketonuria (PKU) Treatment Market Size 2033: USD 218.6 Million
- Japan Phenylenketonuria (PKU) Treatment Market CAGR: 7.51%
- Japan Phenylenketonuria (PKU) Treatment Market Segments: By Type (Gene Therapy, Drug Therapy, Physical Therapy, Stem Cell Therapy, Supportive Care, Others); By Application (Muscular Dystrophy Treatment, Rehabilitation, Neuromuscular Care, Genetic Disorder Treatment, Clinical Trials, Others); By End-User (Hospitals, Clinics, Rehab Centers, Research Institutes, Specialty Centers, Homecare, Others); By Distribution (Hospitals, Clinics, Specialty Pharmacies, Others).
To learn more about this report, Download Free Sample Report
Japan Phenylenketonuria (PKU) Treatment Market Summary
The Japan Phenylenketonuria (PKU) Treatment Market was valued at USD 122.5 Million in 2025. It is forecast to reach USD 218.6 Million by 2033. That is a CAGR of 7.51% over the period.
The Japan Phenylenketonuria (PKU) Treatment Market supports lifelong management of a rare metabolic disorder which prevents patients from metabolizing phenylalanine. Patients require medical diets which include specialized formulas and specific treatments to stop their condition from causing permanent brain damage. The system operates as a permanent healthcare solution which includes three components: neonatal screening, clinical monitoring, and nutrition-linked pharmaceutical support. The system functions as a permanent healthcare solution which provides medical treatment through three stages: neonatal screening, clinical monitoring, and nutritional pharmaceutical assistance.
The market has transformed during the last three to five years because people now prefer to treat their conditions with enzyme and cofactor therapies instead of traditional diet-based treatments. The COVID-19 pandemic caused imported medical food supply interruptions which exposed supply chain dependency issues. This situation prompted Japan to develop stronger systems for managing medical supply shortages and prescribing practices. The industry showed better profitability for expensive orphan drugs. This occurred after doctors started diagnosing patients at earlier stages and patients received treatments throughout their entire life. The medical system established reliable reimbursement methods which hospital systems used to support more advanced PKU treatments.
Key Market Insights
- The Japan Phenylenketonuria (PKU) Treatment Market currently powers its development through two main treatment options which include enzyme replacement therapy and pharmacological drugs.
- Newborn screening programs now cover over 95% of infants, which leads to better outcomes for both the detection of PKU in its early stages and the beginning of treatment.
- The Kanto region controls almost 40% of the market share because it possesses advanced genetic clinics and a greater number of doctors who treat metabolic disorders.
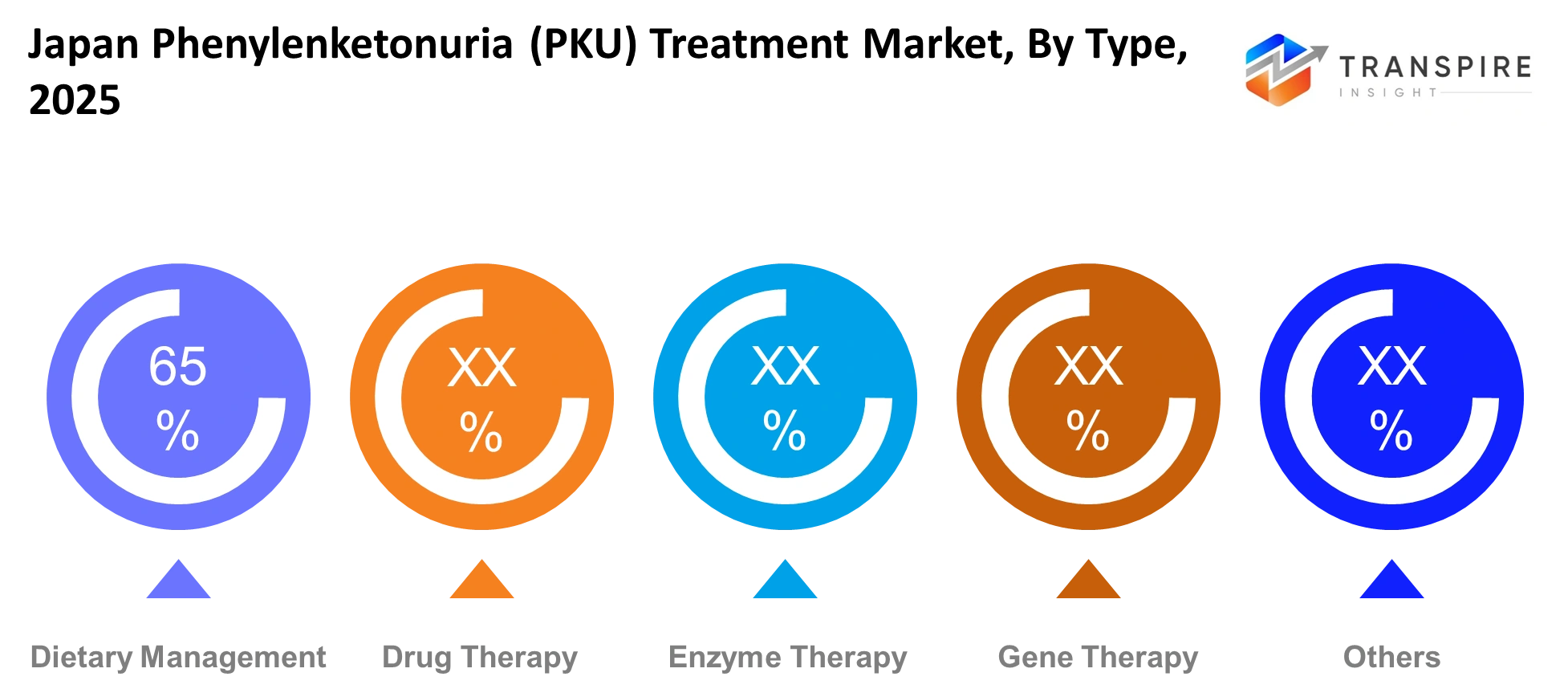
- The Japan Phenylenketonuria (PKU) Treatment Market maintains its leadership position through medical nutrition formulas which create approximately 55% market share.
- The second treatment option for enzyme substitution therapies exists because patients show better clinical results while needing fewer dietary restrictions.
- The fastest-growing segment of medical treatment development shows increased adoption of cofactor-based therapies which use BH4 analogs between 2024 and 2030.
- The requirement for lifelong treatment in PKU leads to chronic metabolic management which controls 70 percent of all medical treatment.
- Treatment delivery is primarily handled by hospitals and specialized metabolic clinics which control approximately 60 percent of the medical services.
- Digital monitoring tools together with telehealth expansion drive homecare and outpatient nutrition management to become the most rapidly expanding user segment.
- The companies expand their regional operations by developing local nutrition products and working with regulatory bodies.
What are the Key Drivers, Restraints, and Opportunities in the Japan Phenylenketonuria (PKU) Treatment Market?
The implementation of universal PKU testing for newborns in Japan together with improved funding for sapropterin dihydrochloride (BH4 therapy) and structured dietary management shows positive results through enhanced early disease detection and increased treatment rates which last throughout a person's lifetime. National screening programs enable hospitals to identify patients sooner which results in fewer neurological complications and higher treatment rates for patients with amino acid formula and adjunct therapy prescriptions. The new treatment procedures at hospitals and metabolic clinics start their processes immediately after screening confirmation which creates stable income sources for the facilities.
The PKU market remains restricted because the Japanese patient population consists of extremely rare cases which prevent medical food manufacturers from achieving manufacturing efficiency. The practice of lifelong dietary restriction for natural protein poses significant challenges which result in inconsistent treatment outcomes due to patients' varying levels of dietary compliance. The Japanese healthcare system does not provide broad access to emerging high-cost therapies like pegvaliase because they lack sufficient approval and reimbursement, which creates barriers that prevent people from using newer treatment methods.
The development of gene therapy and enzyme substitution technologies which target the PAH metabolic pathway presents future potential for innovation because Japan provides Sakigake fast-track designation for rare disease treatments. Digital metabolic monitoring systems together with AI-powered dietary planning solutions are becoming more popular because they help users maintain better health throughout their lives. The combination of increased biotech funding and international business partnerships will lead to faster clinical trial progress and market entry for products in Japan's rare disease market.
What Has the Impact of Artificial Intelligence Been on the Japan Phenylenketonuria (PKU) Treatment Market?
The reference to scrubber monitoring and marine emission control systems does not align with the Phenylenketonuria (PKU) treatment landscape which operates as a clinical and metabolic healthcare domain. The Japan PKU ecosystem uses artificial intelligence to operate digital metabolic monitoring platforms and treatment adherence systems according to this interpretation.
The automated dietary management applications which use AI technology to monitor patient methods of consuming phenylalanine whose thresholds hospitals established will notify doctors when their patients have surpassed these limits. The electronic health record system at tertiary care centers provides automated decision support which reduces the need for staff members to calculate amino acid doses and boosts their productivity during clinical procedures.
Machine learning models use predictive abilities to assess patient adherence risks based on their past dietary habits which allows for timely medical intervention that prevents metabolic decompensation. The systems assist with predicting long-term results which enable doctors to evaluate sapropterin responsiveness more effectively. Hospitals experience operational benefits because their treatment monitoring systems offer greater accuracy and their treatment adjustment processes work faster which leads to improved metabolic control and fewer urgent medical treatments even though different hospitals show different results.
The ultra-rare occurrence of PKU in Japan leads to a major research obstacle because there are insufficient substantial high-quality datasets to study. The restricted availability of large datasets prevents accurate model training while actual clinical situations restrict the predictive abilities of algorithms.
Key Market Trends
- Japanese universal newborn screening programs implemented earlier diagnosis methods which transformed their treatment process from treating symptoms to administering immediate metabolic treatment for newborns.
- The introduction of sapropterin dihydrochloride reimbursement allowed medical professionals to conduct pharmacological responsiveness tests which enabled them to shift from strict dietary management to personalized treatment approaches based on patient characteristics.
- Metabolic clinics adopted digital dietary tracking platforms which replaced manual food logs to enhance their ability to track phenylalanine levels while decreasing patient compliance fluctuations over extended periods.
- BioMarin's pegvaliase adoption discussions in Japan brought attention to enzyme replacement therapies yet regulatory bodies prevented hospitals from adopting these treatments on a large scale.
- AI-driven predictive adherence models now detect high-risk PKU patients earlier, which allows metabolic clinics to start preventive treatment before patients experience severe phenylalanine buildup.
- The funding reforms for rare diseases in Japan resulted in changed reimbursement practices which dedicated more resources to permanent treatment solutions.
- The funding reforms established permanent financial stability for amino acid formula producers which lasted throughout the implementation of their new reimbursement policies.
- The metabolic centers in hospitals developed unified treatment pathways which replaced the previous system of divided outpatient care by providing centralized PKU treatment through specialist-led programs across all major urban areas.
- The gene therapy research partnerships between Japanese research institutes and international biotech companies showed a steady progression toward developing single-treatment PKU curing solutions.
Japan Phenylenketonuria (PKU) Treatment Market Segmentation
By Type :
The Japanese PKU treatment market will use advanced methods to make permanent genetic corrections through gene therapy. Research groups and medical institutions show strong interest in this developing type of research. The development of the project depends on the outcomes of safety tests and the extended patient research results.
The primary treatment method for phenylketonuria will continue to be drug therapy. The treatment controls phenylalanine levels in the bloodstream through persistent medication intake and dietary assistance. Physical therapy helps patients who experience movement and muscle problems because of their metabolic disorder. Stem cell therapy remains in the initial research stage while daily patient management and lifestyle support will continue through existing care methods.
To learn more about this report, Download Free Sample Report
By Application :
The treatment for muscular dystrophy will use similar medical techniques which will help doctors treat patients who experience muscle weakness and ongoing physical deterioration. The rehabilitation program will assist patients in developing their strength while they learn to walk and complete their everyday activities. The implementation of structured therapy programs will lead to improvements in living conditions which will persist throughout the following years. The neuromuscular clinic will provide treatment for patients who experience coordination problems between their nerves and muscles which occur due to metabolic and genetic disorders.
The treatment of genetic disorders will remain essential for managing PKU because it aims to control enzyme levels through dietary management. Clinical trials will enable medical research by evaluating new treatments and enhancing current medical protocols. The organization will use its resources to provide nutritional assistance to patients while conducting their continuous health evaluations.
By End-User :
Hospitals will function as the primary location for patients to receive their initial diagnosis and subsequent treatment of PKU while providing specialized medical staff and emergency treatment services. Clinics will manage ongoing patient check-ups and necessary changes to their prescribed medications. Rehab centers will provide support to patients who require assistance in their physical rehabilitation process and their development of permanent mobility skills.
Research institutes will contribute to the advancement of new medical treatments through their dedicated research work on genetic disorders. Specialty centers will deliver expert care for metabolic disorders and rare diseases through their specialized medical teams. Homecare will continue to grow as patients prefer long-term dietary and medication management at home. The final users of the system will consist of community health organizations and support networks.
By Distribution :
The hospitals will function as main distribution centers for PKU medications and treatment supplies which they will distribute under controlled access that requires medical supervision. The clinics will provide standard prescription medications to patients while they handle ongoing treatment needs through continuous patient assessment and medication dose modifications. Specialty pharmacies will support patients requiring long-term therapy by providing specialized drugs and dietary products.
The pharmacies will establish correct storage procedures and appropriate dosage instructions and maintain a steady supply of products. The different distribution systems will work together to provide continuous treatment to patients while helping to improve disease management throughout Japan.
What are the Key Use Cases Driving the Japan Phenylenketonuria (PKU) Treatment Market?
The primary application which exists within Japan's PKU treatment market operates through lifelong metabolic management which uses medical nutrition therapy methods that include phenylalanine-restricted amino acid formulas as their main component. The process creates its highest demand because newborn screening tests find patients at an early stage who need ongoing dietary treatment to stop their brain development from being harmed which results in them needing lifetime medical care.
The current use of sapropterin dihydrochloride extends to pharmacological responsiveness management particularly in both pediatric metabolic clinics and tertiary hospital environments. Hospitals now use BH4 therapy combined with dietary programs that doctors use to track patients who possess partial enzyme activity. The digital dietary tracking system which supports home monitoring has become popular with outpatient caregivers because it helps them maintain their diet plan when they are away from hospitals.
The clinical trials which are under development test both enzyme substitution therapy and gene-based correction methods that will allow patients to stop following their lifelong dietary restrictions. The integration of AI-driven personalized nutrition planning systems with hospital electronic medical records enables healthcare professionals to obtain real-time metabolic adjustment recommendations to optimize patient treatment. Existing applications have not yet achieved widespread use but they will transform treatment methods when Japan implements precision medicine for rare metabolic disorders.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 122.5 Million |
|
Market size value in 2026 |
USD 131.7 Million |
|
Revenue forecast in 2033 |
USD 218.6 Million |
|
Growth rate |
CAGR of 7.51% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Japan |
|
Key company profiled |
BioMarin, Pfizer, Novartis, Roche, Sanofi, Takeda, Abbott, Nestle Health Science, Mead Johnson, Danone, Cambrooke Therapeutics, Vitaflo, Nutricia, Ajinomoto, Meiji. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Gene Therapy, Drug Therapy, Physical Therapy, Stem Cell Therapy, Supportive Care, Others); By Application (Muscular Dystrophy Treatment, Rehabilitation, Neuromuscular Care, Genetic Disorder Treatment, Clinical Trials, Others); By End-User (Hospitals, Clinics, Rehab Centers, Research Institutes, Specialty Centers, Homecare, Others); By Distribution (Hospitals, Clinics, Specialty Pharmacies, Others). |
Which Regions are Driving the Japan Phenylenketonuria (PKU) Treatment Market Growth?
The Kanto region of Japan controls the PKU treatment market because Tokyo possesses numerous advanced hospitals and metabolic treatment centers. The national children's hospital network together with university research institutions enables medical professionals to conduct early diagnosis and initiate treatment after newborn screening. The treatment of sapropterin-based therapies shows permanent treatment patterns because hospitals show complete reimbursement capacity for all patient groups. The organization maintains its leading position because it operates multiple pharmaceutical headquarters and clinical trial networks while employing specialized doctors.
The Kansai region serves as a reliable market contributor because its two main cities Osaka and Kyoto possess hospital networks that connect with academic medical centers and local healthcare organizations. The Kanto region depends on research for its advantages but Kansai operates through patient care coordination and the implementation of medical procedures in its prefecture hospitals. The presence of pharmaceutical manufacturing facilities together with medical nutrition product supply chains enables healthcare organizations to deliver uninterrupted patient care. The system provides consistent treatment options while decreasing differences between treatment results among patients with metabolic disorders.
The recent establishment of digital health systems and wider availability of neonatal screening services across Kyushu's rural areas make this region the fastest developing area in Japan. Telemedicine services have expanded, which makes it easier for remote PKU patients to connect with specialists who can diagnose their condition more quickly. The governmental support system for rare diseases together with hospital improvement programs has enhanced metabolic treatment resources since the year 2024. The market shift creates new demand for business operators and investors because it generates interest in non-mainstream urban areas that will last until the year 2026 and into 2030.
Who are the Key Players in the Japan Phenylenketonuria (PKU) Treatment Market and How Do They Compete?
The PKU treatment market in Japan presents a partial market consolidation for prescription drugs while showing multiple competing companies in the medical nutrition sector. The market competition depends more on product performance and regulatory approval and capacity for extended dietary assistance than it does on product pricing. Global rare disease companies maintain their market share through establishing early diagnosis systems and building connections with doctors, while specialized metabolic nutrition companies compete by creating better-tasting and more precise product formulations.
Digital adherence technologies that connect nutritional products to patient tracking systems are starting to transform the current market environment. BioMarin Pharmaceutical develops enzymatic and cofactor-based treatments with a focus on sapropterin and pegvaliase development, using technology to create metabolic correction methods that do not require dietary restrictions. Danone Nutricia creates PKU medical foods that offer better taste experience and essential nutrient content, which Japanese hospitals now use for their nutritional treatment programs.
Nestlé Health Science uses Vitaflo to extend its reach through clinical amino acid products and collaborations with metabolic treatment centers, which help them develop relationships with young patients. Ajinomoto uses its domestic production capabilities to create amino acids, which provide hospitals with price advantages and dependable supply chains. Recordati Orphan Europe develops its orphan metabolic disease portfolio through licensing deals and partnerships with Asian rare disease distributors.
Company List
- BioMarin
- Pfizer
- Novartis
- Roche
- Sanofi
- Takeda
- Abbott
- Nestle Health Science
- Mead Johnson
- Danone
- Cambrooke Therapeutics
- Vitaflo
- Nutricia
- Ajinomoto
- Meiji
Recent Development News
In December 2025, PTC Therapeutics announced approval of Sephience™ (sepiapterin). The Japanese Ministry of Health, Labour and Welfare (MHLW) approved Sequence for treating children and adults with phenylketonuria across all disease severities, marking the company’s first product approval in Japan and enabling an upcoming commercial launch after pricing negotiations.
Source: https://www.nasdaq.com/
In 2026, PTC Therapeutics advanced regulatory expansion for its oral PKU therapy Sephience, which was already approved in Europe and under review in Japan. The company’s FDA approval momentum and ongoing Japanese regulatory evaluation support potential market entry in Japan, increasing competition in non-dietary PKU treatment options and accelerating oral enzyme-activity enhancer adoption.
Source: https://www.reuters.com/
What Strategic Insights Define the Future of the Japan Phenylenketonuria (PKU) Treatment Market?
The Japan PKU treatment market is developing new treatment methods which use genetic testing and real-time dietary monitoring to provide continuous metabolic management through digital technologies. The newer treatment procedures now connect three networks which include newborn screening systems and hospital metabolic databases and reimbursement systems that support early and ongoing medical treatment.
Hybrid treatment methods which combine enzyme therapies with medical nutrition and AI-based patient support systems will become the main treatment model in the next 5 to 7 years. The system faces a hidden danger which results from its reliance on a limited number of imported specialty nutrition products and orphan drugs because this dependency creates supply risks and pricing risks when manufacturers change their product lines or experience regulatory setbacks.
Japan presents a new business opportunity through its growing digital health ecosystem for rare diseases which features AI-powered metabolic optimization tools that hospitals use with their electronic medical record systems. Leading pediatric hospitals now begin using these tools after completing their pilot testing. Market participants should prioritize partnerships with hospital networks and digital health developers to embed their solutions directly into clinical decision workflows, ensuring long-term therapeutic stickiness and data access advantage.
Japan Phenylenketonuria (PKU) Treatment Market Report Segmentation
By Type
- Gene Therapy
- Drug Therapy
- Physical Therapy
- Stem Cell Therapy
- Supportive Care
By Application
- Muscular Dystrophy Treatment
- Rehabilitation
- Neuromuscular Care
- Genetic Disorder Treatment
- Clinical Trials
By End-User
- Hospitals
- Clinics
- Rehab Centers
- Research Institutes
- Specialty Centers
- Homecare
By Distribution
- Hospitals
- Clinics
- Specialty Pharmacies
Frequently Asked Questions
Find quick answers to common questions.
The Japan Phenylenketonuria (PKU) Treatment Market size is USD 218.6 Million in 2033.
Key segments for the Japan Phenylenketonuria (PKU) Treatment Market are By Type (Gene Therapy, Drug Therapy, Physical Therapy, Stem Cell Therapy, Supportive Care, Others); By Application (Muscular Dystrophy Treatment, Rehabilitation, Neuromuscular Care, Genetic Disorder Treatment, Clinical Trials, Others); By End-User (Hospitals, Clinics, Rehab Centers, Research Institutes, Specialty Centers, Homecare, Others); By Distribution (Hospitals, Clinics, Specialty Pharmacies, Others).
Major Japan Phenylenketonuria (PKU) Treatment Market players are BioMarin, Pfizer, Novartis, Roche, Sanofi, Takeda, Abbott, Nestle Health Science, Mead Johnson, Danone, Cambrooke Therapeutics, Vitaflo, Nutricia, Ajinomoto, Meiji.
The Japan Phenylenketonuria (PKU) Treatment Market size is USD 122.5 Million in 2025.
The Japan Phenylenketonuria (PKU) Treatment Market CAGR is 7.51% from 2026 to 2033.
- BioMarin
- Pfizer
- Novartis
- Roche
- Sanofi
- Takeda
- Abbott
- Nestle Health Science
- Mead Johnson
- Danone
- Cambrooke Therapeutics
- Vitaflo
- Nutricia
- Ajinomoto
- Meiji
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


