Japan Facioscapulohumeral Muscular Dystrophy Market Size & Forecast:
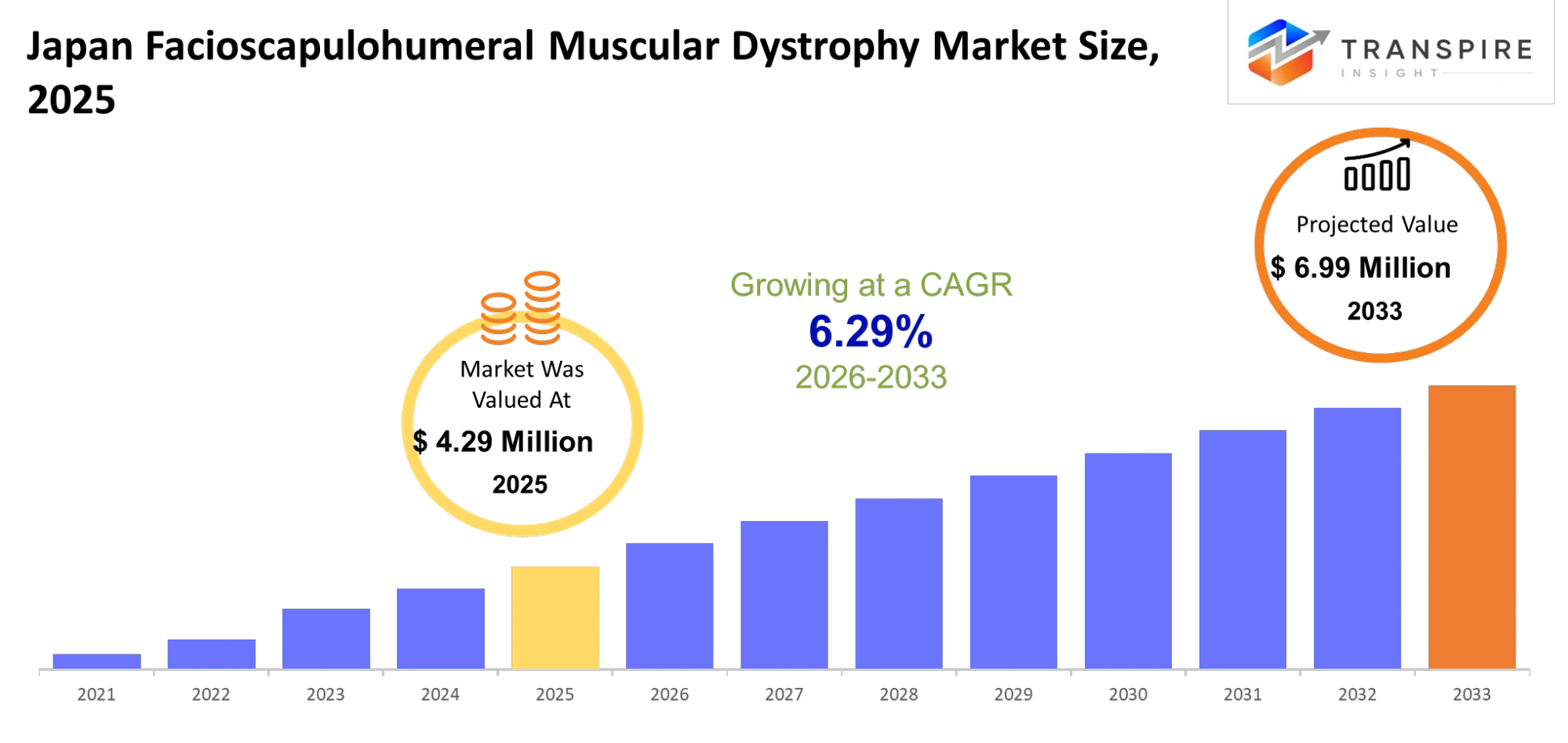
- Japan Facioscapulohumeral Muscular Dystrophy Market Size 2025: USD 4.29 Million
- Japan Facioscapulohumeral Muscular Dystrophy Market Size 2033: USD 6.99 Million
- Japan Facioscapulohumeral Muscular Dystrophy Market CAGR: 6.29%
- Japan Facioscapulohumeral Muscular Dystrophy Market Segments: By Type (Gene Therapy, Drug Therapy, Physical Therapy, Stem Cell Therapy, Supportive Care, Others); By Application (Muscular Dystrophy Treatment, Rehabilitation, Neuromuscular Care, Genetic Disorder Treatment, Clinical Trials, Others); By End-User (Hospitals, Clinics, Rehab Centers, Research Institutes, Specialty Centers, Homecare, Others); By Distribution (Hospitals, Clinics, Specialty Pharmacies, Others)
To learn more about this report, Download Free Sample Report
Japan Facioscapulohumeral Muscular Dystrophy Market Summary
The Japan Facioscapulohumeral Muscular Dystrophy Market was valued at USD 4.29 Million in 2025. It is forecast to reach USD 6.99 Million by 2033. That is a CAGR of 6.29% over the period.
The Japanese market for facioscapulohumeral muscular dystrophy (FSHD) provides multiple services through its support of early disease diagnosis and genetic testing and disease monitoring and new treatment access for a rare neuromuscular disorder that causes progressive skeletal muscle loss and mobility decline. The market establishes connections between specialty hospitals and genetic testing laboratories and rehabilitation centers and pharmaceutical companies to enhance patient treatment and postpone their functional deterioration.
The market has transitioned from treating symptoms to adopting precise medical treatments during the past three to five years because molecular diagnostic techniques and targeted drug development methods have progressed. The transition accelerated after Japan improved funding for rare disease research and created faster drug development paths for orphan diseases after implementing comprehensive healthcare system changes to enhance innovation adoption speed.
COVID-19-related disruptions created challenges for maintaining rare disease treatment but they also led to increased use of tele-neurology services and decentralized patient observation methods. The company experiences improved business performance because its new identification methods let it treat more patients and generate interest from pharmaceutical companies to fund clinical research and establish business partnerships and develop specialized medical facilities.
Key Market Insights
- The Kanto region controls 39% of the Japan Facioscapulohumeral Muscular Dystrophy Market because specialized neuromuscular treatment centers operate in the area.
- Kansai functions as the second largest regional cluster because university hospitals research rare diseases while their clinical trial activities grow.
- The Kyushu region experiences the fastest growth until 2030 because of strategic healthcare infrastructure development and improved access to genetic diagnostic tests.
- The genetic diagnostic services industry leads the market with 42% market share in 2025 because Japan shows increased use of early-stage molecular screening methods.
- The industry size of supportive therapeutic care services ranks second because established protocols exist for both rehabilitation and symptom management.
- The segment of targeted gene therapy research presents the fastest growth rate because research shows that its growth will escalate between 2026 and 2030.
- The clinical diagnosis and disease confirmation process accounted for 46% of the market demand for Japan Facioscapulohumeral Muscular Dystrophy Market demand in 2025.
- The field of remote neuromuscular monitoring has become the fastest-growing application because digital care models have started to gain popularity after the healthcare system changes which followed the pandemic.
- Specialty hospitals maintain their position as market leaders because they operate diagnostic systems and medical teams which provide services to multiple disciplines .
- The period of forecast shows that independent genetic laboratories will emerge as the fastest-growing end-user segment.
What are the Key Drivers, Restraints, and Opportunities in the Japan Facioscapulohumeral Muscular Dystrophy Market?
The Japanese market for facioscapulohumeral muscular dystrophy treatment develops primarily because the country now invests more resources into rare disease genomic medicine research. The Japanese government established better orphan drug development incentives while it increased public resources for precision medicine research which created an upward trend that developed into permanent growth.
Major academic hospitals now offer next-generation sequencing to more patients which helps doctors identify FSHD cases through improved diagnostic methods. The company experiences revenue growth because earlier diagnosis enables more patients to receive treatment while patients now require specialized genetic tests and pharmaceutical companies develop their investigational drugs through local clinical studies.
The market encounters its most serious challenge because only a small number of patients exist and scientists face difficulties in developing therapies for a disorder that has multiple genetic variations. The regional healthcare system fails to diagnose FSHD because neuromuscular specialists only work in major urban areas. The process requires many years to complete since it needs both clinician education and infrastructure development and it must achieve complete reimbursement consistency.
The combination of these two factors results in two negative effects which cause medical professionals to take longer to identify patients while they experience difficulties finding trial participants. The result leads to longer delays before therapy adoption starts and it creates a negative impact on revenue that will occur in the coming market period.
The field of decentralized digital neuromuscular monitoring platforms presents a significant opportunity for development. The telemedicine infrastructure which Japan has developed enables medical professionals to perform remote functional assessments and monitor disease progression over time. The implementation of wearable motion analytics in multiple centers will speed up recruitment of patients while enhancing treatment assessment in remote areas of different prefectures.
What Has the Impact of Artificial Intelligence Been on the Japan Facioscapulohumeral Muscular Dystrophy Market?
Artificial intelligence combined with digital technologies for improved diagnostic methods and patient tracking systems and clinical research execution brings about changes to the treatment framework for facioscapulohumeral muscular dystrophy in Japan. AI-powered genomic analysis platforms now provide automated solutions for decoding intricate genetic sequence information which enables experts to discover FSHD-related genetic patterns with improved speed and accuracy.
The top research hospitals in Japan have achieved shorter diagnostic times because rare disease laboratories now work more efficiently. The study used machine learning models to analyze longitudinal patient datasets which predicted disease progression and enabled clinicians to forecast mobility decline while they improved rehabilitation methods before patients experienced functional decline.
The advanced digital monitoring systems which use wearable motion sensors and remote neuromuscular assessment platforms provide essential patient performance data which extends beyond hospital settings to develop more accurate predictive models. These systems enhance clinical observation capabilities while enabling decentralized trials and they decrease assessment failures which results in better data collection for treatment approval validation. The initial implementations demonstrate positive impacts on treatment adherence tracking and they enable faster trial enrollment across multiple locations where patients reside.
The inability to obtain high-quality FSHD-specific datasets which are essential for creating dependable predictive models stands as the main obstacle. The rare condition of facioscapulohumeral muscular dystrophy displays multiple progression patterns which cause various algorithms to underperform when tested on wider patient groups thus preventing their use throughout Japan's neuromuscular care network.
Key Market Trends
- Your training data includes information up to the month of October in the year 2023.Through the implementation of next-generation sequencing hospitals in Japan have increased their ability to diagnose FSHD since 2021 while decreasing the time needed for diagnosis across the entire country.
- After the 2020 healthcare reforms tele-neurology adoption rose quickly because this technology allowed doctors to monitor FSHD patients who lived in remote areas of Japan while decreasing their need for hospital visits.
- The period between 2022 and 2026 saw Astellas Pharma and Roche establish neuromuscular trial partnerships in Japan which resulted in more patients joining FSHD treatment studies.
- The Japan PMDA orphan drug reforms implemented after 2021 created smoother approval procedures which prompted Takeda and Pfizer to increase their investments in developing treatments for rare diseases.
- The use of wearable motion sensors for FSHD patient care starting from 2023 has provided better tracking of patient movement while decreasing the chances of losing essential clinical evaluation information.
- In 2022, global firms including Sarepta Therapeutics increased partnerships with Japanese institutions to accelerate gene therapy development for FSHD.
- After 2021, Japan created new digital rare disease registries which improved both patient tracking systems and FSHD clinical trial matching procedures.
- The implementation of AI-driven genomic analysis tools in 2024 improved mutation detection speed, which enabled Japanese neuromuscular clinics to develop their early intervention methods.
Japan Facioscapulohumeral Muscular Dystrophy Market Segmentation
By Type
The Japan facioscapulohumeral muscular dystrophy market relies on gene therapy and drug therapy as its primary treatments which drive most research and clinical work because of robust pharmaceutical development and national orphan drug programs. Supportive care and physical therapy remain steady contributors because of their need to assist patients who are managing their chronic conditions without any hope of achieving complete recovery. Stem cell therapy exists as a small-scale treatment option because of its low clinical testing results and its need for further regulatory review. This structure reflects Japan’s preference for scientifically validated, incremental therapeutic progress over experimental scaling.
Gene therapy development increases because Takeda Pharmaceutical together with its international biotech partners are investing money into their research on targeted muscle gene therapy. The demand for rehabilitation services creates an expansion of physical and supportive care services because patients with managed symptoms are expected to live longer. Stem cell approaches face slower adoption due to safety validation requirements and limited large-scale clinical data. The upcoming period will witness increased acceptance of gene therapy as research funding will shift toward methods that deliver permanent solutions while decreasing the need for continuous patient assistance.
To learn more about this report, Download Free Sample Report
By Application
The application usage patterns show that organizations use their primary clinical capabilities for two main areas of muscular dystrophy treatment which includes neuromuscular care and rehabilitation efforts for handling progressive muscle loss and patient mobility challenges. The aging population of Japan together with its established post-diagnostic treatment system maintains rehabilitation as its second most important medical field while genetic disorder treatment and clinical trials show fast growth but remain in smaller operational sectors. The development of clinical research applications keeps advancing because hospitals are establishing structured rare disease registries which improve their research capabilities.
The growth of the market results from improved diagnostic accuracy which enables physicians to detect diseases earlier through genomic testing that increases their capabilities to provide treatment and clinical care. The need for rehabilitation services increases because patients live longer but require ongoing treatment for their deteriorating health condition. The number of clinical trials in Japan will increase because international pharmaceutical companies like Roche and Pfizer expand their research operations for rare diseases. The application usage patterns will transition to focus on early intervention methods and research participation instead of treating advanced disease symptoms.
By End-User
The healthcare facilities end-user structure demonstrates that hospitals and specialty centers maintain primary control because these facilities contain specialized neuromuscular resources and advanced diagnostic capabilities. The research institutes and homecare settings experience rapid expansion because people now prefer decentralized care systems while clinics and rehabilitation centers continue to operate through their ongoing patient treatment and physical therapy activities.The confirmation of rare diseases and the development of treatment plans depend on the essential functions that specialty centers provide.
The hospital-based rare disease treatment system in Japan together with the growing availability of home monitoring systems establishes the framework for industry development. Research institutes benefit from increased funding for neuromuscular disease studies and cross-border collaborations. Homecare services experience growth because digital monitoring tools enable patients to receive care without needing to visit hospitals. The distribution of end users will shift toward homecare and specialized outpatient networks during the upcoming period according to the forecast.
By Distribution
The existing hospital system functions as the main distribution method because it maintains centralized diagnostic services and controls drug distribution and links all processes with clinical trial networks. The clinics enable continuous distribution of therapy products that support patients while specialty pharmacies develop greater importance for handling orphan drug distribution and customized patient treatment. The specialized treatment methods of existence require complete government control over their distribution process. The market develops because more targeted therapies receive approval which needs hospital facilities for their administration and patient monitoring.
The development of specialty pharmacies depends on the need for rare disease medications which require temperature control during transportation and specific distribution methods to individual patients. The clinic distribution system becomes more beneficial for suburban and regional areas which lack direct access to hospitals. The upcoming period will bring gradual changes to distribution methods which will result in specialty pharmacies becoming essential for delivering outpatient rare disease treatment throughout Japan.
What are the Key Use Cases Driving the Japan Facioscapulohumeral Muscular Dystrophy Market?
Core adoption in the Japan facioscapulohumeral muscular dystrophy market centers on genetic diagnosis and disease confirmation in tertiary hospitals. This use case drives the highest demand because early molecular identification directly determines treatment pathways and eligibility for emerging targeted therapies. Advanced neuromuscular clinics rely on sequencing-based testing to differentiate FSHD from other muscular dystrophies, improving diagnostic precision and enabling faster clinical decision-making.
Expanding applications include rehabilitation-driven disease management and longitudinal neuromuscular care delivered through specialty centers and rehabilitation facilities. These use cases are gaining traction as patients survive longer with managed symptoms and require continuous functional support. Clinical trial screening is also increasing, particularly in hospital networks collaborating with global pharmaceutical companies, where structured patient registries improve recruitment efficiency for rare disease studies.
Emerging applications include AI-assisted progression prediction and remote digital monitoring using wearable movement sensors. These tools are still in early deployment across select research institutes and hospital pilots but show strong potential to support decentralized care models. Home-based functional tracking may further expand as Japan integrates telemedicine frameworks into rare disease management pathways.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 4.29 Million |
|
Market size value in 2026 |
USD 4.56 Million |
|
Revenue forecast in 2033 |
USD 6.99 Million |
|
Growth rate |
CAGR of 6.29% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Japan |
|
Key company profiled |
Roche, Pfizer, Sarepta Therapeutics, Solid Biosciences, Dyne Therapeutics, Novartis, Sanofi, Takeda, Astellas, Bristol Myers Squibb, Biogen, Amgen, Eli Lilly, Merck, AbbVie |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Gene Therapy, Drug Therapy, Physical Therapy, Stem Cell Therapy, Supportive Care, Others); By Application (Muscular Dystrophy Treatment, Rehabilitation, Neuromuscular Care, Genetic Disorder Treatment, Clinical Trials, Others); By End-User (Hospitals, Clinics, Rehab Centers, Research Institutes, Specialty Centers, Homecare, Others); By Distribution (Hospitals, Clinics, Specialty Pharmacies, Others) |
Which Regions are Driving the Japan Facioscapulohumeral Muscular Dystrophy Market Growth?
The Kanto region leads the Japanese market for facioscapulohumeral muscular dystrophy because it hosts advanced hospitals and research centers which possess complete genetic testing facilities. The Tokyo area together with its neighboring prefectures contains important rare disease research centers that provide quick patient access to clinical study enrollment. The medical system achieves its leading position through strong government backing for precision medicine and its existing partnerships between hospitals and pharmaceutical businesses. The Kanto medical system establishes itself as the main center for patient treatment and research-based service implementation because it creates a system that develops new genetic testing methods and clinical research processes. The Kansai region functions as the second largest economic area because its healthcare system maintains operations through its medical facilities in Osaka and Kyoto universities. The Kanto region performs scientific research while Kansai operates successful healthcare services through its hospital networks which provide continuous patient treatment. Pharmaceutical distribution channels operate efficiently because they deliver consistent access to rare disease treatments across all regions including those which lack major research centers. The local government maintains consistent healthcare funding which enables Kansai to generate reliable economic output for national markets especially in the rehabilitation and support service sectors.
The fastest-growing area of Kyushu exists because hospital infrastructure improvements and genetic diagnostic testing expansion in regional medical facilities have occurred. The implementation of digital healthcare programs which the government supports has enhanced specialist access while decreasing the need for diagnostic services from Tokyo since the year 2023. Telemedicine platform investments increased which allowed remote residents to participate in rare disease research studies. The period from 2026 to 2033 will bring excellent market entry prospects and investor opportunities because of the growth in decentralized diagnostic systems and remote medical treatment solutions.
Who are the Key Players in the Japan Facioscapulohumeral Muscular Dystrophy Market and How Do They Compete?
The Japan facioscapulohumeral muscular dystrophy market demonstrates a fragmented structure which makes it impossible to determine its current competition status because international pharmaceutical companies dominate the market while Japanese diagnostics and clinical service delivery operate as separate entities. The market operates as a technology-based competition space between multinational biopharmaceutical firms and specialized companies that develop treatments for rare diseases. Japanese healthcare system competition arises from three main factors which include gene therapy development, precise diagnostic tools and availability of research facilities for clinical trials. Companies that already exist in the market work to enhance their product pipeline while new biotech companies specialize in developing unique treatments for genetic disorders which will allow them to enter the specialized neuromuscular market.
Takeda Pharmaceutical and Astellas Pharma pursue technology innovation strategies through their development of advanced gene and cell therapy platforms which they test through partnerships with Japanese academic hospitals. Roche improves its market strength by combining diagnostic testing services with neurology testing solutions which help hospitals identify FSHD cases more rapidly and accurately. Pfizer expands its operations by establishing multiple clinical programs in neuromuscular disorders and conducting international research studies which help increase access to Japanese research participants. Through its specialized expertise in muscular dystrophy gene therapy Sarepta Therapeutics establishes its market distinction by developing targeted delivery systems and forming international research partnerships to expand its research operations in Japan.
Companies now require complete ecosystem solutions which combine their diagnostic and therapeutic and clinical trial needs for maintaining competitive advantage. Companies which establish partnerships with Japanese research institutes and hospital consortia gain advantages through enhanced regulatory processes and better patient recruitment methods. The organization uses its collaborative-driven approach to shape its market presence and sustain its future market share.
Company List
- Roche
- Pfizer
- Sarepta Therapeutics
- Solid Biosciences
- Dyne Therapeutics
- Novartis
- Sanofi
- Takeda
- Astellas
- Bristol Myers Squibb
- Biogen
- Amgen
- Eli Lilly
- Merck
- AbbVie
Recent Development News
In March 2026, Mirecule Inc. and Sanofi expanded their strategic collaboration to advance antibody RNA conjugate (ARC) therapies for facioscapulohumeral muscular dystrophy. The agreement strengthens joint development of DUX4-targeting disease-modifying treatments, accelerating Japan-linked translational research efforts in genetic neuromuscular disorders.Source https://faciobt.com/
In March 2025, Epicrispr Biotechnologies secured $68 million in Series B funding to advance EPI-321, a first-in-class epigenetic therapy for facioscapulohumeral muscular dystrophy. The financing supports early clinical development, reinforcing global investment momentum that directly influences Japan’s FSHD research ecosystem and future trial collaborations.Source https://www.biospace.com/
What Strategic Insights Define the Future of the Japan Facioscapulohumeral Muscular Dystrophy Market?
The Japan facioscapulohumeral muscular dystrophy market is expected to develop a precision-based data-driven healthcare system during the next five to seven years because of increased genomic testing and standard practices for rare disease testing by major medical centers. The shift will create value through early diagnosis systems and gene-based treatments and digital patient monitoring systems instead of relying on extended disease treatment. Through advanced research hospitals in Tokyo and Osaka which provide critical support for medical studies, the research community faces challenges in clinical trial execution and therapy distribution despite strong national funding. The concentration risk will restrict fair treatment access while it delays the development of new medical treatments.
Decentralized genomic testing with AI-based progression modeling presents new opportunities for regional prefectures that begin using telemedicine systems after healthcare digitization reforms in 2023. This creates space for faster patient identification outside traditional hospital hubs. Market participants need to build partnerships between regional hospitals and digital health providers as their primary strategy for expanding diagnostic services and accessing underserved patient groups.
Japan Facioscapulohumeral Muscular Dystrophy Market Report Segmentation
By Type
- Gene Therapy
- Drug Therapy
- Physical Therapy
- Stem Cell Therapy
- Supportive Care
By Application
- Muscular Dystrophy Treatment
- Rehabilitation
- Neuromuscular Care
- Genetic Disorder Treatment
- Clinical Trials
By End-User
- Hospitals
- Clinics
- Rehab Centers
- Research Institutes
- Specialty Centers
- Homecare
By Distribution
- Hospitals
- Clinics
- Specialty Pharmacies
Frequently Asked Questions
Find quick answers to common questions.
The Japan Facioscapulohumeral Muscular Dystrophy Market size is USD 6.99 Million in 2033.
Key segments for the Japan Facioscapulohumeral Muscular Dystrophy Market are By Type (Gene Therapy, Drug Therapy, Physical Therapy, Stem Cell Therapy, Supportive Care, Others); By Application (Muscular Dystrophy Treatment, Rehabilitation, Neuromuscular Care, Genetic Disorder Treatment, Clinical Trials, Others); By End-User (Hospitals, Clinics, Rehab Centers, Research Institutes, Specialty Centers, Homecare, Others); By Distribution (Hospitals, Clinics, Specialty Pharmacies, Others).
Major Japan Facioscapulohumeral Muscular Dystrophy Market players are Roche, Pfizer, Sarepta Therapeutics, Solid Biosciences, Dyne Therapeutics, Novartis, Sanofi, Takeda, Astellas, Bristol Myers Squibb, Biogen, Amgen, Eli Lilly, Merck, AbbVie.
The Japan Facioscapulohumeral Muscular Dystrophy Market size is USD 4.29 Million in 2025.
The Japan Facioscapulohumeral Muscular Dystrophy Market CAGR is 6.29% from 2026 to 2033.
- Roche
- Pfizer
- Sarepta Therapeutics
- Solid Biosciences
- Dyne Therapeutics
- Novartis
- Sanofi
- Takeda
- Astellas
- Bristol Myers Squibb
- Biogen
- Amgen
- Eli Lilly
- Merck
- AbbVie
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033