Europe Asthma Drugs Market Size & Forecast:
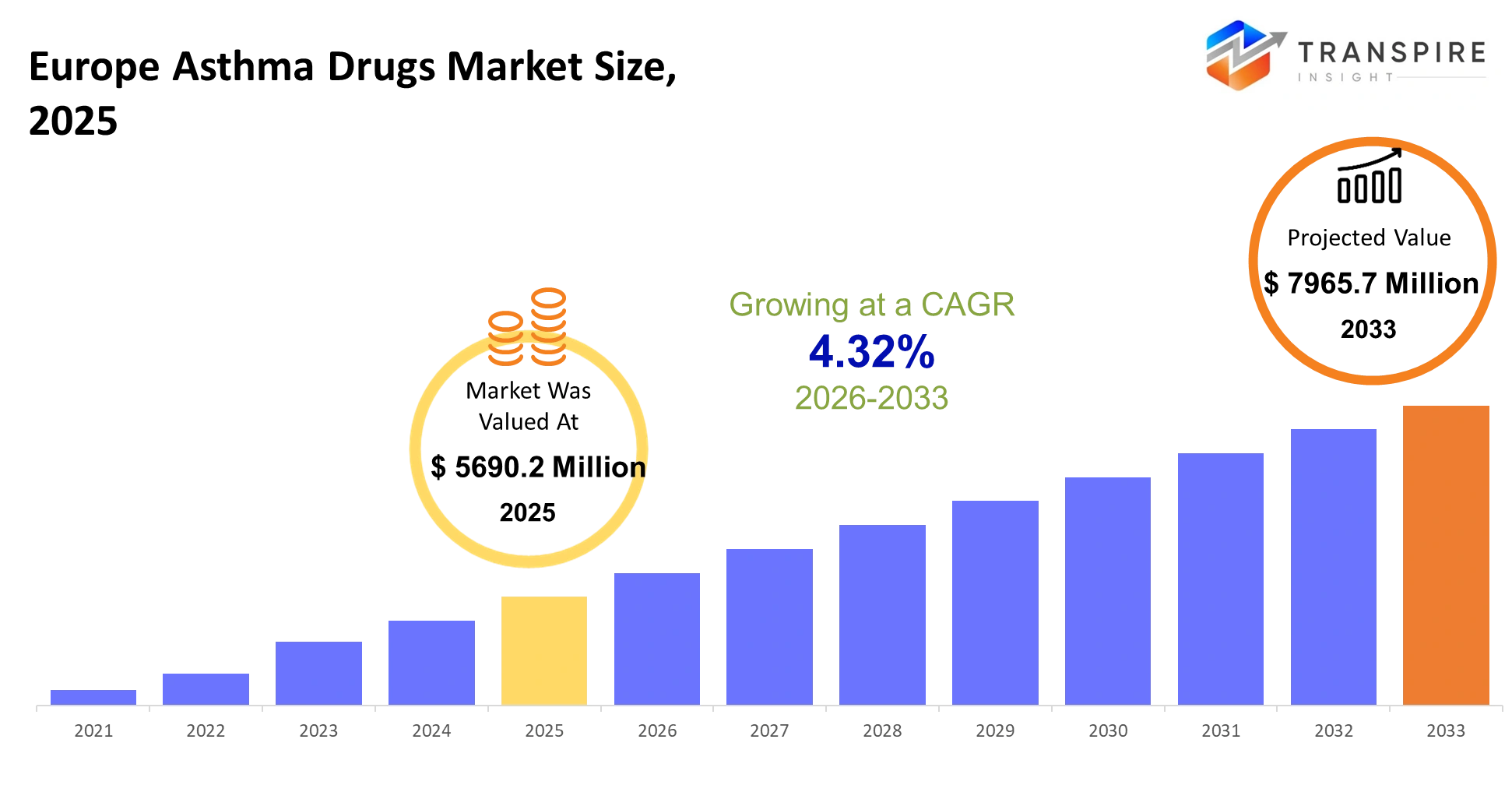
- Europe Asthma Drugs Market Size 2025: USD 5690.2 Million
- Europe Asthma Drugs Market Size 2033: USD 7965.7 Million
- Europe Asthma Drugs Market CAGR: 4.32%
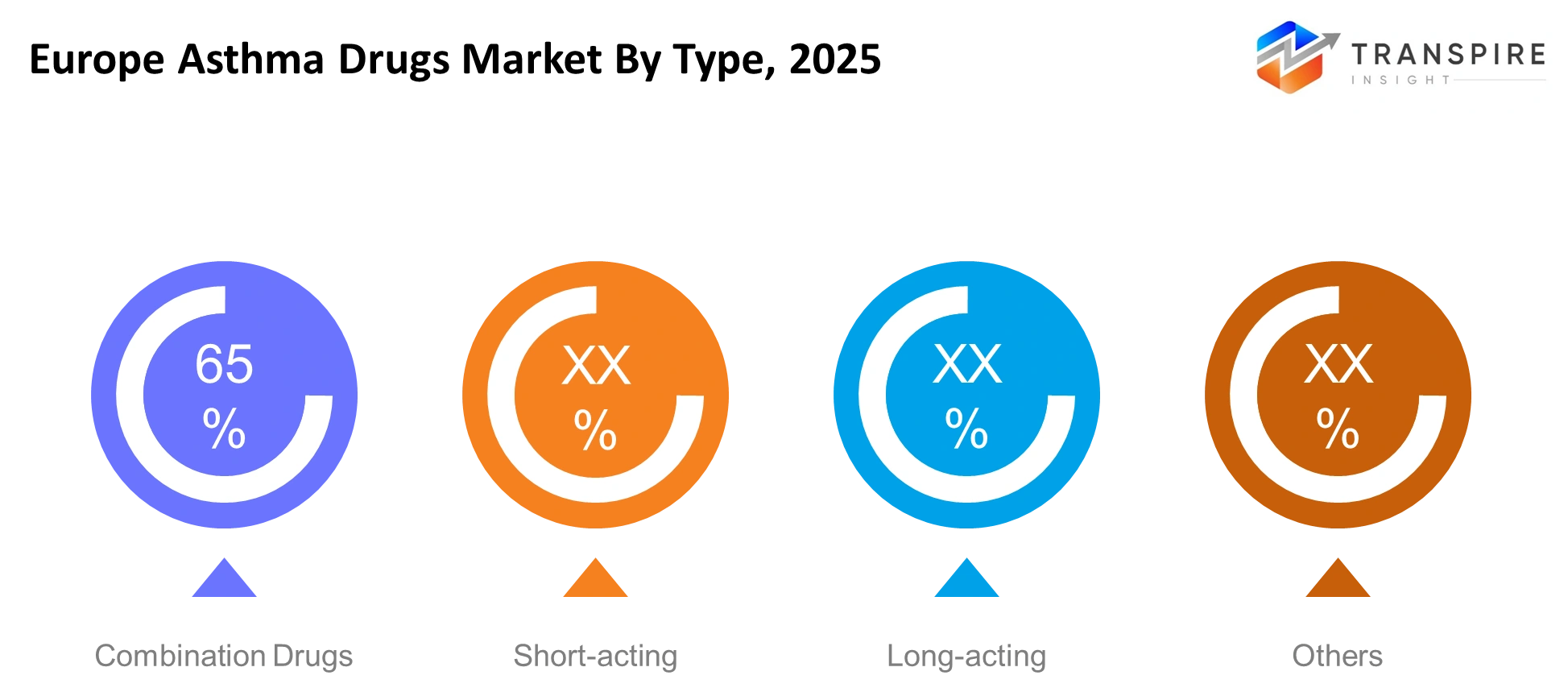
- Europe Asthma Drugs Market Segments: By Type (Short-acting, Long-acting, Combination Drugs, Others), By Application (Acute Treatment, Maintenance Therapy, Others), By End-User (Hospitals, Clinics, Pharmacies, Others), By Route (Inhalation, Oral, Injectable, Others).
To learn more about this report, Download Free Sample Report
Europe Asthma Drugs Market Summary:
The Europe Asthma Drugs Market size is estimated at USD 5690.2 Million in 2025 and is anticipated to reach USD 7965.7 Million by 2033, growing at a CAGR of 4.32% from 2026 to 2033. The Europe asthma drugs market exists to keep patients out of emergency rooms and maintain day-to-day lung function, enabling consistent participation in work, school, and physical activity. The system provides long-term disease management through inhalers and biologics and combination therapies that decrease patient flare-ups and hospital visits.
A structural shift toward targeted biologics and personalized treatment pathways has transformed prescribing practices for severe asthma cases during the past three to five years. The transition shows improved diagnostic segmentation together with health insurance companies' readiness to cover expensive treatments that stop serious health problems from happening. The COVID-19 pandemic created a major turning point because it revealed how people with respiratory problems were at risk which led healthcare systems to focus on treating chronic airway diseases through remote monitoring.
Healthcare organizations currently experience growth through their implementation of treatment methods which enable them to start patient care earlier while making use of advanced medical solutions.
Key Market Insights
- Western Europe controls almost 45% of the European asthma medication market in 2025 because its healthcare systems are highly developed.
- Germany France and the UK together lead revenue production through their effective reimbursement systems and their early adoption of biologic treatments.
- Eastern Europe shows the fastest growth rate because its respiratory care services become more accessible throughout the period until 2030 at a compound annual growth rate higher than 6 percent.
- Inhaled corticosteroids dominate the market because they will hold a 40 percent share in 2025 which establishes their function as essential components of long-term asthma control treatment.
- The second-largest market share belongs to combination inhalers which gain popularity because they help patients use their medications and provide two different treatment methods.
- Biologics represent the fastest-growing segment which is projected to grow above 8 percent annually through 2030 because targeted treatments deliver better medical results.
- Chronic asthma management which requires lifelong treatment and therapy adherence, accounts for more than 65% of all applications.
- The treatment of severe asthma shows the highest growth rate because more doctors begin using monoclonal antibody therapies together with precision diagnostic tools.
- Hospitals drive about 50% of total market demand for 2025 because their doctors write prescriptions that lead to advanced medical treatments.
- The fastest-growing market segment belongs to specialty clinics, which expand quickly because they use outpatient care models together with customized treatment methods.
What are the Key Drivers, Restraints, and Opportunities in the Europe Asthma Drugs Market?
The Europe Asthma Drugs Market experiences its primary growth engine through the increasing use of biologic treatments for severe asthma which started after major EU countries began implementing new clinical guidelines and reimbursement policies in 2020. Health systems increasingly support high-cost monoclonal antibodies because they reduce hospital admissions and emergency interventions. The cost-offset system benefits severe asthma patient treatment through two mechanisms which lead to higher revenue generation from premium therapies causing market value per patient to increase instead of base patient count.
The most significant structural barrier remains the high cost and restricted access to advanced biologics. The therapies need specialist diagnosis which includes biomarker testing and biomarker testing plus strict reimbursement rules that control patient access to their benefits. Public healthcare budgets in parts of Southern and Eastern Europe cannot absorb widespread biologic use, creating uneven access across regions. The constraint prevents eligible patients from receiving timely treatment while it maintains a broad segment of the population on less effective therapies that cost less.
Digital inhalers and connected respiratory care platforms provide a key opportunity which organizations can use to advance their operations. The two technologies in this system offer health professionals the ability to monitor patient behavior in real time while they provide intervention solutions, which lead to better results among patients with moderate asthma. The UK and Germany have begun testing digital therapeutics reimbursement, which will help these technologies achieve greater market acceptance. The combination of drug-device systems will lead to extended treatment periods while new revenue sources will begin to generate recurring income.
What Has the Impact of Artificial Intelligence Been on the Europe Asthma Drugs Market?
The combination of artificial intelligence and advanced digital technologies is starting to transform respiratory drug management throughout Europe because these technologies improve therapy delivery and monitoring and optimization processes. The integration of AI-driven platforms into connected inhalers enables automatic tracking of patient adherence while the system analyzes their dosage patterns. The systems notify doctors about patient medication schedule violations because this information helps them take action that decreases the possibility of severe health problems.
Machine learning models use real-world patient data to create predictive care systems which analyze both inhaler usage and environmental triggers and historical flare-up events. The models have the ability to predict asthma attacks up to two days before their occurrence which enables doctors to modify treatment strategies without delay. Pharmaceutical companies and healthcare providers use these insights to optimize therapy effectiveness and reduce hospital admissions which directly improves cost efficiency and patient outcomes.
Digital health integration has produced observable operational advantages which include a 15 to 25 percent increase in medication adherence and a decrease in emergency visits among monitored populations. The extended treatment periods and increased revenue stability from advanced therapies create financial advantages for healthcare providers.
Data fragmentation across healthcare systems represents a major obstacle which needs to be addressed. The process of large-scale AI model training faces obstacles because electronic health record systems lack consistent integration and privacy regulations create limitations.
Key Market Trends
- European treatment guidelines established after 2020 prescribed biologics as the primary treatment for severe asthma cases which resulted in a new trend towards using specified medications that provided increased treatment benefits for individual patients.
- Post-pandemic, physicians accelerated early intervention strategies which included faster patient transitions from monotherapy inhalers to combination therapies that would decrease their hospitalization risks.
- The period between 2021 and 2025 saw smart inhalers reach a 20% adoption increase which marked a transition towards respiratory care methods that used adherence tracking and data analytics.
- The reimbursement policies which existing in Germany and the UK now provide coverage for high-cost biologics, which results in better access for patients who require these medications and helps drug manufacturers to increase their market share of expensive medications.
- AstraZeneca and GlaxoSmithKline have emerged as pharmaceutical industry leaders who now allocate more resources to their monoclonal antibody development programs which will transform their market competition strategies.
- Hospital-based asthma treatment practices have evolved into specialized asthma clinics which provide outpatient services because hospitals face budget constraints while needing to track patient progress.
- Digital health systems which health systems implemented between 2022 and 2026, led to a 25% increase in medication adherence which resulted in payers supporting drug-device systems that connect medications with their accompanying devices.
- Eastern Europe saw its advanced therapy access improve after 2021 because of health system funding changes and international pharmaceutical companies who entered the region.
- The increased pollution levels in urban areas prompted environmental organizations to demand that health authorities include preventive respiratory care in their comprehensive public health strategies.
Europe Asthma Drugs Market Segmentation
By Type
The two types of long-acting and combination drugs present themselves as the leading treatment options because their effectiveness enables patients to stay compliant with their treatment, while their ability to maintain symptom control over extended periods. Healthcare providers prefer combination inhalers that combine corticosteroids with long-acting bronchodilators because these devices simplify treatment methods and decrease the occurrence of breathing problems. The medical community uses short-acting medications to provide immediate relief from symptoms, but the need for these drugs has decreased because treatment protocols now recommend preventive methods instead of emergency solutions.
The market for biologic therapies represents its fastest expansion because these treatments target specific inflammatory pathways which exist in severe asthma cases. The segment will expand when reimbursement policies improve and diagnostic tools become more accurate. The future manufacturing process will focus on delivering two product categories, which include biologic medications that deliver high therapeutic value and combination treatments that will help medical providers treat complex patient needs.
To learn more about this report, Download Free Sample Report
By Application
The therapy method which doctors use to treat patients with asthma who need ongoing treatment to control their symptoms. Doctors increasingly provide patients with long-term control medications because these drugs help protect against disease outbreaks while generating steady income for their practices. The medical field requires both urgent care and essential services, but emergency departments need to decrease their usage of urgent care services.
The field of severe asthma treatment is experiencing rapid growth because doctors are using biologic drugs and personalized treatment methods to treat their patients. The shift results from people wanting to control their diseases before they need emergency treatment. The upcoming years will see investment funding for therapies which extend treatment effects and decrease costs for healthcare providers, which will support maintenance treatment methods.
By End-User
The hospital system maintains its top position as healthcare providers because it offers doctors access to specialized doctors and advanced medical equipment and advanced treatment methods which include biologic drugs. The expansion of outpatient care models together with healthcare systems quest for budget control has enabled clinics to achieve rapid growth. The pharmacies function as essential providers which distribute maintenance drugs to patients while offering them multiple chances to follow their medication schedule. The development of specialized respiratory centers has established new centers which provide customized asthma treatment especially for patients with severe asthma conditions.
The increasing number of non-hospital facilities indicates a societal movement towards decentralized medical treatment which includes ongoing patient observation. The trend motivates manufacturers to create products and distribution methods which match outpatient medical treatment requirements while investors are likely to concentrate on business models that combine therapy with patient observation and continuous healthcare delivery.
By Route
Inhalation remains the most common treatment method because it delivers medication directly to the lungs which leads to faster treatment effects and produces fewer side effects throughout the body. Patients receive oral therapies as second-line treatments because these medications only benefit mild cases and cannot match the effectiveness of inhaled treatments. The fastest-growing treatment method depends on injectable biological therapies because these medications focus on treating severe asthma through extended dosing times.
The accessibility of injectables depends on two factors: first, they require doctor supervision and second, they need insurance companies to provide financial backing which establishes different access levels throughout different areas. The development of inhaler technology together with drug formulation improvements has resulted in better user experience and treatment compliance. Future research will direct its efforts towards developing digital monitoring systems that work together with medication delivery systems to improve inhalation methods and enhance the distribution of new injectable drug delivery systems.
What are the Key Use Cases Driving the Europe Asthma Drugs Market?
The main use case investigates how maintenance therapies help patients with asthma to achieve better disease control in outpatient settings while preventing hospital admissions which would incur high costs. Physicians select combination inhalers and corticosteroids as their preferred treatment option because these medications help to reduce exacerbation instances while allowing patients to maintain their daily activities. The application generates its highest user demand because asthma requires ongoing management which needs continuous treatment instead of occasional medical care.
The new use cases enable severe asthma management through specialist clinics and hospitals which now use biologic treatments for patients who do not respond to conventional therapies. The field of pediatric asthma treatment is expanding because customized dosing and inhaler design improvements help young patients use their treatment better.
The new use cases investigate digital systems that support respiratory treatment through inhaler devices which monitor patient usage and allow healthcare providers to start treatment before emergencies happen. Healthcare organizations now adopt remote monitoring systems together with AI technologies that predict patient risks in their medical operations at advanced healthcare facilities.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 5690.2 Million |
|
Market size value in 2026 |
USD 5923.3 Million |
|
Revenue forecast in 2033 |
USD 7965.7 Million |
|
Growth rate |
CAGR of 4.32% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain and Rest of Europe) |
|
Key company profiled |
GSK, AstraZeneca, Novartis, Sanofi, Roche, Teva, Merck, Pfizer, Boehringer Ingelheim, Amgen, Regeneron, AbbVie, Bayer, Mylan, Cipla |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Short-acting, Long-acting, Combination Drugs, Others), By Application (Acute Treatment, Maintenance Therapy, Others), By End-User (Hospitals, Clinics, Pharmacies, Others), By Route (Inhalation, Oral, Injectable, Others) |
Which Regions are Driving the Europe Asthma Drugs Market Growth?
The market is dominated by Western Europe because of its strong reimbursement systems and early adoption of advanced respiratory treatments. The three nations Germany, the UK and France implement structured clinical guidelines which promote quick use of combination drugs and biologics. The medical system includes numerous specialists and diagnostic centers and hospital networks which enable precise disease identification and precise medical solutions. The integrated healthcare system maintains high patient treatment success rates while generating steady income for the system.
Northern Europe implements a stability-based economic model which results from its citizens having access to universal healthcare and doctors using controlled medication distribution methods. The Swedish and Danish healthcare systems focus on maintaining continuous disease management together with preventive health solutions which drives the need for ongoing maintenance treatment. The region possesses advanced digital health systems which enable extensive remote patient monitoring and treatment compliance assessment. The market environment establishes predictable conditions which remain stable when compared to the high-cost market for innovative solutions found in Western Europe.
Healthcare systems in Eastern Europe expand parent asthma treatment access to modern treatments which makes this region the fastest expanding market. The recent policy changes together with increased EU funding for healthcare have established better reimbursement systems which facilitate advanced therapy accessibility. Multinational pharmaceutical companies have accelerated their regional growth by establishing partnerships and building distribution networks. The market shift between the years 2026 to 2033 creates new business possibilities which will benefit companies that serve underprivileged patient groups who will soon receive conventional medical treatment.
Who are the Key Players in the Europe Asthma Drugs Market and How Do They Compete?
The European asthma medicine market shows moderate market concentration because several major pharmaceutical companies control valuable market segments while generic drug manufacturers battle for market share in established treatments through price competition. Companies which operate in the market currently use new product development to protect their market share, especially through their development of biologic drugs and combination inhalers, instead of depending on price changes. The new competition framework requires organizations to establish their market position through measurable clinical results and patient treatment compliance and their capacity to prove healthcare system cost reductions across extended periods.
The company AstraZeneca develops new biologic treatments, which create medicines that interrupt specific inflammatory pathways found in severe asthma cases. The company uses its precise method to create distinct products, which enables them to charge high prices backed by their excellent clinical results. GlaxoSmithKline maintains its market dominance in maintenance therapies through its expertise in inhaler technology, which enables the company to create new drug delivery systems that enhance its existing respiratory product range. The two companies use clinical research and partnerships to boost their biologic research programs.
Sanofi develops immunology-based therapies that treat patients suffering from allergic diseases with its biologic treatments. Novartis establishes its market presence through specific treatment options, which it supports with its global research capabilities and extensive specialty healthcare services. Teva Pharmaceuticals operates in the European market through its generic inhalers, which it sells at lower prices to customers who choose economical options by leveraging its manufacturing power and distribution system.
Company List
- GSK
- AstraZeneca
- Novartis
- Sanofi
- Roche
- Teva
- Merck
- Pfizer
- Boehringer Ingelheim
- Amgen
- Regeneron
- AbbVie
- Bayer
- Mylan
- Cipla
Recent Development News
In April 2026, AstraZeneca Warns Germany’s Drug Pricing Reforms Could Delay New Respiratory Drug Launches: AstraZeneca CEO Pascal Soriot warned that proposed German pharmaceutical pricing reforms may discourage companies from introducing innovative medicines in Europe, including respiratory and asthma therapies. The comments highlighted growing concerns about Europe’s competitiveness in advanced drug development and commercialization.
Source: https://www.reuters.com
In April 2026, AstraZeneca Beats Q1 Expectations as Company Pushes Forward with New Drug Launch Pipeline: AstraZeneca exceeded Q1 2026 profit expectations and confirmed plans for multiple new drug launches through 2030. The company also cautioned that international drug pricing pressure could impact future launches in Europe, including respiratory and chronic disease therapies tied to the asthma treatment market.
Source: https://www.reuters.com
What Strategic Insights Define the Future of the Europe Asthma Drugs Market?
The Europe Asthma Drugs Market is changing its fundamental structure to focus on precision medicine and outcome-based treatment systems which depend on therapy results and treatment length rather than patient numbers. The shift occurs because payers demand funding for treatments which decrease hospital usage thus they prefer biologic drugs and combination treatments with digital patient compliance systems. Over the next 5–7 years healthcare providers will develop new treatment pathways which unite diagnostic testing and medication administration with patient monitoring into one comprehensive system.
The less visible danger of biotech drugs being used by more patients results in increased financial pressure. Governments may tighten reimbursement criteria or introduce reference pricing which could compress margins despite rising adoption.
The opportunity arises from digital therapeutics combining with respiratory medications in Germany which has developing reimbursement systems. Companies should invest early in drug-device ecosystems and real-world data capabilities to secure long-term differentiation and payer alignment.
Europe Asthma Drugs Market Report Segmentation
By Type
- Short-acting
- Long-acting
- Combination Drugs
- Others
By Application
- Acute Treatment
- Maintenance Therapy
- Others
By End-User
- Hospitals
- Clinics
- Pharmacies
- Others
By Route
- Inhalation
- Oral
- Injectable
- Others
Frequently Asked Questions
Find quick answers to common questions.
The approximate Europe Asthma Drugs Market size for the market will be USD 7965.7 Million in 2033.
The key segments of the Europe Asthma Drugs Market are By Type (Short-acting, Long-acting, Combination Drugs, Others), By Application (Acute Treatment, Maintenance Therapy, Others), By End-User (Hospitals, Clinics, Pharmacies, Others), By Route (Inhalation, Oral, Injectable, Others).
Major players in the Europe Asthma Drugs Market are GSK, AstraZeneca, Novartis, Sanofi, Roche, Teva, Merck, Pfizer, Boehringer Ingelheim, Amgen, Regeneron, AbbVie, Bayer, Mylan, Cipla.
The current market size of the Europe Asthma Drugs Market is USD 5690.2 Million in 2025.
The Europe Asthma Drugs Market CAGR is 4.32%.
- GSK
- AstraZeneca
- Novartis
- Sanofi
- Roche
- Teva
- Merck
- Pfizer
- Boehringer Ingelheim
- Amgen
- Regeneron
- AbbVie
- Bayer
- Mylan
- Cipla
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033