Japan Acute Inflammatory Demyelinating Polyneuropathy (AIDP) Guillain-Barré Syndrome Market Size & Forecast:
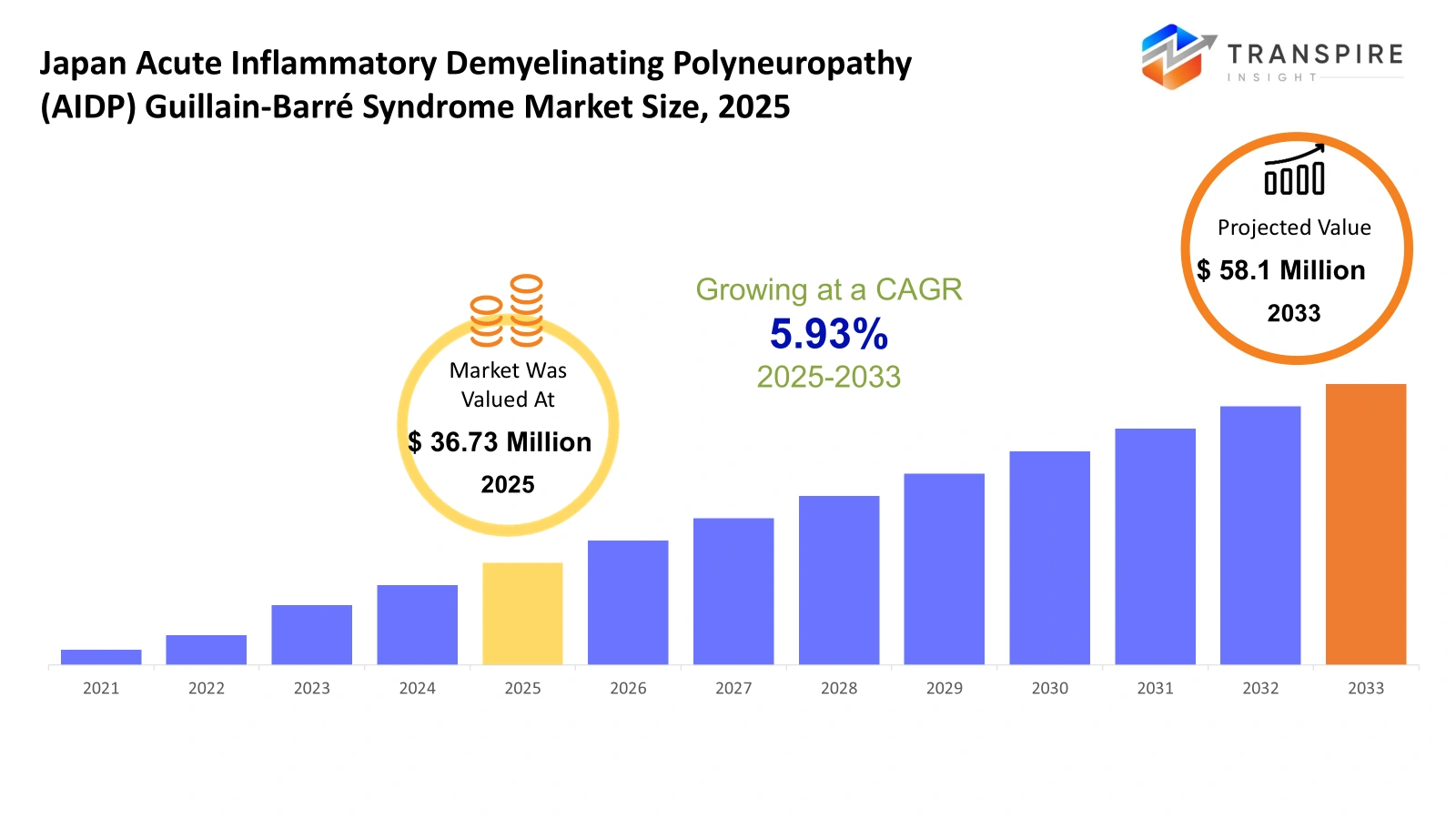
- Japan Acute Inflammatory Demyelinating Polyneuropathy (AIDP) Guillain-Barré Syndrome Market Size 2025: USD 36.73 Million
- Japan Acute Inflammatory Demyelinating Polyneuropathy (AIDP) Guillain-Barré Syndrome Market Size 2033: USD 58.1 Million
- Japan Acute Inflammatory Demyelinating Polyneuropathy (AIDP) Guillain-Barré Syndrome Market CAGR: 5.93%
- Japan Acute Inflammatory Demyelinating Polyneuropathy (AIDP) Guillain-Barré Syndrome Market Segments: By Treatment Type (Intravenous Immunoglobulin Therapy, Plasma Exchange Therapy, Corticosteroids), By End User (Hospitals, Speciality Clinics, Research Institutes).
To learn more about this report, Download Free Sample Report
Japan Acute Inflammatory Demyelinating Polyneuropathy (AIDP)Guillain-Barrée Syndrome Market Summary:
The Japan Acute Inflammatory Demyelinating Polyneuropathy (AIDP) Guillain-Barré Syndrome Market size is estimated at USD 36.73 Million in 2025. It is anticipated to reach USD 58.1 Million by 2033, growing at a CAGR of 5.93% from 2026 to 2033. The healthcare market of Japan shows special development through its most common form of Guillain-Barré Syndrome, Acute Inflammatory Demyelinating Polyneuropathy (AIDP). The country experiences improved patient results because people now understand more medical conditions while diagnostic methods become better and immunotherapy treatments develop. Japanese hospitals and neurological centers now prioritize early diagnosis together with quick treatment methods that use intravenous immunoglobulin and plasma exchange therapies. The combination of clinical research and government support for rare disease management drives innovation, which improves Japan's capacity to treat AIDP and other neurological disorders.
Key Market Trends & Insights:
- The Japanese healthcare system needs stronger capabilities which allow for earlier detection of neurological disorders. Advanced diagnostic technologies together with clinical knowledge improvements help doctors diagnose AIDP cases faster, which enables patients to receive necessary medical care without waiting.
- The Japanese medical system uses plasma exchange and intravenous immunoglobulin (IVIG) as standard treatment options. The therapies become more accessible to hospitals which results in hospitals providing better patient management while establishing consistent demand for the treatments.
- The Japanese healthcare system functions as an essential resource for managing AIDP and other uncommon neurological disorders. Specialised medical facilities together with skilled healthcare staff provide patients with appropriate medical treatment and rehabilitation solutions.
- Japanese research facilities, together with pharmaceutical companies, conduct research studies to examine neurological autoimmune disorders. The combination of clinical trials and academic partnerships works to create better treatment options and develop comprehensive knowledge about Guillain-Barré Syndrome.
- The Focus on Rehabilitation and Long-Term Care AIDP patients need physical therapy services to support their recovery process after they finish the most critical stage of their disease. The Japanese healthcare system is developing its capacity to provide complete neurological recovery support through the expansion of physiotherapy services and rehabilitation programs and long-term care solutions.
Japan Acute Inflammatory Demyelinating Polyneuropathy (AIDP) Guillain-barre Syndrome Market Segmentation
By Treatment Type
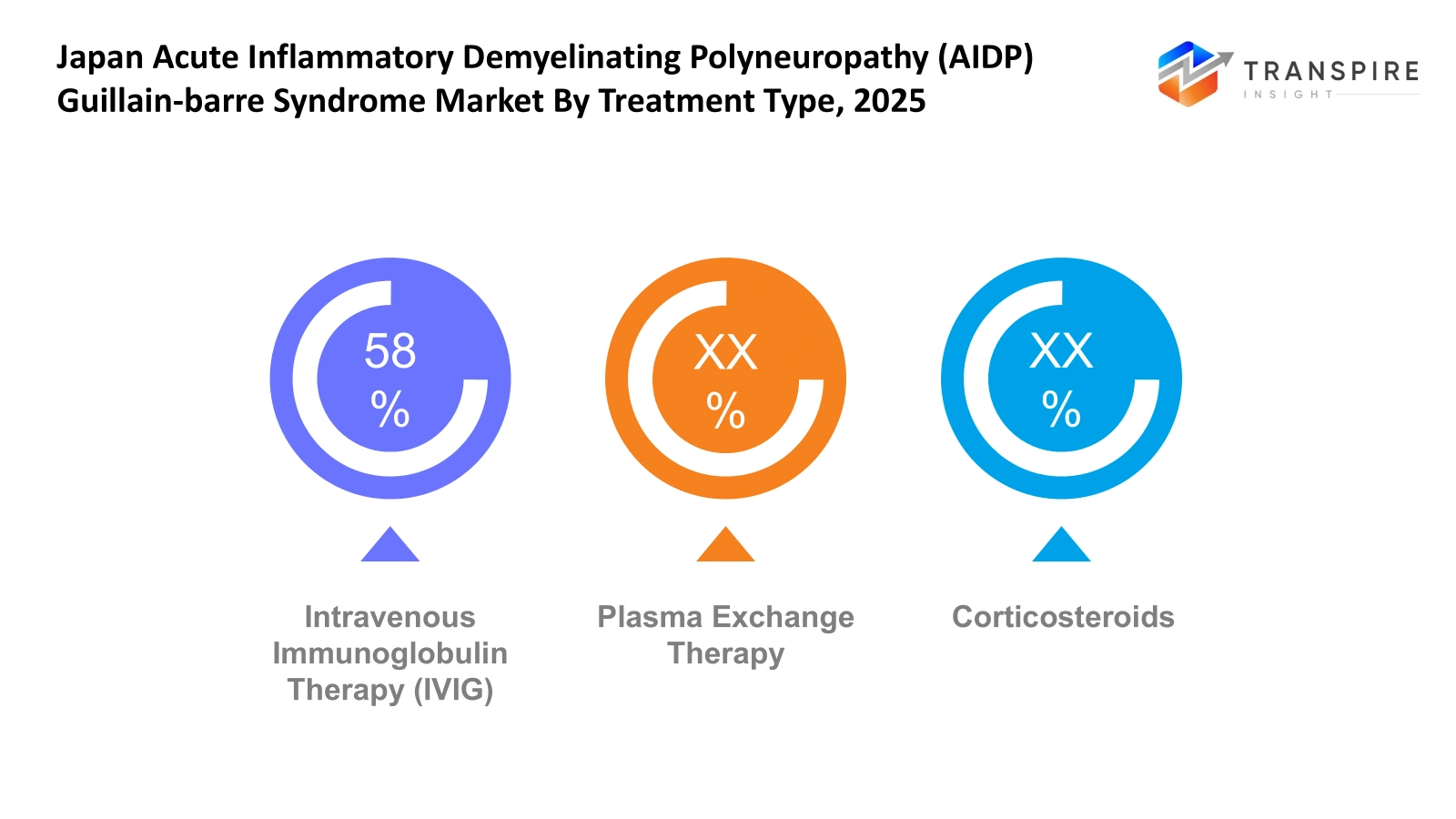
- Intravenous Immunoglobulin Therapy (IVIG): The medical procedure called intravenous immunoglobulin therapy serves as a standard treatment method which physicians use throughout Japan to treat patients who have acute inflammatory demyelinating polyradiculoneuropathy linked with Guillain-Barré Syndrome. The treatment method decreases immune system damage to nerves while helping patients heal more quickly. Hospitals choose intravenous immunoglobulin treatment because it produces effective results which medical professionals can deliver to patients with neurological autoimmune diseases through fast treatment methods.
- Plasma Exchange Therapy: The medical procedure known as plasma exchange therapy uses the method of plasmapheresis to treat AIDP patients throughout Japan. The medical procedure eliminates dangerous antibodies from the body to decrease nerve tissue swelling. The medical procedure takes place in specialised medical centres which possess advanced technology and skilled staff members who can deal with complicated brain disorders.
- Corticosteroids: Doctors use corticosteroids to treat autoimmune-related inflammation in some cases. The medical treatment AIDP shows primary treatment methods for patients, but healthcare professionals can use secondary treatment methods based on specific patient situations. The medical team assesses their benefits and drawbacks to use them in complete treatment plans for neurological disorders.
To learn more about this report, Download Free Sample Report
By Application
- Hospitals: The primary location for treating AIDP patients in Japan exists in hospitals. The majority of cases which need emergency medical treatment require both advanced diagnostic procedures and intensive medical care. The treatment of acute disease through IVIG therapy and plasma exchange procedures depends on the capabilities of large hospitals and their neurological departments.
- Specialty Clinics: Specialty clinics which specialise in neurology provide essential assistance to patients through their follow-up treatment and initial diagnostic assessments. The clinics work together with large hospitals to provide their patients with complete medical care. The process of rehabilitation supervision together with symptom management and recovery assessment helps the patient achieve better results in the future.
- Research Institutes: Japanese research institutes study autoimmune neurological disorders, which include AIDP as their primary area of research. The organisation conducts clinical trials while developing new treatments and studying Guillain-Barré Syndrome. The organisation develops new treatment methods while advancing the entire field of neurological treatment which exists in Japan.
Country Insights
The Japanese AIDP Guillain-Barré Syndrome market demonstrates both the nation's advanced healthcare system and its increasing research efforts on uncommon neurological disorders. Japanese healthcare facilities currently prioritise both early disease detection methods and successful treatment methods because of two main factors: the nation has an ageing population, and people show more understanding of autoimmune disorders. The advanced medical facilities, together with the skilled neurologists, provide necessary medical care that helps doctors treat AIDP conditions while preventing serious health issues.
Japanese hospitals implement intravenous immunoglobulin therapy together with plasma exchange procedures as their primary methods for aiding patient recovery. Government funding for rare disease studies together with academic-pharmaceutical partnerships for research has created an environment that fosters breakthroughs in neurological disorder treatment. Research initiatives currently work to enhance knowledge about immune-mediated nerve disorders and their therapeutic results.
Japan's healthcare system, together with its medical research activities and patient treatment approaches, enables better treatment of Guillain-Barré Syndrome while advancing the development of this specialised medical field.
Recent Development News
Experimental Drug ANX005 Shows Positive Results in Late-Stage Guillain-Barré Syndrome Trial.
Post-Marketing Studies Raise Awareness of Guillain-Barré Syndrome Risk Following RSV Vaccination.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 36.73 Million |
|
Market size value in 2026 |
USD 38.82 Million |
|
Revenue forecast in 2033 |
USD 58.1 Million |
|
Growth rate |
CAGR of 5.93% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Japan |
|
Key company profiled |
Takeda Pharmaceutical Company, CSL Behring, Grifols SA, Octapharma AG, Pfizer Inc., F. Hoffmann-La Roche Ltd., Novartis AG, Sanofi SA, Baxter International Inc., Kedrion Biopharma, Biotest AG, ADMA Biologics, Emergent BioSolutions, AstraZeneca PLC, Bayer AG. |
|
Customization scope |
Free report customization (country, regional & segment scope). AvAIl customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Treatment Type (Intravenous Immunoglobulin Therapy, Plasma Exchange Therapy, Corticosteroids), By End User (Hospitals, Specialty Clinics, Research Institutes). |
Key Japan Acute Inflammatory Demyelinating Polyneuropathy (AIDP) Guillain-barre Syndrome Company Insights
The Japan Acute Inflammatory Demyelinating Polyneuropathy (AIDP) Guillain-Barré Syndrome market involves a mix of pharmaceutical companies, biotechnology firms, and healthcare institutions which are developing better treatments for this uncommon neurological disorder. The companies are developing treatments which include intravenous immunoglobulin and advanced immunomodulatory therapies to treat immune-related nerve damage. Many organisations are investing their resources into clinical research while building partnerships with medical institutions to test new treatment methods. The Japanese regulatory system, together with its modern healthcare system, enables companies in this industry to enhance patient results while contributing to the development of neurological care practices.
Company List
- Takeda Pharmaceutical Company
- CSL Behring
- Grifols SA
- Octapharma AG
- Pfizer Inc.
- F. Hoffmann-La Roche Ltd
- Novartis AG
- Sanofi SA
- Baxter International Inc
- Kedrion Biopharma
- Biotest AG
- ADMA Biologics
- Emergent BioSolutions
- AstraZeneca PLC
- Bayer AG
Japan Acute Inflammatory Demyelinating Polyneuropathy (AIDP) Guillain-Barré Syndrome Market Report Segmentation
By Treatment Type
- Intravenous Immunoglobulin Therapy
- Plasma Exchange Therapy
- Corticosteroids
By End User
- Hospitals
- Specialty Clinics
- Research Institutes
Frequently Asked Questions
Find quick answers to common questions.
The approximate Japan Acute Inflammatory Demyelinating Polyneuropathy (AIDP) Guillain-Barré Syndrome Market size will be USD 58.1 Million in 2033.
The key segments of the Japan Acute Inflammatory Demyelinating Polyneuropathy (AIDP) Guillain-Barré Syndrome Market are: By Treatment Type (Intravenous Immunoglobulin Therapy, Plasma Exchange Therapy, Corticosteroids); By End User (Hospitals, Speciality Clinics, Research Institutes).
Major players in the Japan Acute Inflammatory Demyelinating Polyneuropathy (AIDP) Guillain-Barré Syndrome Market are Takeda Pharmaceutical Company, CSL Behring, Grifols SA, Octapharma AG, Pfizer Inc., F. Hoffmann-La Roche Ltd., Novartis AG, Sanofi SA, Baxter International Inc., Kedrion Biopharma, Biotest AG, ADMA Biologics, Emergent BioSolutions, AstraZeneca PLC, and Bayer AG.
The current market size of the Japan Acute Inflammatory Demyelinating Polyneuropathy (AIDP) Guillain-Barré Syndrome Market is USD 36.73 Million in 2025.
The Japan Acute Inflammatory Demyelinating Polyneuropathy (AIDP) Guillain-Barré Syndrome Market CAGR is 5.93%.
- Takeda Pharmaceutical Company
- CSL Behring
- Grifols SA
- Octapharma AG
- Pfizer Inc.
- F. Hoffmann-La Roche Ltd.
- Novartis AG
- Sanofi SA
- Baxter International Inc.
- Kedrion Biopharma
- Biotest AG
- ADMA Biologics
- Emergent BioSolutions
- AstraZeneca PLC
- Bayer AG.
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


