Japan Intracranial Pressure Monitoring Devices Market Size & Forecast:
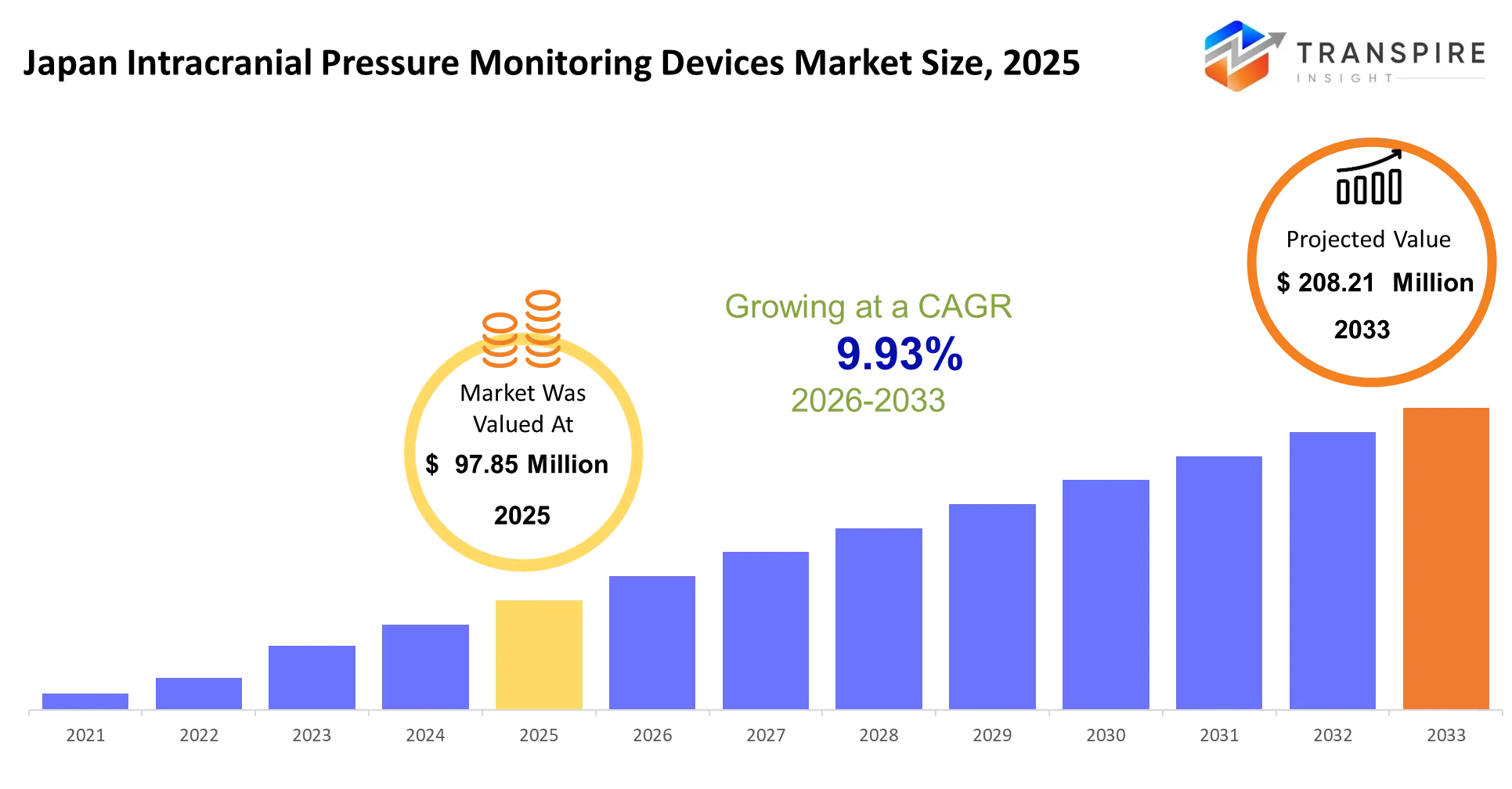
- Japan Intracranial Pressure Monitoring Devices Market Size 2025: USD 97.85 Million
- Japan Intracranial Pressure Monitoring Devices Market Size 2033: USD 208.21 Million
- Japan Intracranial Pressure Monitoring Devices Market CAGR: 9.93%
- Japan Intracranial Pressure Monitoring Devices Market Segments: By Type (Invasive Monitoring Devices, Non-invasive Monitoring Devices, Accessories, Others); By Application (Traumatic Brain Injury, Hydrocephalus, Stroke, Neurosurgery, Others); By End-User (Hospitals, Clinics, Neurosurgical Centers, Research Institutes, Others); By Technology (Fiber Optic Sensors, Microtransducers, Wireless Monitoring, Others)
To learn more about this report, Download Free Sample Report
Japan Intracranial Pressure Monitoring Devices Market Summary
The Japan Intracranial Pressure Monitoring Devices Market was valued at USD 97.85 Million in 2025. It is forecast to reach USD 208.21 Million by 2033. That is a CAGR of 9.93% over the period.
The Japan intracranial pressure monitoring devices market supports one of the most time-sensitive functions in acute neurological care: helping clinicians detect dangerous pressure changes inside the skull before they lead to irreversible brain damage. The systems operate in trauma centers and neurosurgical units and critical care settings to assist medical personnel in making treatment choices for patients who suffer from traumatic brain injuries and strokes and hydrocephalus and post-surgical complications.
The market has progressed during the past five years from traditional invasive monitoring methods to modern digital systems which use sensor technology and minimally invasive techniques for precise continuous monitoring at the patient bedside. The shift gained traction after Japan implemented hospital digitalization initiatives which included development of state-of-the-art critical care systems for hospitals that emerged from the challenges faced during the COVID-19 health crisis.
The global semiconductor shortage created supply chain disruptions which limited device distribution but hospitals responded by selecting dependable domestic and regional vendors. The rising incidence of cerebrovascular diseases in Japan because of its aging population has led hospitals to develop better diagnostic procedures and real-time monitoring systems which results in increased adoption of advanced technologies and stronger procurement activity in tertiary healthcare facilities.
Key Market Insights
- The Japan Intracranial Pressure Monitoring Devices Market reaches its highest point in 2025 because Kanto controls 38% of the market through its network of advanced tertiary hospitals which operate throughout Tokyo and Yokohama.
- Kansai represents the second-largest regional market with approximately 24% share, supported by strong neurosurgical infrastructure across Osaka and Kyoto.
- The Chubu region will experience its fastest growth until 2030 because healthcare systems modernize and new neurological treatment facilities open throughout the area.
- Invasive ICP monitoring systems dominate, contributing nearly 57% of Japan intracranial pressure monitoring devices industry size in 2025 due to clinical precision.
- External ventricular drainage systems hold a significant share, which emergency neurosurgical procedures use to support their operations.
- The segment of minimally invasive digital sensors shows the highest growth rate which will continue to rise until 2030 because its use results in decreased operational hazards.
- The market needs for emergency treatment will drive therapeutic brain injury solutions which account for 41% of total market demand in 2025.
- Hospitals with tertiary care facilities control 61% of the market for neurocritical care services which are offered at advanced neurocritical care units.
- The second-largest share of the market belongs to specialized neurology centers which experience increasing demand for their services.
- Private multi-specialty hospitals represent the fastest expanding end-user segment because they strategically develop their high-acuity medical services.
What are the Key Drivers, Restraints, and Opportunities in the Japan Intracranial Pressure Monitoring Devices Market?
The strongest force accelerating the Japan intracranial pressure monitoring devices market is the modernization of neurocritical care infrastructure across major hospitals. The shift began to develop after healthcare system capacity reviews which occurred after the COVID-19 period found deficiencies in continuous monitoring systems that should operate during high-acuity neurological patient care. The government-backed hospital digitalization programs together with the funding for intensive care system improvements led to the acquisition of advanced intracranial pressure monitoring systems which offer real-time data integration. The technology adoption rates at tertiary care hospitals improved because facilities selected technologies that offered faster diagnostic results and better surgical decision-making, which created continuous earnings through equipment upgrades and sensor consumption.
The most significant structural barrier is the high cost of installation and specialized clinical training required for invasive monitoring systems. The regional hospitals do not have the necessary neurosurgical expertise or intensive care infrastructure that enables safe operation of these devices. The challenge operates at a structural level because institutions need multiple years to develop trained neurocritical care personnel and enhance their hospital infrastructure. The adoption process remains limited to large urban medical centers which prevents secondary healthcare facilities from accessing the market and generates lost revenue opportunities.
The field of minimally invasive wireless monitoring technologies presents a significant opportunity for development. Japanese hospitals are currently testing compact sensor-based systems which enable them to conduct procedures with increased operational efficiency. The medical sector will experience growth through digital health integration investments that focus on Chubu and Kyushu's emerging medical centers.
What Has the Impact of Artificial Intelligence Been on the Japan Intracranial Pressure Monitoring Devices Market?
The International Neurocritical Care Society recognized the need for continuous brain monitoring methods which can offer real-time data analysis to support critical care treatment. The use of AI systems in advanced neurology departments enables automatic processing of intracranial sensor data to identify and track abnormal pressure changes while minimizing interference from background noise.
This system enables medical professionals to handle their duties more efficiently in intensive care units while it enhances their ability to react quickly during critical situations which include brain injuries and post-surgical swelling. The digital monitoring systems hospitals have installed show improved accuracy in tracking pressure changes which helps them make better decisions about patient treatment and achieve better results in dangerous situations.
Healthcare organizations now use machine learning models to develop predictive risk scoring systems which analyze past patient data to forecast intracranial hypertension attacks that will happen within the next few hours. The system enables better ICU resource distribution while decreasing emergency situations, which leads to reduced treatment expenses and faster bed availability in neurosurgical units.
The primary obstacle they face involves connecting AI technologies with existing hospital monitoring systems, which requires extensive effort to achieve successful results. The majority of hospitals maintain separate data systems, which create obstacles for real-time data sharing and lead to decreased diagnostic accuracy because medical data remains either incomplete or inconsistent. The deployment of AI-assisted medical decision tools across Japan's hospital network faces delays because of stringent security validation processes, which prevent hospitals from using these technologies even though they have great potential.
Key Market Trends
- The ICP monitoring devices market in Japan shifted from its exclusive use of invasive systems to its current state which combines both invasive systems and digital sensor-based platforms. The tertiary hospitals of Japan will achieve approximately 45% installed base of digital sensor-based platforms by 2025.
- Japanese neuroICU units implemented AI waveform analytics from 2023 to 2026 because hospitals experienced better outcomes through quicker IH detection and improved response capabilities.
- The rising number of cerebrovascular cases among Japan's senior citizens since 2020 has created an increased need for continuous ICP monitoring in stroke and trauma treatment facilities throughout the country.
- During the 2021 to 2022 period when semiconductor shortages occurred Japanese hospitals began acquiring medical devices from local manufacturers instead of their previous practice which involved purchasing neuro-monitoring equipment from overseas sources.
- The digital health policies of Japan's Ministry of Health established after 2022 public health scenarios enabled hospitals to expedite their approval processes for connected neurocritical monitoring devices.
- Between 2023 and 2026, Medtronic, Integra LifeSciences, and Natus expanded their partnerships in Japan to develop integrated neuro monitoring systems and their partnerships with intensive care units.
- Japanese hospitals improved their neurocritical care equipment after COVID-19 ICU stress testing which resulted in tertiary centers purchasing more real-time ICP monitoring systems.
- The adoption of minimally invasive ICP devices started to increase after 2024 as Japanese neurosurgical departments began to use them instead of traditional invasive catheters.
Japan Intracranial Pressure Monitoring Devices Market Segmentation
By Type :
The use of invasive monitoring devices in intracranial pressure management will continue because surgical insertion provides direct measurement, which delivers precise readings for critical neurological cases. The medical field utilizes these devices to monitor severe brain injuries because they provide continuous monitoring, which enables doctors to deliver precise treatment and medical response based on exact data.
Non-invasive monitoring devices will gain steady attention as they reduce infection risks and patient discomfort while still offering useful pressure estimates. The system relies on accessories and supporting components to maintain operational performance while ensuring system compatibility and enhanced reliability for both invasive and non-invasive monitoring systems.
By Application :
The need for intracranial pressure monitoring devices will increase because traumatic brain injury cases require medical professionals to obtain brain condition results within short timeframes for accurate treatment. Consistent monitoring enables hydrocephalus management because it helps doctors maintain control over fluid buildup and pressure fluctuations.
Doctors will need to monitor stroke-related cases continuously because they need to track brain swelling and secondary health problems. These devices will support neurosurgery applications during operations and post-operative care by providing doctors with safe pressure monitoring. Improved monitoring will help doctors make better clinical decisions while also keeping patients stable, which will benefit all neurological conditions.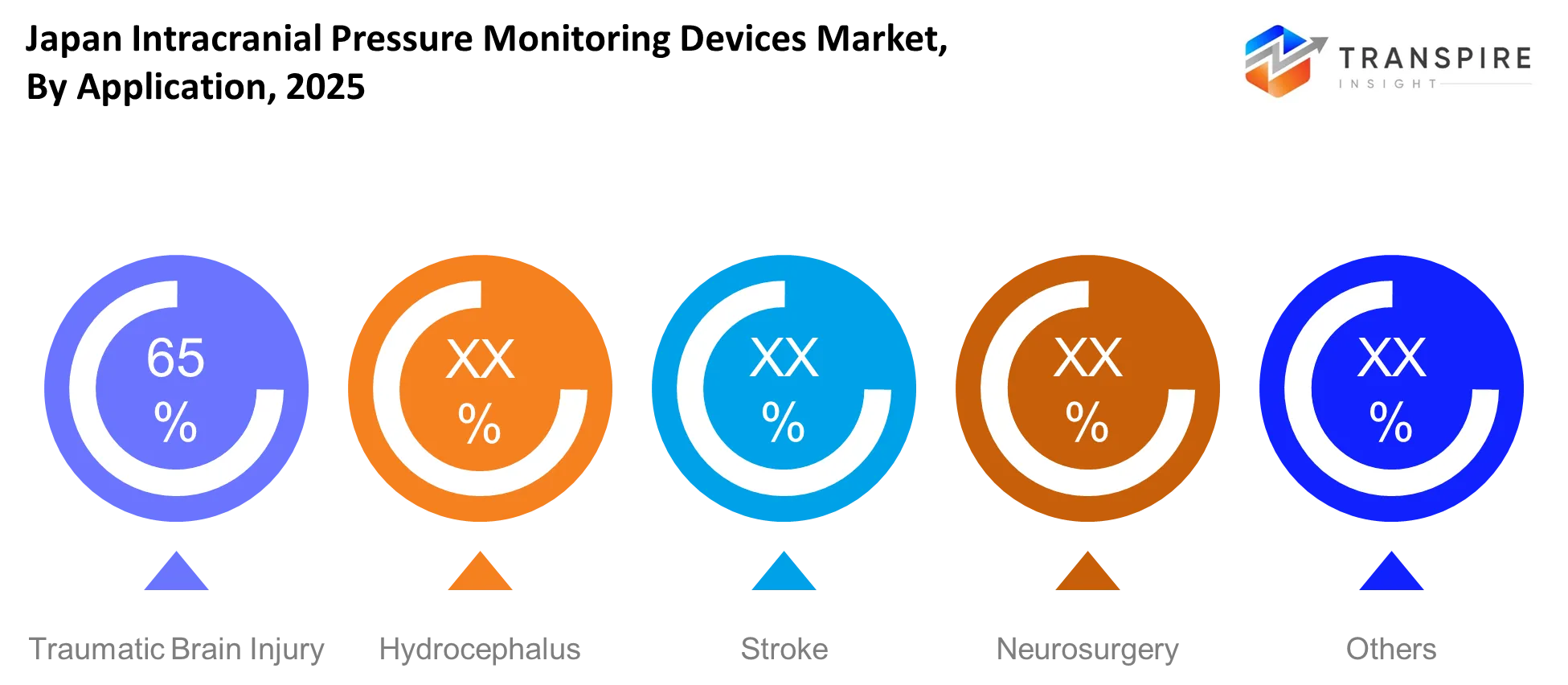
To learn more about this report, Download Free Sample Report
By End-User :
The primary users of emergency and intensive care units which treat critical brain injury cases will be hospitals because these units need to provide continuous patient monitoring. The advanced intracranial pressure systems will be used because the system requires both strong infrastructure and trained personnel to operate it.
The devices will be utilized by clinics and neurosurgical centers to deliver specialized neurological treatment and conduct follow-up assessments. Research institutes will drive technological progress through their work on testing new technologies while healthcare facilities will implement these systems when they recognize their clinical necessity.
By Technology :
Fiber optic sensors will support high-precision monitoring by delivering stable and accurate pressure readings even in complex clinical environments. The critical care field will choose these sensors because they demonstrate high sensitivity and dependable performance when monitoring brain pressure changes.
Microtransducers will maintain their status as popular devices because their small size allows users to measure results without needing extra equipment. Wireless monitoring technologies will gain attention for improving patient mobility and reducing wired system limitations which enable hospitals to transmit data and manage patients with greater flexibility.
What are the Key Use Cases Driving the Japan Intracranial Pressure Monitoring Devices Market?
The primary application of intracranial pressure monitoring devices in Japan medical facilities, which includes emergency departments and intensive care units, focuses on managing traumatic brain injuries. The highest rate of medical equipment usage at tertiary hospitals and neurocritical care centers occurs during cases that need constant monitoring of intracranial pressure to safeguard against secondary brain injury.
The medical field uses its applications for both stroke treatment and post-neurosurgery patient observation at major university medical centers and dedicated neurology research facilities. The Japanese healthcare system needs real-time pressure monitoring systems because the country's elderly population experiences increased cases of cerebrovascular diseases, which helps doctors to make faster decisions and improves surgical results during acute ischemic and hemorrhagic stroke treatment.
The new use cases include outpatient recovery monitoring through minimal invasive methods and establishing hybrid neuro-ICU environments, which allow remote data monitoring. Several advanced hospitals are testing portable sensor systems, which emergency transport personnel can use to identify neurotrauma cases at their initial stages, thus demonstrating the technology's potential to spread beyond standard ICU settings.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 97.85 Million |
|
Market size value in 2026 |
USD 107.34 Million |
|
Revenue forecast in 2033 |
USD 208.21 Million |
|
Growth rate |
CAGR of 9.93% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Japan |
|
Key company profiled |
Medtronic, Integra LifeSciences, Codman, Raumedic, Spiegelberg, Natus Medical, GE Healthcare, Philips, Siemens Healthineers, Boston Scientific, Abbott, B. Braun, NeuroWave, Sophysa, Tokai Medical |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Invasive Monitoring Devices, Non-invasive Monitoring Devices, Accessories, Others); By Application (Traumatic Brain Injury, Hydrocephalus, Stroke, Neurosurgery, Others); By End-User (Hospitals, Clinics, Neurosurgical Centers, Research Institutes, Others); By Technology (Fiber Optic Sensors, Microtransducers, Wireless Monitoring, Others) |
Which Regions are Driving the Japan Intracranial Pressure Monitoring Devices Market Growth?
The Japanese market for intracranial pressure monitoring devices exists within Tokyo and the larger Kanto region because those areas possess many advanced tertiary hospitals and national neuroscience research centers. The government provides substantial financial support which enables critical care facilities to improve their neuro-ICU equipment through ongoing upgrades at top medical centers. The equipment usage rates increase when hospitals treat more patients who need treatment for traumatic injuries and stroke conditions. The ecosystem benefits because university hospitals and medical device manufacturers and clinical training programs work together to advance monitoring technology adoption.
The Kansai region maintains its economic stability through established healthcare networks which operate across Osaka and Kyoto and their nearby prefectures. The region develops its strength through operational reliability while Kanto depends on its national institution concentration because private hospital networks and regional medical consortiums maintain continuous investment in the area. The hospitals in this area establish their procurement schedules while using standardized neurocritical care practices. The planned investment activities create consistent demand patterns for intracranial pressure monitoring systems which enables manufacturers to establish a dependable revenue stream from Kansai territory despite its lower innovation output compared to Tokyo.
The recent development of emergency services and hospital upgrades throughout Kyushu has transformed the region into its fastest-growing area. Fukuoka now has multiple stroke care units and trauma centers which create a need for advanced neurological monitoring systems that operate in real time. The government healthcare decentralization policies promote infrastructure development to areas outside of established metropolitan regions. The period from 2026 to 2033 will bring about a transformation which will draw medical device suppliers to discover new markets outside of their established urban areas.
Who are the Key Players in the Japan Intracranial Pressure Monitoring Devices Market and How Do They Compete?
The Japan market for intracranial pressure monitoring devices shows moderate competition because a limited number of global neuro-monitoring firms control hospital purchasing choices. Hospitals select systems which have received clinical validation and can work together with ICU monitoring systems while decreasing calibration errors that occur during critical care operations. Companies now compete based on their sensor accuracy and real-time data integration capabilities and the training assistance they provide instead of using price as their main competitive element. The purchasing behavior of tertiary care networks depends on two main factors: medical facilities require long-term service contracts and need to make their systems compatible with existing hospital digital networks.
Medtronic develops complete neurocritical care systems which combine intracranial pressure monitoring technology with intensive care unit operational information to help hospitals build permanent relationships within the Japanese healthcare system. Integra LifeSciences markets its trauma-focused ICP catheters and neurosurgical products to meet the needs of high-intensity emergency medical environments. Natus Medical employs neurodiagnostic integration to create a unique offering that connects ICP monitoring with EEG and multimodal brain function analysis which enables deeper clinical decision-making for complex neurological conditions.
Sophysa designs external ventricular drainage systems and hydrocephalus management devices which serve specific neurosurgical operations in Japanese university hospitals. Raumedic competes through high-precision fiber optic ICP sensors designed for continuous monitoring in critical care environments. Both companies expand their operations through distributor partnerships and clinical collaboration programs which they conduct with Japanese hospitals to achieve certification compliance and provide procedural training support for specialized neurosurgical departments.
Company List
- Medtronic
- Integra LifeSciences
- Codman
- Raumedic
- Spiegelberg
- Natus Medical
- GE Healthcare
- Philips
- Siemens Healthineers
- Boston Scientific
- Abbott
- B. Braun
- NeuroWave
- Sophysa
- Tokai Medical
Recent Development News
“In September 2025, researchers at multiple institutions published a fully automated intracranial pressure (ICP) grading framework integrating optic nerve sheath ultrasound analysis and clinical data fusion.” The study demonstrated improved accuracy in non-invasive ICP evaluation, highlighting the shift toward AI-assisted monitoring approaches that are increasingly relevant to Japan’s neurocritical care ecosystem. Source https://arxiv.org/
In January 2026, researchers introduced a neural network-based framework for intracranial pressure waveform analysis using attention mechanisms to improve diagnostic interpretation of ICP monitoring data.” The development supports next-generation signal interpretation tools that can be integrated into Japan’s hospital-based ICP monitoring systems for enhanced neurocritical care decision-making. Sourcehttps://arxiv.org/
What Strategic Insights Define the Future of the Japan Intracranial Pressure Monitoring Devices Market?
The Japan intracranial pressure monitoring devices market is moving toward a digitally integrated, minimally invasive monitoring ecosystem over the next 5 to 7 years. The hospital needs continuous neurological data streams which enable integration into ICU decision-support systems because Japan's aging population will increase cerebrovascular case volumes. Devices that cannot connect with real-time analytics systems will lose their usefulness in environments which require high-acuity medical care.
The main danger of centralized procurement arises from its tendency to concentrate purchasing power among major institutional hospitals. The shift toward centralized purchasing systems forces mid-tier device suppliers to decrease their bargaining power while facing increased pricing pressure. The lengthy regulatory validation process for AI-based medical devices will delay the rollout of advanced monitoring technologies which will result in clinical use delays that follow behind technological progress.
The development of portable intracranial monitoring systems which serve emergency transport and regional stroke networks in Kyushu and Shikoku's underserved regions represents an emerging business opportunity. Companies that align with Japan’s regional healthcare decentralization programs can capture early adoption advantages. Market participants should develop device platforms which use modular components and work with hospital digital systems to create future AI-based clinical workflows.
Japan Intracranial Pressure Monitoring Devices Market Report Segmentation
By Type
- Invasive Monitoring Devices
- Non-invasive Monitoring Devices
- Accessories
By Application
- Traumatic Brain Injury
- Hydrocephalus
- Stroke
- Neurosurgery
By End-User
- Hospitals
- Clinics
- Neurosurgical Centers
- Research Institutes
By Technology
- Fiber Optic Sensors
- Microtransducers
- Wireless Monitoring
Frequently Asked Questions
Find quick answers to common questions.
The Japan Intracranial Pressure Monitoring Devices Market size is USD 208.21 Million in 2033.
Key Segments for the Japan Intracranial Pressure Monitoring Devices Market are By Type (Invasive Monitoring Devices, Non-invasive Monitoring Devices, Accessories, Others); By Application (Traumatic Brain Injury, Hydrocephalus, Stroke, Neurosurgery, Others); By End-User (Hospitals, Clinics, Neurosurgical Centers, Research Institutes, Others); By Technology (Fiber Optic Sensors, Microtransducers, Wireless Monitoring, Others).
Major Japan Intracranial Pressure Monitoring Devices Market Players are Medtronic, Integra LifeSciences, Codman, Raumedic, Spiegelberg, Natus Medical, GE Healthcare, Philips, Siemens Healthineers, Boston Scientific, Abbott, B. Braun, NeuroWave, Sophysa, Tokai Medical.
The Current Japan Intracranial Pressure Monitoring Devices Market size is USD 97.85 Million in 2025.
The Japan Intracranial Pressure Monitoring Devices Market CAGR is 9.93% from 2026 to 2033.
- Medtronic
- Integra LifeSciences
- Codman
- Raumedic
- Spiegelberg
- Natus Medical
- GE Healthcare
- Philips
- Siemens Healthineers
- Boston Scientific
- Abbott
- B. Braun
- NeuroWave
- Sophysa
- Tokai Medical
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


