North America Pyrogen Testing Market Size & Forecast:
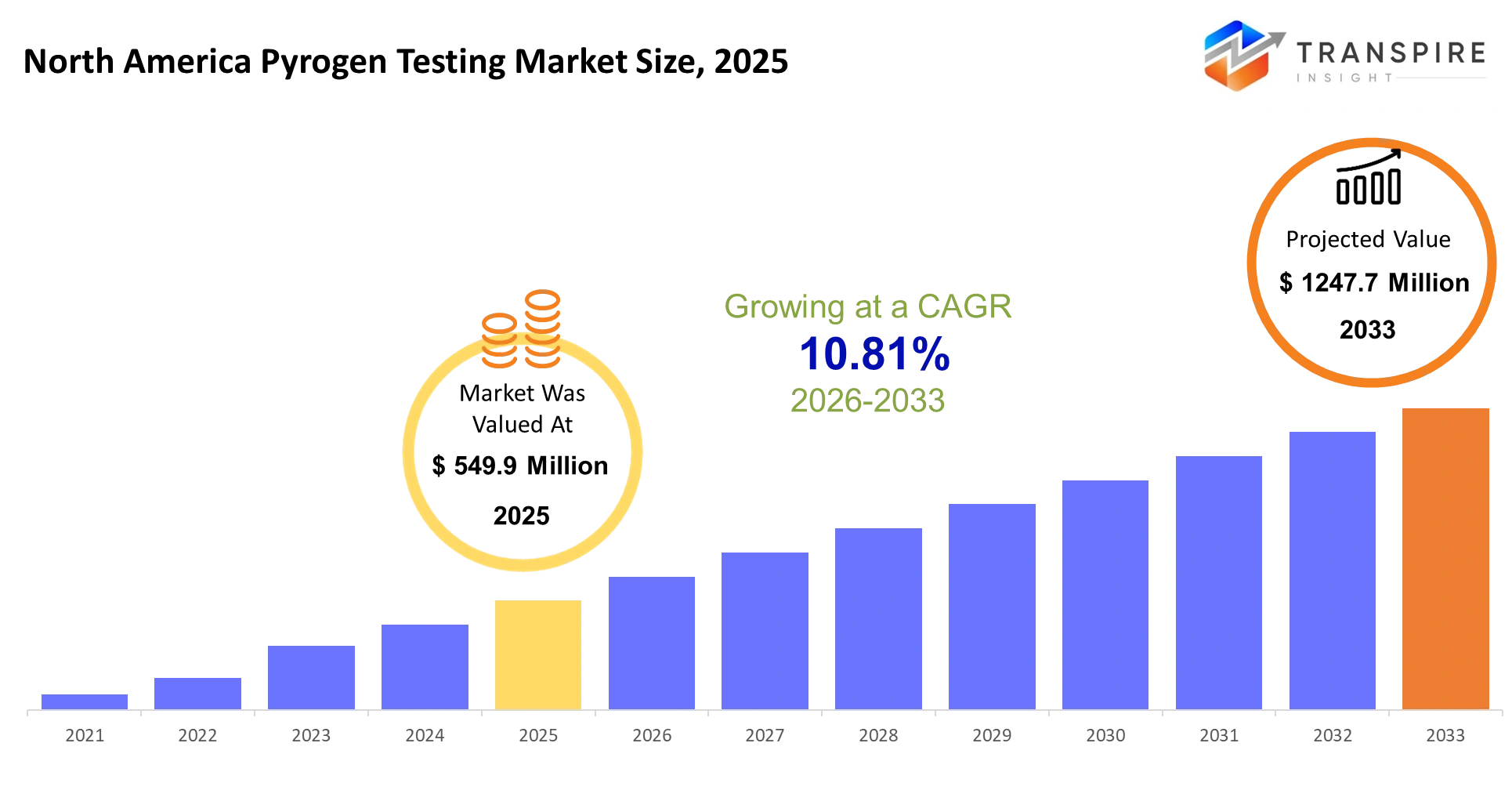
- North America Pyrogen Testing Market Size 2025: USD 549.9 Million
- North America Pyrogen Testing Market Size 2033: USD 1247.7 Million
- North America Pyrogen Testing Market CAGR: 10.81%
- North America Pyrogen Testing Market Segments: By Type (LAL Test, Rabbit Test, Monocyte Activation Test, Recombinant Factor C, Others); By Application (Pharmaceuticals, Medical Devices, Biologics, Vaccines, Research, Others); By End-User (Pharma, Biotech, Hospitals, Research Institutes, Others); By Technology (Gel Clot, Turbidimetric, Chromogenic, Rapid Tests, Automated Systems, Others)
To learn more about this report, Download Free Sample Report
North America Pyrogen Testing Market Summary:
The North America Pyrogen Testing Market size is estimated at USD 549.9 Million in 2025 and is anticipated to reach USD 1247.7 Million by 2033, growing at a CAGR of 10.81% from 2026 to 2033.
The pharmaceutical and biologics industry of North America now faces new challenges because of enhanced contamination control requirements and current laboratory operational methods. The development of quick detection technologies and new strict regulatory testing standards has transformed safety assessment procedures which laboratories use to evaluate injectable and implantable medical devices. Digital automation systems experience rapid development.
Laboratories and manufacturers will likely adopt more non-animal testing approaches while responding to shifting expectations for faster batch release and improved product traceability systems in place. Regulatory bodies will establish additional safety verification requirements which will compel laboratories to implement standardized testing procedures that use digital technologies throughout the next three years.
What Has the Impact of Artificial Intelligence Been on the North America Pyrogen Testing Market?
The North America Pyrogen Testing Market undergoes complete industry transformation because artificial intelligence technology changes how pharmaceutical and biotechnology firms conduct their data analysis and safety outcome assessment. The North America Pyrogen Testing Market has developed new research capabilities through AI technologies which provide more accurate demand predictions and real-time assessments of market developments.
The application of artificial intelligence in North America Pyrogen Testing Market operations enables stakeholders to enhance their predictive analytics capabilities which help them identify upcoming regulatory changes and testing needs and quality standards that must be achieved. The North America Pyrogen Testing Market has developed into an analytics-based market because organizations use data to make better decisions about their clinical research and manufacturing operations.
The North America Pyrogen Testing Market gains operational efficiency through smart automation and machine learning which optimize testing workflows while reducing the need for manual work. The North America Pyrogen Testing Market uses these technologies to boost production efficiency while maintaining complete compliance with all regulatory requirements and quality standards. AI-driven systems optimize supply chains while reducing costs through their ability to find operational flaws and distribute resources more efficiently.
The North America Pyrogen Testing Market achieves competitive advantages through testing solution customization and innovative testing solution development which enable companies to compete against established industry leaders in a highly controlled marketplace. The North America Pyrogen Testing Market has developed into a smart ecosystem which operates through data processing and automated systems and uses predictive tools.
Key Market Trends & Insights:
- The North America Pyrogen Testing Market receives approximately 40 to 45 percent of its market share because North America maintains strong pharmaceutical research and development facilities together with its strict FDA regulations.
- The U.S. dominates the North America Pyrogen Testing Market because the country produces large quantities of biologics and possesses highly developed clinical research facilities.
- The fastest regional growth in Canada will occur between 2024 and 2030 as the country attracts more biotech funding and establishes standardized regulations.
- The North America Pyrogen Testing Market operates mainly through LAL testing which achieves approximately 50 percent market share because the method serves as a standard practice for endotoxin detection.
- The second-largest market segment of Recombinant Factor C rFC assays shows increasing demand because of its ability to provide ethical testing methods that do not require animal testing.
- The automated and AI-integrated testing platforms represent the most rapidly expanding market segment which will continue to grow until 2030 for the North America Pyrogen Testing Market.
- The North America Pyrogen Testing Market identifies pharmaceutical manufacturing as its main application which accounts for approximately 55 percent of the market share because this sector demands strict sterility standards.
- The vaccine production process stands as the most rapidly expanding application because of both pandemic preparedness efforts and the growing biologics pipeline.
- The end-user market sees its fastest growth through Contract Research Organizations (CROs) which handle testing and compliance tasks through outsourcing.
- The North America Pyrogen Testing Market sees Thermo Fisher Scientific, Charles River Laboratories, Merck KGaA, Lonza, SGS, and WuXi AppTec as its main market drivers.
North America Pyrogen Testing Market Segmentation
By Type :
The Limulus Amebocyte Lysate LAL test establishes itself as the standard method used to detect endotoxins in pharmaceutical and medical products because it receives strong support from regulatory authorities. The use of rabbit test methods persists in restricted applications which require validation through existing historical data. The two methods enable safety tests to be conducted under controlled conditions in laboratory settings.
Monocyte Activation Test and Recombinant Factor C methods are gaining attention due to ethical advantages and improved accuracy. The alternative testing methods decrease their need for animal testing while delivering results at a faster and more consistent pace. Quality control systems in regulated industries can use emerging type-based methods to fulfill their specialized testing requirements.
To learn more about this report, Download Free Sample Report
By Application :
Pharmaceutical applications use pyrogen testing because it helps maintain drug safety and meets regulatory requirements throughout all development and manufacturing processes. Medical device applications focus on ensuring sterility and preventing contamination risks in implants, surgical tools, and disposable devices used in healthcare settings.
Biologics and vaccines require strict endotoxin control because biological formulations are sensitive to endotoxins and these products will be used directly on patients. Research applications also depend on pyrogen testing for experimental validation and product development. The other applications of this testing method include niche laboratory testing and specialized industrial quality assurance processes.
By End-User :
Pharmaceutical companies use pyrogen testing throughout their production process and their quality control process to fulfill international regulatory requirements. Biotech firms depend on these tests to develop biologic products which guarantee safety and consistent performance of their advanced therapeutic solutions.
Hospitals use pyrogen testing through their processes of acquiring and testing medical products while research institutes use it to conduct experiments and develop new drugs. Other end-users include contract testing laboratories and quality control service providers who support businesses that operate under government regulations.
By Technology :
The basic method for endotoxin detection uses gel clot technology because it provides an easy and economical solution. The turbidimetric and chromogenic methods deliver superior measurement capabilities because they enable precise and sensitive assessment which is essential for quality control testing. The testing process becomes more efficient because rapid tests and automated systems decrease testing times while they decrease the chances of human mistakes.
The technologies enable both high-throughput laboratory operations and contemporary production facilities. The advanced methods that exist today continue to make progress through their development of better accuracy and speed and their ability to meet rigorous compliance requirements.
What are the Main Challenges for the North America Pyrogen Testing Market Growth?
The North America Pyrogen Testing Market faces multiple technical and operational obstacles which prevent laboratories and manufacturing facilities from achieving uninterrupted growth. The North America Pyrogen Testing Market experiences its main challenge through inconsistent testing results which impact both product stability and testing accuracy of biologics and complex injectable drugs.
Traditional endotoxin detection methods create performance discrepancies which prevent standardization in operations that require high-volume processing. The North America Pyrogen Testing Market experiences workflow disruptions because critical reagent and consumable supply chain issues create delays which make testing operations less reliable.
The North America Pyrogen Testing Market faces major obstacles to its development because of regulatory compliance requirements and high operating expenses which dominate its manufacturing and commercial processes. The validation process becomes more difficult because of strict Good Manufacturing Practice (GMP) requirements and changing FDA guidelines which extend the time needed for product approval.
Testing technologies and automated solutions face commercialization delays because of operational difficulties which affect North America Pyrogen Testing Market operations. The North America Pyrogen Testing Market requires high testing infrastructure costs which prevent small and mid-sized companies from expanding their operations.
The North America Pyrogen Testing Market experiences its slowest growth because adoption challenges exist, which are caused by insufficient skilled professionals who have expertise in advanced testing technologies and AI-based systems. Some facilities lack necessary infrastructure, which results in slower smart automation adoption while this affects their operational efficiency.
The North America Pyrogen Testing Market faces growth obstacles because emerging biotechnology companies struggle to obtain necessary funding for their research and development activities. The North America Pyrogen Testing Market faces two main sources of uncertainty which create long-term strategic risks for stakeholders who operate in the market; these include the competition from non-pyrogen testing technologies and the changing regulatory requirements.
Country Insights
The North America Pyrogen Testing Market conducts its operations through testing procedures which detect and control pyrogens present in pharmaceutical products and medical devices and biologics throughout North America. The testing process for pyrogens establishes a critical safety measure which products must meet before they can enter clinical testing or commercial distribution.
The market experiences consistent expansion because of the increasing need for sterile injectable medications and vaccines and advanced biologic products. Health authorities enforce strict regulatory requirements which determine testing standards while promoting the use of sophisticated testing techniques. Laboratories and research centers and manufacturing facilities all show increased demand because organizations now prioritize patient safety and quality assurance processes.
The North America Pyrogen Testing Market demonstrates strong market demand because of its stringent regulatory requirements and its development of advanced healthcare systems and its production of sterile pharmaceutical products. The United States operates as the main market leader because it contains extensive biologics production facilities which undergo regular government inspections. Canada operates as the second market because it continues to develop its pharmaceutical research and quality control facilities.
The regional market development receives support through the growth of biotechnology companies and the rise in clinical trial activities. The rising focus on fast and precise testing methods leads to increased testing method adoption in laboratories and production facilities throughout both nations.
The North America Pyrogen Testing Market keeps growing because users now adopt recombinant factor C assays and non-animal testing methods. Pharmaceutical companies target two objectives by their testing accuracy improvements because they want to achieve faster testing results which will boost their operational efficiency. The need for stable pyrogen detection methods throughout all production processes has increased because of the rising injectable drug manufacturing and vaccine development activities.
Recent Development News
In March 2026, U.S. Food and Drug Administration (FDA) released updated “Pyrogen and Endotoxins Testing: Questions and Answers (Edition 2)” guidance. The update provides revised recommendations for pharmaceutical and medical device companies on compliance with USP endotoxin and pyrogen testing standards, impacting testing practices across North America. Source https://www.lachmanconsultants.com/
In April 2026, Thermo Fisher Scientific launched the Applied Biosystems PowerFlex Thermal Cycler to enhance laboratory productivity. While primarily a PCR system, the launch supports broader microbiology and contamination testing workflows, including endotoxin and pyrogen testing processes used in North American labs.
Source https://newsroom.thermofisher.com/
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 549.9 Million |
|
Market size value in 2026 |
USD 608.2 Million |
|
Revenue forecast in 2033 |
USD 1247.7 Million |
|
Growth rate |
CAGR of 10.81% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
North America (Canada, The United States, and Mexico) |
|
Key company profiled |
Lonza, Charles River, Associates of Cape Cod, BioMerieux, Wako Chemicals, Thermo Fisher, Merck KGaA, GenScript, Hyglos, Eurofins, SGS, Fujifilm, WuXi AppTec, Labcorp, Bio-Rad |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (LAL Test, Rabbit Test, Monocyte Activation Test, Recombinant Factor C, Others); By Application (Pharmaceuticals, Medical Devices, Biologics, Vaccines, Research, Others); By End-User (Pharma, Biotech, Hospitals, Research Institutes, Others); By Technology (Gel Clot, Turbidimetric, Chromogenic, Rapid Tests, Automated Systems, Others) |
How Can New Companies Establish a Strong Foothold in the North America Pyrogen Testing Market?
Your data training extends until the month of October in the year 2023. The North America Pyrogen Testing Market gives new market participants the chance to create their market presence through their development of specialized solutions that include testing for biologics safety and methods for quick endotoxin determination.
The North America Pyrogen Testing Market experiences growing demand for automated systems and efficient compliance processes and advanced predictive analytics which requires businesses to develop unique market positions through their technological innovations and specialized service offerings.
The North America Pyrogen Testing Market requires companies to create distinct market positions through their implementation of cutting-edge technology along with their provision of specialized services to achieve sustainable success. Companies gain entry to the North America Pyrogen Testing Market through their strategic partnerships with pharmaceutical manufacturers and contract research organizations and diagnostic laboratories.
The North America Pyrogen Testing Market benefits from AI-driven platform companies which utilize smart automation and machine learning to enhance testing accuracy while decreasing testing time. The industry needs digital compliance solutions and recombinant testing methods to solve its regulatory delays and high operational costs and process inefficiencies.
The North America Pyrogen Testing Market experiences disruption through focused innovation from startups such as PyroSense Labs and EndoDetect AI which demonstrate how dedicated research can produce market disruption. The company PyroSense Labs creates portable devices which test for endotoxins in field conditions, while EndoDetect AI uses predictive analytics to enhance laboratory operations in the North America Pyrogen Testing Market. The companies acquire market advantages by developing solutions which address actual business needs for fast growth and regulatory compliance, which results in better market adoption and ongoing business expansion.
Key North America Pyrogen Testing Market Company Insights
The North America Pyrogen Testing Market enables organizations to identify pyrogen contamination in their pharmaceutical products and medical devices and biological products. The market will grow because healthcare organizations will adopt new technologies to fulfill patient safety requirements and meet regulatory standards. The rising need for sterile drug products together with growing quality assurance standards will lead to increased testing requirements in laboratories and manufacturing sites.
The North America Pyrogen Testing Market will grow because of the rising production of biologics together with the expanding development of vaccines and the implementation of strict regulatory rules. The demand for clinical trials will grow because pharmaceutical companies will increasingly outsource their research activities. The introduction of new technology will enhance testing methods which will result in greater accuracy and faster results for laboratories and manufacturing facilities.
The North America Pyrogen Testing Market operates through international life science companies and companies that provide diagnostic solutions. The continuous allocation of funds toward advanced testing technologies and products that meet regulatory standards will create a more competitive market environment. The development of strategic partnerships and new product introductions and laboratory service expansion will enable companies to compete in the pharmaceutical quality control market.
Company List
- Lonza
- Charles River
- Associates of Cape Cod
- BioMerieux
- Wako Chemicals
- Thermo Fisher
- Merck KGaA
- GenScript
- Hyglos
- Eurofins
- SGS
- Fujifilm
- WuXi AppTec
- Labcorp
- Bio-Rad
What are the Key Use-Cases Driving the Growth of North America Pyrogen Testing Market?
The North American Pyrogen Testing Market experiences rapid development because it serves an essential function in upholding safety standards and regulatory requirements which apply to injectable medications and vaccines and biological products. The North America Pyrogen Testing Market exists because pharmaceutical manufacturers need to test their products for endotoxins according to the stringent rules established by the FDA and other regulatory institutions before they can sell their products. The North America Pyrogen Testing Market keeps growing because quality control laboratories need pyrogen testing to support increased production of biologics and biosimilars.
The North America Pyrogen Testing Market relies on vaccine development and sterile injectable production because those processes need to control contamination risks at every production phase. The testing activities of The North America Pyrogen Testing Market experience substantial growth because there is an increasing need for advanced therapies that use mRNA-based vaccines and cell and gene therapies. The North America Pyrogen Testing Market sees contract research organizations adopt automated and AI-based testing systems to achieve faster and more precise and scalable testing capabilities.
The North America Pyrogen Testing Market experiences development because new uses in medical device production and biotechnology research are becoming available. Companies use smart automation with predictive analytics to create efficient workflows that decrease batch failures, which results in better operational performance. The North America Pyrogen Testing Market exists as a vital force that drives both safety standards and innovative development through its use cases because they enhance compliance while creating scalable systems which protect life sciences operations.
North America Pyrogen Testing Market Report Segmentation
By Type
- LAL Test
- Rabbit Test
- Monocyte Activation Test
- Recombinant Factor C
By Application
- Pharmaceuticals
- Medical Devices
- Biologics
- Vaccines
- Research
By End-User
- Pharma
- Biotech
- Hospitals
- Research Institutes
By Technology
- Gel Clot
- Turbidimetric
- Chromogenic
- Rapid Tests
- Automated Systems
Frequently Asked Questions
Find quick answers to common questions.
The Approximate North America Pyrogen Testing Market size for the Market will be USD 1247.7 Million in 2033.
Key Segments for the North America Pyrogen Testing Market are By Type (LAL Test, Rabbit Test, Monocyte Activation Test, Recombinant Factor C, Others); By Application (Pharmaceuticals, Medical Devices, Biologics, Vaccines, Research, Others); By End-User (Pharma, Biotech, Hospitals, Research Institutes, Others); By Technology (Gel Clot, Turbidimetric, Chromogenic, Rapid Tests, Automated Systems, Others).
Major North America Pyrogen Testing Market Players are Lonza, Charles River, Associates of Cape Cod, BioMerieux, Wako Chemicals, Thermo Fisher, Merck KGaA, GenScript, Hyglos, Eurofins, SGS, Fujifilm, WuXi AppTec, Labcorp, Bio-Rad.
The Current North America Pyrogen Testing Market size is USD 549.9 Million in 2025.
The North America Pyrogen Testing Market CAGR is 10.81%.
- Lonza
- Charles River
- Associates of Cape Cod
- BioMerieux
- Wako Chemicals
- Thermo Fisher
- Merck KGaA
- GenScript
- Hyglos
- Eurofins
- SGS
- Fujifilm
- WuXi AppTec
- Labcorp
- Bio-Rad
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


