Japan Alkylating Agents Market Size & Forecast:
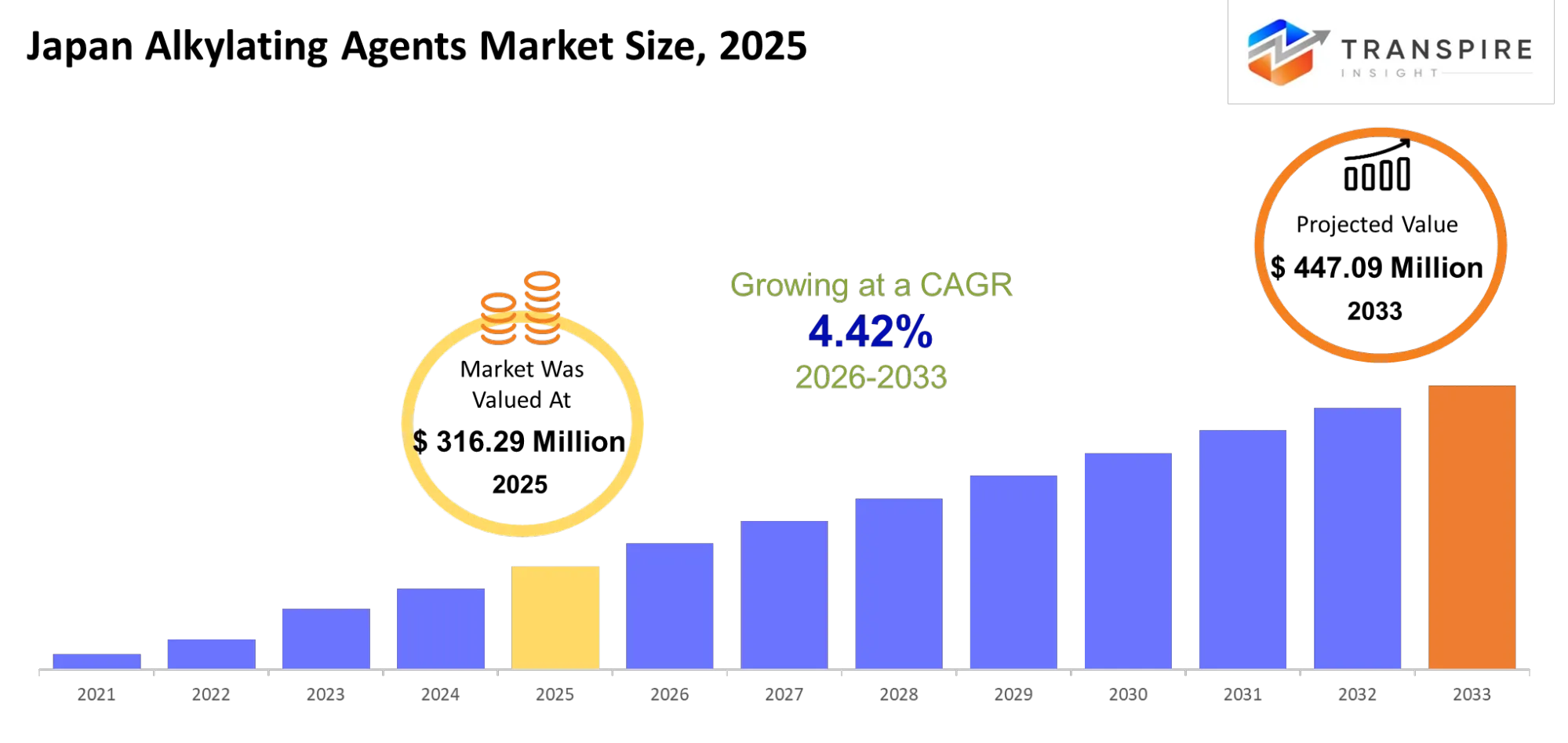
- Japan Alkylating Agents Market Size 2025: USD 316.29 Million
- Japan Alkylating Agents Market Size 2033: USD 447.09 Million
- Japan Alkylating Agents Market CAGR: 4.42%
- Japan Alkylating Agents Market Segments: By Type (Nitrogen Mustards, Nitrosoureas, Alkyl Sulfonates, Triazines, Others); By Application (Cancer Treatment, Chemotherapy, Oncology Research, Clinical Trials, Others); By End-User (Hospitals, Cancer Centers, Clinics, Research Institutes, Specialty Oncology Centers, Others); By Distribution (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Specialty Pharmacies, Others)
To learn more about this report, Download Free Sample Report
Japan Alkylating Agents Market Summary
The Japan Alkylating Agents Market was valued at USD 316.29 Million in 2025. It is forecast to reach USD 447.09 Million by 2033. That is a CAGR of 4.42% over the period.
The Japan Alkylating Agents Market underpins a critical segment of the country’s oncology treatment ecosystem by supplying chemotherapy drugs which disrupt cancer cell replication in hematologic cancers and brain tumors and aggressive solid malignancies that require rapid treatment. The market has evolved during the past three to five years because it now uses specific targeted combination regimens which include alkylating agents paired with immunotherapies and precision oncology through these two treatment methods.
The healthcare system faced two problems during the COVID-19 period when it experienced delayed cancer screenings and treatment backlogs which resulted in more patients receiving later stage diagnoses that required advanced treatment methods. Japan's growing elderly population has resulted in more patients who require specialized treatment for complex cancer cases. Healthcare organizations are acquiring more hospital supplies while investing in new drug formulations which offer better tolerability because these products lead to ongoing revenue growth as healthcare providers focus on effective treatments which have lower toxicity levels and enhanced patient management systems.
Key Market Insights
- The Japan Alkylating Agents Market is expanding because Japan experiences higher cancer rates which result in more than 1 million new cancer cases every year since 2023.
- The demand drivers for oncology drug procurement of tertiary care centers exist because hospitals are increasing their chemotherapy regimen implementation and their oncology drug purchasing efforts.
- The Japan alkylating agents industry underwent transformation through precision oncology development which enabled chemotherapy treatment success through its targeted treatment combination with hematologic and solid tumor therapies.
- Kanto dominates the Japan Alkylating Agents Market with a 2025 market share of 38 percent because Tokyo contains multiple advanced oncology hospitals.
- Chubu will experience its most rapid growth until 2031 because of its planned development of healthcare facilities and its upcoming enhancements to oncology services.
- The 2025 market shows 42% share for Nitrogen Mustards which demonstrates their common use in treating lymphoma and leukemia.
- The second biggest market segment consists of platinum-based alkylating agents which medical professionals use to treat lung and ovarian cancer.
- The most rapidly expanding treatment option uses Temozolomide-based therapies because doctors increasingly adopt glioblastoma treatment methods until 2030.
- Standardized chemotherapy regimens for hematologic malignancies account for approximately 36 percent of total market revenue in 2025.
- The Japanese market shows specialty oncology hospitals holding 54 percent market share because these facilities provide centralized cancer treatment across the country.
What are the Key Drivers, Restraints, and Opportunities in the Japan Alkylating Agents Market?
The fastest growing element of the Japan Alkylating Agents Market stems from two factors which include the aging Japanese population and the increasing need for oncology treatments. The Japanese healthcare system underwent five years of medical system modifications which now provide better funding for advanced cancer treatment.
The current policy framework enables hospitals to purchase more alkylating agents for use in their treatment of hematologic cancers and aggressive solid tumors. The post-pandemic recovery in cancer screening has further intensified this effect, as delayed diagnoses have translated into a larger share of late-stage cases requiring aggressive chemotherapy intervention.
The main obstacle which exists in the healthcare system arises from the ongoing toxic effects of alkylating agents, which restrict doctors from prescribing these medications to elderly patients with multiple health conditions. The blood pressure treatment problem requires permanent solutions because the current design of medication treatments needs both time and effort for scientists to develop new formulas and complete full testing. The treatment process in Japan needs to follow medication guidelines but severe treatment effects force clinicians to reduce dosages and switch patients to different therapies which results in financial losses for pharmaceutical companies and slow advancement of new treatments to the public.
The next-generation formulation development field which focuses on creating liposomal systems and targeted delivery alkylating agents represents a crucial opportunity for future growth. The Japanese pharmaceutical industry is investing in precision oncology platforms which will enable better chemotherapy treatments for patients. The research collaborations established between oncology centers in Tokyo and Osaka are developing new treatment methods which will benefit previously untreated patient groups.
What Has the Impact of Artificial Intelligence Been on the Japan Alkylating Agents Market?
Artificial intelligence is transforming the Japanese alkylating agents industry by its capability to speed up cancer treatment drug development and enhance the efficiency of pharmaceutical manufacturing processes. Japanese pharmaceutical companies are using AI-based process analytics systems to control the complete production process of alkylating compounds which includes monitoring batches and identifying impurities and managing quality assurance.
The systems evaluate reaction conditions throughout the production process which helps decrease production variations and leads to faster validation processes. Digital monitoring systems have enabled better production results and reduced batch failure rates between 10% and 15% in multiple advanced manufacturing facilities. Machine learning technology is enhancing predictive capabilities which analyze oncology treatment pathways.
Hospitals and research institutions use AI models to examine genomic data together with treatment-response histories in order to assist clinicians who need to determine which patients will gain from alkylating agent combination therapies. The system enables doctors to select better treatment options because it decreases the need for patients to undergo dangerous procedures while helping hospitals use their medical resources more efficiently. Supply chain predictive analytics systems enable manufacturers to forecast hospital purchasing schedules which results in less inventory interruptions.
These technologies create operational improvements through their ability to accelerate production processes and enhance regulatory compliance and optimize clinical trial design. The adoption process faces challenges because of two factors: high integration costs and insufficient availability of extensive Japanese oncology datasets which decrease algorithm performance in actual medical environments.
Key Market Trends
- The Japanese oncology centers have increased their use of alkylating agents in combination treatments since 2022 because they obtained clinical evidence for using immunotherapy as a treatment combination.
- The treatment requirements for cancer patients who recovered from medical care after 2021 shifted towards advanced-stage treatments, which led hospitals to focus on obtaining high-intensity chemotherapy medications instead of standard outpatient cancer treatment methods.
- Takeda Pharmaceutical Company and Chugai Pharmaceutical established precision oncology research partnerships which they expanded between 2023 and 2025 to enhance targeted chemotherapy treatment methods.
- The 2023 reimbursement changes in Japan led hospitals to start using high-value oncology treatments, which made manufacturers focus on developing new alkylating drugs that have better side effect control.
- Domestic manufacturers have reduced their need for foreign active pharmaceutical ingredients because they started producing these ingredients domestically after 2022 to address supply chain issues which revealed their purchasing weaknesses.
- Japanese pharmaceutical companies adopted AI-based production systems in their manufacturing plants after 2024, which led to a 10 to 15 percent reduction in product quality problems and faster equipment testing processes.
- The pattern of prescribing medications has changed since doctors now choose treatment methods based on blood test results instead of using standard chemotherapy procedures which were common practice before 2020.
- The academic oncology centers located in Tokyo and Osaka have increased their clinical trial operations since 2023 to expedite the testing of new alkylating treatment combinations which target treatment-resistant cancers.
- The international market competition has changed because pharmaceutical companies such as Pfizer and Bristol Myers Squibb expanded their regional licensing agreements to improve their access to the Japanese market.
- The post-market pharmacovigilance requirements which regulators established in 2022 have made it necessary for manufacturers to develop advanced systems that monitor real-world drug safety.
Japan Alkylating Agents Market Segmentation
By Type :
The Japan Alkylating Agents Market will show steady demand across different drug types used in cancer care. Nitrogen Mustards will remain widely used because they provide effective results in chemotherapy treatments. The combination of Nitrosoureas and Alkyl Sulfonates will help medical professionals treat patients who have rare or challenging cancers. Triazines and other forms will add variety for specific clinical needs and patient response.
Each type will serve different stages of cancer management because drug strength and patient condition determine which treatment to use. Hospitals and oncology centers will continue to select these agents based on treatment goals and safety profile and response rate. Research activity will also support gradual improvement in drug combinations which will lead to better outcomes.
By Application :
The primary field of application for cancer treatment will continue to use Alkylating Agents because they serve as the most effective method to control cancer cell growth. The drugs will function as essential components of chemotherapy treatment which will be used to treat various cancer types. The research in oncology will aim to develop drugs with better effectiveness while delivering fewer harmful effects.
The clinical trials will evaluate new treatment combinations and dosage methods to enhance patient results. Researchers will investigate Alkylating Agents for their potential use in supportive treatment applications beyond their current medical range. The ongoing medical research process will promote hospital and research institution implementation of new findings.
To learn more about this report, Download Free Sample Report
By End-User :
The primary field of application for cancer treatment will continue to use Alkylating Agents because they serve as the most effective method to control cancer cell growth. The drugs will function as essential components of chemotherapy treatment which will be used to treat various cancer types. The research in oncology will aim to develop drugs with better effectiveness while delivering fewer harmful effects.
The clinical trials will evaluate new treatment combinations and dosage methods to enhance patient results. Researchers will investigate Alkylating Agents for their potential use in supportive treatment applications beyond their current medical range. The ongoing medical research process will promote hospital and research institution implementation of new findings.
By Distribution :
Hospital Pharmacies will dominate distribution due to direct use of Alkylating Agents in chemotherapy settings. Specialty Pharmacies will control cancer drug distribution through their specialized storage and administration procedures. Retail Pharmacies will provide minimal service because they only handle prescribed medications.
Online Pharmacies will experience gradual growth yet will face ongoing limitations from regulatory requirements and safety standards. Distribution systems will depend on hospital-connected networks to maintain proper material handling and correct dosage delivery and protect patient safety during cancer treatments throughout medical facilities.
What are the Key Use Cases Driving the Japan Alkylating Agents Market?
The main purpose of the study focuses on cancer treatment in tertiary hospitals which uses alkylating agents to treat both hematologic cancers and aggressive solid tumors. The medical field considers the treatment method to be highly dependable because it successfully treats patients who need swift control of cancer cell growth during their advanced stage which requires them to undergo strong chemotherapy treatments.
Research facilities and specialized cancer treatment centers are starting to implement new technologies which enable them to conduct research on combination treatments that use immunotherapy and targeted biologic agents. Research organizations use clinical trial environments to test their improved drug dosing methods and safer drug formulations which they developed for use in brain cancer and treatment-resistant lymphoma patient care programs.
The development of new personalized cancer treatment methods which use genomic information and biomarker data for treatment choices exists as an emerging medical application. Japanese academic medical centers research alkylating agents to develop targeted conditioning methods for stem cell transplants which will become a big research area after Japan builds more cancer research facilities for clinical applications.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 316.29 Million |
|
Market size value in 2026 |
USD 330.27 Million |
|
Revenue forecast in 2033 |
USD 447.09 Million |
|
Growth rate |
CAGR of 4.42% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Japan |
|
Key company profiled |
Pfizer, Roche, Novartis, Bristol Myers Squibb, Merck, AstraZeneca, GSK, Sanofi, Takeda, AbbVie, Amgen, Eli Lilly, Johnson & Johnson, Bayer, Teva |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Nitrogen Mustards, Nitrosoureas, Alkyl Sulfonates, Triazines, Others); By Application (Cancer Treatment, Chemotherapy, Oncology Research, Clinical Trials, Others); By End-User (Hospitals, Cancer Centers, Clinics, Research Institutes, Specialty Oncology Centers, Others); By Distribution (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Specialty Pharmacies, Others) |
Which Regions are Driving the Japan Alkylating Agents Market Growth?
The Kanto region leads the Japan alkylating agents market because it has advanced oncology hospitals and pharmaceutical headquarters and effective national healthcare policy execution. Academic medical centers located in Tokyo handle the highest volume of complex cancer treatments which leads to continuous need for alkylating therapies. Market leadership exists because of reimbursement systems which support access to new oncology treatment protocols. The pharmaceutical research ecosystem develops through its clinical trial networks and regulatory systems which enable quick adoption of new drug treatments into medical practice.
The Kansai region holds the second-largest position, but growth dynamics differ from Kanto because strength comes from manufacturing depth and stable pharmaceutical investment rather than clinical concentration alone. The drug development and production facilities located in Osaka and Kyoto established a trustworthy supply chain which delivers oncology therapeutics. Local universities and pharmaceutical companies maintain consistent collaboration which drives ongoing development of new treatment methods. Kansai serves as a reliable source of national market revenue because its economic strength and institutional commitment prove strong even when healthcare budgets face reduction.
The Chubu region has become the fastest developing area because its cancer treatment facilities expanded and healthcare digital systems developed since 2024. The establishment of advanced oncology treatment centers through prefectural funding has enabled patients to receive specialized chemotherapy services beyond conventional city centers. The implementation of AI-based treatment planning combined with precision medicine programs has led to faster adoption of differentiated alkylating treatment methods. The period from 2026 until 2033 creates substantial market entry possibilities which extend to both targeted formulation development and regional pharmaceutical collaborations for investors.
Who are the Key Players in the Japan Alkylating Agents Market and How Do They Compete?
The Japanese market for alkylating agents exhibits moderate competition because multinational pharmaceutical companies and certain domestic manufacturers capture most of the market share. The competition between businesses depends more on their product development and research results and their ability to meet regulations and their capacity to introduce new oncology treatments than on pricing strategies.
Existing companies maintain their market presence through established hospital partnerships and their extensive oncology product offerings while new companies focus on precision oncology research and development of unique treatment methods. The market increasingly rewards companies that combine reliable manufacturing processes with unique delivery systems and established methods for validating their clinical results.
Through its oncology product range and its specialized clinical testing capabilities Takeda Pharmaceutical Company establishes a competitive advantage. Takeda establishes faster access to treatment-response data through its close partnerships with Japanese academic oncology institutions which enables them to develop targeted product improvements.
Chugai Pharmaceutical uses its biomarker-based oncology research and its partnership relationships to develop new treatment combinations which create unique market advantages. The expertise in precision medicine enables organizations to establish a stronger presence in targeted chemotherapy treatments which require personalized treatment plans.
The company uses its worldwide production capacity together with its comprehensive cancer treatment distribution network to achieve steady supply delivery throughout hospitals in Japan. The company establishes regional licensing agreements to expand its market reach while decreasing obstacles to regulatory approval.
The company develops new cancer treatment methods by combining alkylating agents with its immuno-oncology platforms. Nippon Kayaku establishes its market advantage through its specialized development of injectable cancer treatments and its ability to maintain production through domestic manufacturing during times when supply chain control becomes crucial.
Company List
- Pfizer
- Roche
- Novartis
- Bristol Myers Squibb
- Merck
- AstraZeneca
- GSK
- Sanofi
- Takeda
- AbbVie
- Amgen
- Eli Lilly
- Johnson & Johnson
- Bayer
- Teva
Recent Development News
"In February 2026, Sarepta Therapeutics and Chugai Pharmaceutical (Roche Group) launched ELEVIDYS in Japan following reimbursement listing under the National Health Insurance system. While not an alkylating agent itself, the launch reflects expanding high-cost oncology and rare disease therapy commercialization pathways that directly influence hospital oncology drug procurement structures where alkylating agents are also used.
Source https://www.nasdaq.com/
In March 2026, Pharming Group N.V. received Japanese Ministry of Health, Labour and Welfare approval for Joenja® (leniolisib) for APDS, expanding Japan’s pipeline of advanced specialty therapeutics entering oncology-adjacent treatment ecosystems. This approval reinforces regulatory acceleration trends that also benefit cytotoxic and alkylating agent approvals in hematology-oncology segments. Source https://www.globenewswire.com/
What Strategic Insights Define the Future of the Japan Alkylating Agents Market?
The Japan Alkylating Agents Market is undergoing a structural transition during its next five to seven years toward specialized chemotherapy systems which combine scientific accuracy with engineered medical treatments. The combination of genomic medicine research and the increasing need for cancer treatment among elderly patients and Japan's ongoing development of data-based cancer treatment systems is creating this movement. Commercial success in the future will require businesses to develop their complementary capabilities to match alkylating agents with specific treatment pathways that use biomarker information and combination immunotherapy methods.
The industry faces hidden dangers from technology replacement because advanced targeted therapies and cell-based oncology treatments are gradually decreasing the need for standard cytotoxic treatments in particular types of cancer. Manufacturers who continue to use existing products will experience revenue concentration compression. The development of AI-based formulation optimization together with the growth of Chubu-based oncology manufacturing hubs is creating new possibilities for the region which is currently improving its healthcare services.
Market participants should prioritize co-development partnerships with Japanese academic oncology centers and invest early in differentiated low-toxicity delivery systems. The suppliers who obtain clinical validation for precision-compatible formulations will achieve long-term business success because treatment standards will change over time.
Japan Alkylating Agents Market Report Segmentation
By Type
- Nitrogen Mustards
- Nitrosoureas
- Alkyl Sulfonates
- Triazines
By Application
- Cancer Treatment
- Chemotherapy
- Oncology Research
- Clinical Trials
By End-User
- Hospitals
- Cancer Centers
- Clinics
- Research Institutes
- Specialty Oncology Centers
By Distribution
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Specialty Pharmacies
Frequently Asked Questions
Find quick answers to common questions.
The Japan Alkylating Agents Market size is USD 447.09 Million in 2033.
Key segments for the Japan Alkylating Agents Market are By Type (Nitrogen Mustards, Nitrosoureas, Alkyl Sulfonates, Triazines, Others); By Application (Cancer Treatment, Chemotherapy, Oncology Research, Clinical Trials, Others); By End-User (Hospitals, Cancer Centers, Clinics, Research Institutes, Specialty Oncology Centers, Others); By Distribution (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Specialty Pharmacies, Others).
Major Japan Alkylating Agents Market players are Pfizer, Roche, Novartis, Bristol Myers Squibb, Merck, AstraZeneca, GSK, Sanofi, Takeda, AbbVie, Amgen, Eli Lilly, Johnson & Johnson, Bayer, Teva.
The Japan Alkylating Agents Market size is USD 316.29 Million in 2025.
The Japan Alkylating Agents Market CAGR is 4.42% from 2026 to 2033.
- Pfizer
- Roche
- Novartis
- Bristol Myers Squibb
- Merck
- AstraZeneca
- GSK
- Sanofi
- Takeda
- AbbVie
- Amgen
- Eli Lilly
- Johnson & Johnson
- Bayer
- Teva
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033