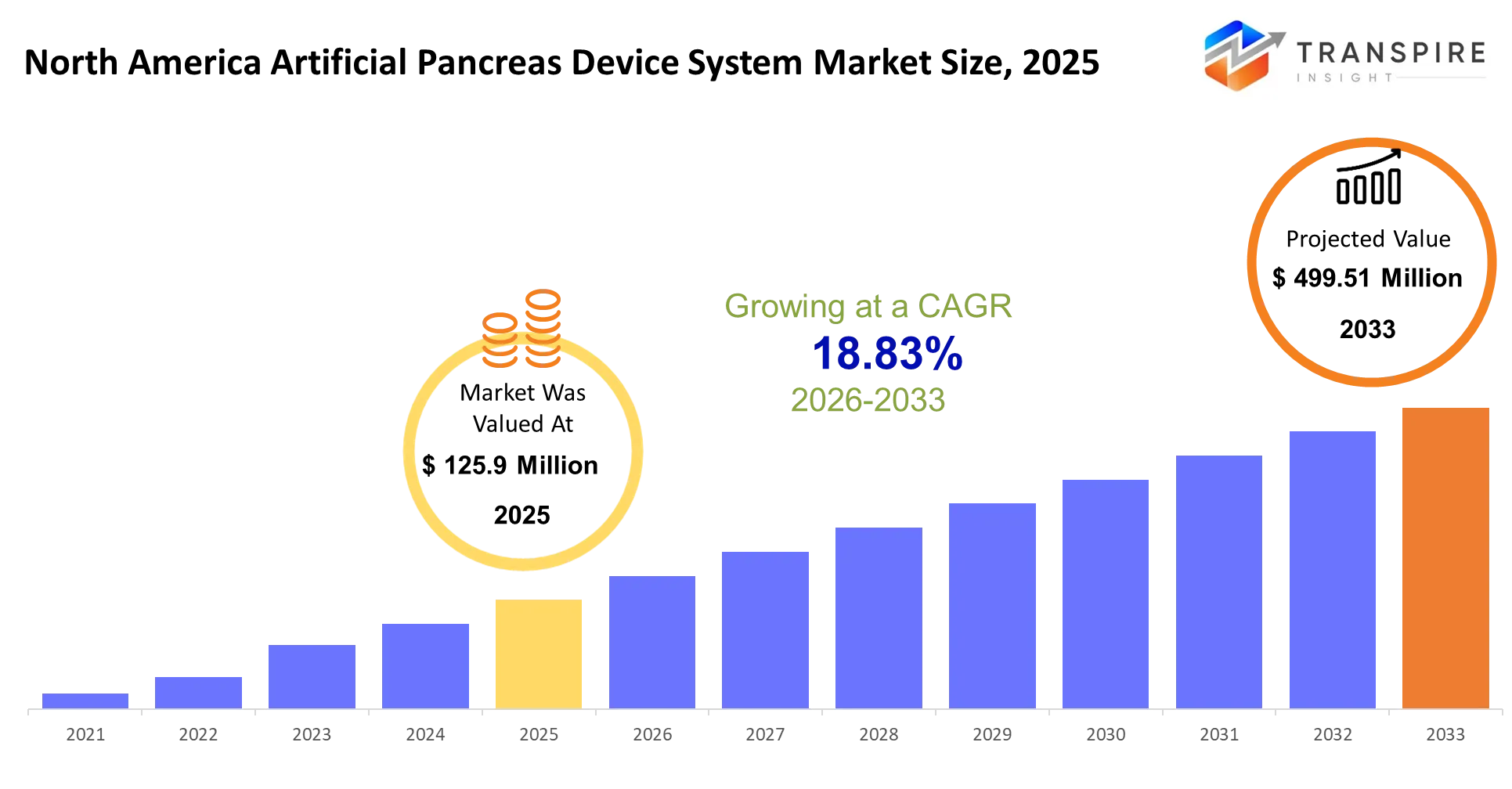
North America Artificial Pancreas Device System Market Size & Forecast:
- North America Artificial Pancreas Device System Market Size 2025: USD 125.9 Million
- North America Artificial Pancreas Device System Market Size 2033: USD 499.51 Million
- North America Artificial Pancreas Device System Market CAGR: 18.83%
- North America Artificial Pancreas Device System Market Segments: By Type (Closed-loop Systems, Hybrid Systems, Insulin Pumps, CGM Systems, Others), By Application (Type 1 Diabetes, Type 2 Diabetes, Research, Others), By End-User (Hospitals, Homecare, Clinics, Others), By Component (Sensors, Pumps, Controllers, Software, Others).
To learn more about this report, Download Free Sample Report
North America Artificial Pancreas Device System Market Summary:
The North America Artificial Pancreas Device System Market size is estimated at USD 125.9 Million in 2025 and is anticipated to reach USD 499.51 Million by 2033, growing at a CAGR of 18.83% from 2026 to 2033. The North America Artificial Pancreas Device System Market is experiencing strong growth because more people develop diabetes and more people want automated insulin delivery systems.
The advanced systems provide continuous glucose monitoring which works together with insulin pumps to enable patients to control their blood sugar levels without needing to do anything. People in the region increase their understanding of diabetes management while technology develops closed-loop systems which drive market growth throughout the area.
The combination of regulatory approvals and medical device companies' innovative development work is driving hospitals and homecare facilities to start using new products. This artificial pancreas device system is now considered the first choice of treatment for these patients due to its improved precision and reduced occurrence of hypoglycemia episodes along with enhanced daily activity levels. There will be strong demand for the North America Artificial Pancreas Device System Market due to increasing health care expenditure and customized treatments for diabetics.
What Has the Impact of Artificial Intelligence Been on the North America Artificial Pancreas Device System Market?
There are a number of major changes that occur due to the use of AI systems in the North America Artificial Pancreas Device System Market. AI systems help improve the accuracy of diabetic care while also helping to create insulin dispensing devices. North America Artificial Pancreas Device System Market makes use of AI to examine data from continuous glucose monitoring.
The North America Artificial Pancreas Device System Market uses artificial intelligence to track glucose patterns through machine learning and predictive analytics which helps reduce hypoglycemia risks while improving patient health results. The North America Artificial Pancreas Device System Market shows rising market trends because these technology improvements create more flexible solutions which meet specific patient needs.
AI-powered smart automation solutions enhance device efficiency while increasing system dependability throughout the North America Artificial Pancreas Device System Market. North America Artificial Pancreas Device System Market manufacturers use predictive analytics to anticipate demand shifts while they optimize production schedules and decrease manufacturing expenses.
North America Artificial Pancreas Device System Market organizations use artificial intelligence to boost supply chain performance through real-time market surveillance and quick inventory management. The North America Artificial Pancreas Device System Market achieves special market advantages through tailored solutions which provide better precision and ongoing product improvements that result from artificial intelligence integration.
Key Market Trends & Insights:
- The United States controls between 70-75 percent of North America Artificial Pancreas Device System Market which will reach that share in 2025 because of its superior healthcare systems and its capacity to use new technologies.
- The North America Artificial Pancreas Device System Market is experiencing its fastest growth in Canada which will develop quickly from 2026 to 2030 because of increasing diabetes rates.
- The North America Artificial Pancreas Device System Market shows product segmentation results that demonstrate insulin pump-integrated systems control the main market share at 45 percent because of their strong clinical performance.
- Systems based on continuous glucose monitoring (CGM) rank as the second biggest market segment while fully closed-loop systems show the fastest market growth because they use artificial intelligence.
- The North America Artificial Pancreas Device System Market shows Type 1 diabetes management as the main application which controls 80 of market share.
- The field of Type 2 diabetes applications is experiencing rapid growth because more patients become eligible for treatment and devices become easier to access.
- The North America Artificial Pancreas Device System Market shows hospitals as the primary end-user segment which controls 60 of the market because of their strong clinical adoption.
- The homecare industry is experiencing its strongest growth because of patients' ability to monitor their health remotely.
- The companies are making big investments in artificial intelligence predictive analytics systems and smart automation together with machine learning algorithms to enhance their ability to control glucose levels.
- The North America Artificial Pancreas Device System Market is experiencing stronger competitive positioning through three main factors which include strategic partnerships and FDA product approvals combined with new product developments.
North America Artificial Pancreas Device System Market Segmentation
By Type
The research shows that closed-loop systems operate insulin delivery through automatic glucose measurement-based adjustments which respond to actual glucose measurements. The system decreases the need for human involvement while it increases the precision of glucose monitoring, which results in improved health results for patients.
The market demand for hybrid systems remains strong because these systems provide automated functions while requiring users to make only minimal inputs, which creates an optimal solution for patients who require partial autonomy. Insulin pumps maintain their popularity among users because medical professionals trust their performance and they can easily connect to monitoring equipment.
CGM systems operate as essential tools which provide continuous glucose level monitoring to empower healthcare providers with advanced treatment decision-making capabilities. The North American artificial pancreas device system market experiences ongoing treatment efficiency improvements because various new technologies emerge in the market.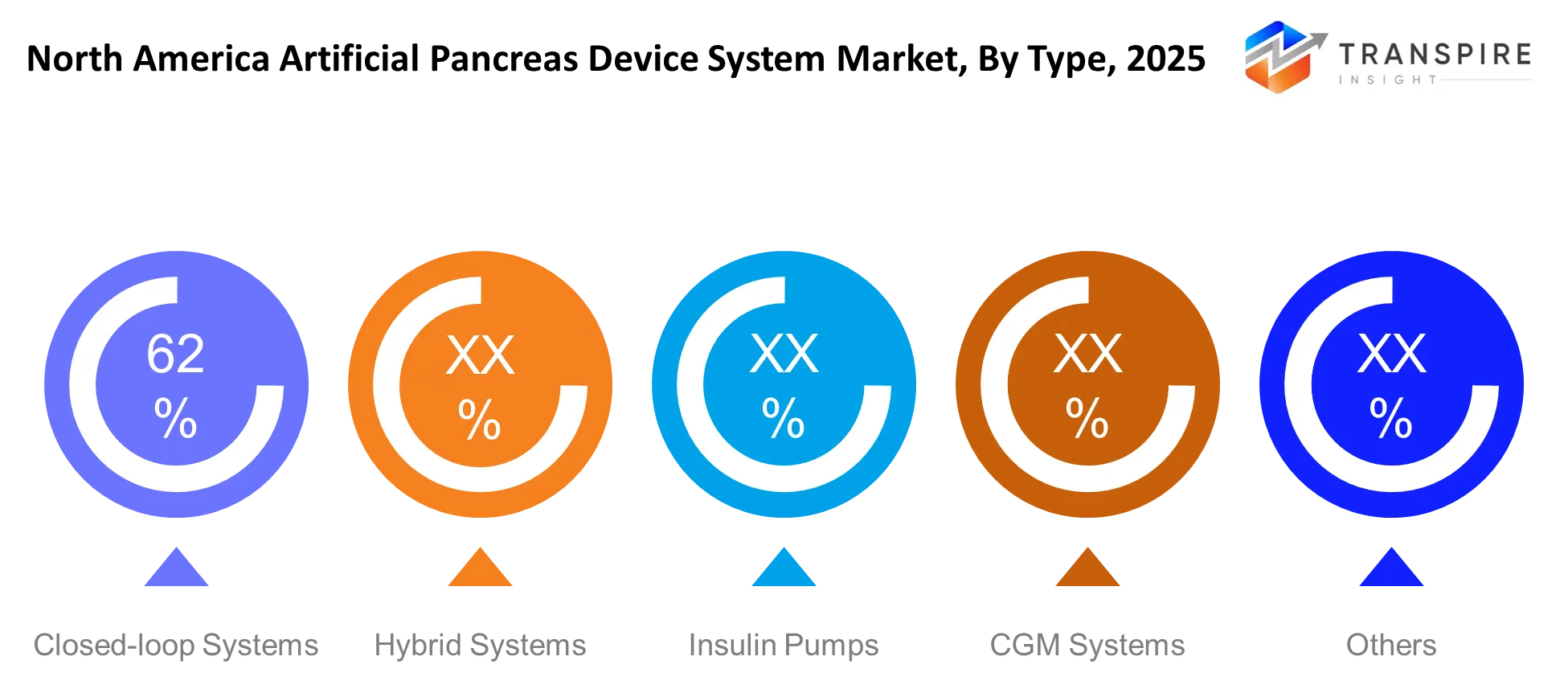
To learn more about this report, Download Free Sample Report
By Application
Patients with Type 1 diabetes need permanent insulin treatment together with exact blood sugar measurement, which makes this condition their primary medical requirement. The segment achieves better disease management results because automated systems have been widely adopted through this segment.
Patient awareness about diabetes and device accessibility have led to increased adoption of Type 2 diabetes treatment methods. Healthcare institutions conduct research studies to evaluate new algorithms together with smart automation systems which enhance their operational accuracy.
The field of research is expanding because organizations are developing new projects which include both experimental and preventive health care solutions. The North America Artificial Pancreas Device System Market experiences demand growth because clinical acceptance and technological advancements are expanding in these two fields.
By End-User
The primary end-user group for advanced monitoring systems at hospitals uses these systems to manage critical diabetes needs and oversee patient care. The presence of strong infrastructure together with skilled medical personnel results in high clinical adoption rates. Homecare usage is growing rapidly because patients prefer receiving treatment in their homes while doctors can monitor them from a distance.
Patients experience better quality of life through this shift because it allows them to track their glucose levels continuously without needing to visit hospitals. Outpatient diabetes care with follow-up services from clinics creates ongoing demand for their services. Research centers and other end-user organizations drive innovation through testing of advanced systems. The North America Artificial Pancreas Device System Market experiences growth through various factors that work together to create market expansion.
By Component
Sensors function as essential elements because they maintain constant glucose level tracking while delivering immediate data which enables precise insulin administration. Pumps serve as vital tools because they enable secure insulin delivery which healthcare facilities use to execute their contemporary automated medication distribution systems. Controllers function as essential components because they handle sensor data processing which enables them to deliver prompt diabetes treatment modifications.
The increasing use of AI algorithms and predictive analytics technologies has made software solutions vital for modern operations. The systems enhance decision-making processes while delivering customized diabetes treatment solutions for patients. The system performance depends on other elements which assist in maintaining operational reliability. The North America Artificial Pancreas Device System Market uses these elements to drive technological progress and market growth.
What are the Main Challenges for the North America Artificial Pancreas Device System Market Growth?
The North American Artificial Pancreas Device System Market encounters multiple technical and operational difficulties which hinder its complete implementation in various healthcare facilities. The system struggles to maintain consistent glucose measurements because it cannot achieve precise results during fast-changing patient conditions.
North America Artificial Pancreas Device System Market needs to achieve exact sensor and pump and control algorithm integration because this requirement adds more complexity to the system operation. The combination of limited device scalability and occasional performance inconsistencies creates obstacles that prevent real-world applications from achieving their full potential.
The North America Artificial Pancreas Device System Market faces major restrictions for its manufacturing and commercialization processes because of the enforced stringent regulatory standards and demanding production requirements. Medical-grade safety protocols require compliance which results in extended development periods and higher production expenses.
The North America Artificial Pancreas Device System Market companies experience product approval delays which result in extended time periods before their products enter the market. Manufacturers face challenges in their ability to conduct large-scale commercialization because of their high R&D expenses and the need to manage complex supply chain operations.
The North America Artificial Pancreas Device System Market faces adoption challenges because certain patient groups need to be educated about the system while there is a shortage of healthcare professionals who have received proper training. High device costs restrict accessibility, especially in non-urban healthcare facilities, reducing overall penetration. Advanced automated solutions face limited implementation because of existing infrastructure deficiencies in diabetes care management systems. The North America Artificial Pancreas Device System Market growth faces major obstacles from these factors.
Country Insights
The North America Artificial Pancreas Device System Market depends on United States territory because of its developed healthcare infrastructure and its quick implementation of diabetes treatment solutions. The automated insulin delivery system has better acceptance because patients know about it and their insurance plans cover it. The region will experience steady development because of ongoing medical device innovations and active clinical research projects.
The North America Artificial Pancreas Device System Market shows continuous development in Canada because of increasing diabetes prevalence and improvements in digital healthcare system capabilities. The government supports chronic disease management through its initiatives which drive people to use smart glucose monitoring systems. The current remote area access restrictions create service gaps but upcoming healthcare investments will improve access to services throughout the next few years.
The North America Artificial Pancreas Device System Market sees higher adoption rates in urban areas because these regions possess better hospital systems and offer modern medical treatments. People living in rural areas experience difficulties with system adoption because they need to spend money on special medical services which are not easily found. The market will expand through the growth of telehealth services and increased public knowledge about the industry.
Recent Development News
In February 2026, Medtronic Diabetes expanded its U.S. insulin delivery ecosystem: Medtronic announced the expansion of its insulin delivery ecosystem in the U.S., combining Medicare access improvements with new FDA clearances for its automated insulin delivery (AID) platforms, strengthening its artificial pancreas portfolio.
Source: https://news.medtronic.com
In February 2026, Portal Diabetes received FDA Breakthrough Device Designation: Portal Diabetes’ implantable insulin pump system—widely described as a next-generation “artificial pancreas”—received FDA Breakthrough Device Designation. This marks a major regulatory milestone, accelerating its clinical pathway toward closed-loop insulin automation.
Source: https://www.hcplive.com
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 125.9 Million |
|
Market size value in 2026 |
USD 149.32 Million |
|
Revenue forecast in 2033 |
USD 499.51 Million |
|
Growth rate |
CAGR of 18.83% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
North America (Canada, The United States, and Mexico) |
|
Key company profiled |
Medtronic, Tandem Diabetes Care, Insulet, Dexcom, Abbott, Roche, Johnson & Johnson, Eli Lilly, Novo Nordisk, Ascensia Diabetes Care, Senseonics, Beta Bionics, Bigfoot Biomedical, Ypsomed, SOOIL Development |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Closed-loop Systems, Hybrid Systems, Insulin Pumps, CGM Systems, Others), By Application (Type 1 Diabetes, Type 2 Diabetes, Research, Others), By End-User (Hospitals, Homecare, Clinics, Others), By Component (Sensors, Pumps, Controllers, Software, Others). |
How Can New Companies Establish a Strong Foothold in the North America Artificial Pancreas Device System Market?
New companies entering the North America Artificial Pancreas Device System Market can build a strong position by focusing on innovation-led differentiation and solving unmet clinical needs in diabetes care. The North America Artificial Pancreas Device System Market rewards technologies that improve real-time glucose control and reduce manual intervention and enhance patient comfort.
Startups that invest in AI-driven insulin automation and wearable integration and predictive analytics will gain faster acceptance in a competitive healthcare environment. The company will achieve better results through patient-centric design since this method will increase product usage and build lasting trust with users. The hospitals and diabetes clinics and established med-tech companies which new partnerships will create in the North America Artificial Pancreas Device System Market will help establish stronger market entry pathways.
Research institutions provide new companies with resources to test product performance while they expedite their clinical trial operations. The North America Artificial Pancreas Device System Market benefits from Beta Bionics and Bigfoot Biomedical because these companies use intelligent automation systems to improve their clinical research partnerships. The research partnerships help both groups achieve quicker regulatory approvals which allow them to access more markets.
The North America Artificial Pancreas Device System Market requires technology differentiation as the demand for closed-loop systems and AI-enabled insulin delivery systems increases. New entrants must focus on reducing device complexity while they improve sensor accuracy and enhance system reliability. Sequel Med Tech has developed automated insulin delivery systems which provide simplified operation to eliminate barriers for multiple patients.
Key North America Artificial Pancreas Device System Market Company Insights
The North America Artificial Pancreas Device System Market experiences strong market competition which results from ongoing technological advancements in both automated insulin delivery systems and AI-powered glucose monitoring technologies. The top companies in the industry work to achieve three main objectives which include enhancing device precision and streamlining operational processes and providing better treatment experiences for patients. The North America Artificial Pancreas Device System Market will continue to see intense rivalry as demand for smarter diabetes management solutions increases across hospitals and homecare settings.
Three companies which include Medtronic and Tandem Diabetes Care and Insulet Corporation dominate the North America Artificial Pancreas Device System Market because they offer advanced product lines which medical facilities across the country use. Dexcom and Abbott support market expansion through high-performance continuous glucose monitoring systems. The companies dedicate significant resources to research activities and partnership development and regulatory approval processes which will enable them to enhance their market position in the North America Artificial Pancreas Device System Market.
The North America Artificial Pancreas Device System Market faces increased competition from new companies which develop user-friendly products that incorporate AI-based automated systems. Beta Bionics and new innovators are working to create complete insulin delivery systems which will minimize the need for patients to interact with their treatment. The North America Artificial Pancreas Device System Market will experience future changes to its competitive landscape because companies will pursue product development and expense management and digital health system integration.
Company List
- Medtronic
- Tandem Diabetes Care
- Insulet
- Dexcom
- Abbott
- Roche
- Johnson & Johnson
- Eli Lilly
- Novo Nordisk
- Ascensia Diabetes Care
- Senseonics
- Beta Bionics
- Bigfoot Biomedical
- Ypsomed
- SOOIL Development
What are the Key Use-Cases Driving the Growth of the North America Artificial Pancreas Device System Market?
The North America Artificial Pancreas Device System Market shows growth because clinical and homecare diabetes management systems use this technology for real-time patient monitoring. The North America Artificial Pancreas Device System Market operates automatic insulin delivery systems because they use continuous glucose monitoring to decrease manual work while enhancing patient protection.
The systems allow Type 1 diabetes patients to obtain exact glucose measurement through continuous monitoring. The region experiences increased adoption of personalized healthcare solutions which results in higher demand for these healthcare products. The North America Artificial Pancreas Device System Market develops through hospital systems which need precise and rapid insulin dose modifications for their critical care operations.
Emergency diabetes management and post-surgery glucose control are key use-cases which enhance treatment results. The North America Artificial Pancreas Device System Market experiences growth from outpatient medical facilities which provide patients with extended medical observation while decreasing their need to visit hospitals. The practical applications of these systems enhance the operational performance of healthcare delivery systems.
The North America Artificial Pancreas Device System Market uses remote patient monitoring systems which depend on digital health platforms and their associated devices. AI predictive systems identify glucose level changes at an early stage which helps to minimize potential health issues. The North America Artificial Pancreas Device System Market develops clinical research activities through its implementation of advanced algorithms which test better insulin delivery systems. The available use-cases create new opportunities for innovation while they help to establish market growth for the long term.
North America Artificial Pancreas Device System Market Report Segmentation
By Type
- Closed-loop Systems
- Hybrid Systems
- Insulin Pumps
- CGM Systems
- Others
By Application
- Type 1 Diabetes
- Type 2 Diabetes
- Research
- Others
By End-User
- Hospitals
- Homecare
- Clinics
- Others
By Component
- Sensors
- Pumps
- Controllers
- Software
- Others
Frequently Asked Questions
Find quick answers to common questions.
The Approximate North America Artificial Pancreas Device System Market size for the Market will be USD 499.51 Million in 2033.
The key segments of the North America Artificial Pancreas Device System Market are By Type (Closed-loop Systems, Hybrid Systems, Insulin Pumps, CGM Systems, Others), By Application (Type 1 Diabetes, Type 2 Diabetes, Research, Others), By End-User (Hospitals, Homecare, Clinics, Others), By Component (Sensors, Pumps, Controllers, Software, Others).
Major Players in the North America Artificial Pancreas Device System Market are Medtronic, Tandem Diabetes Care, Insulet, Dexcom, Abbott, Roche, Johnson & Johnson, Eli Lilly, Novo Nordisk, Ascensia Diabetes Care, Senseonics, Beta Bionics, Bigfoot Biomedical, Ypsomed, SOOIL Development.
The Current Market size of the North America Artificial Pancreas Device System Market is USD 125.9 Million in 2025.
The North America Artificial Pancreas Device System Market CAGR is 18.83%.
- Medtronic
- Tandem Diabetes Care
- Insulet
- Dexcom
- Abbott
- Roche
- Johnson & Johnson
- Eli Lilly
- Novo Nordisk
- Ascensia Diabetes Care
- Senseonics
- Beta Bionics
- Bigfoot Biomedical
- Ypsomed
- SOOIL Development
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


