Germany Pharmaceutical Contract Manufacturing Market Size & Forecast:
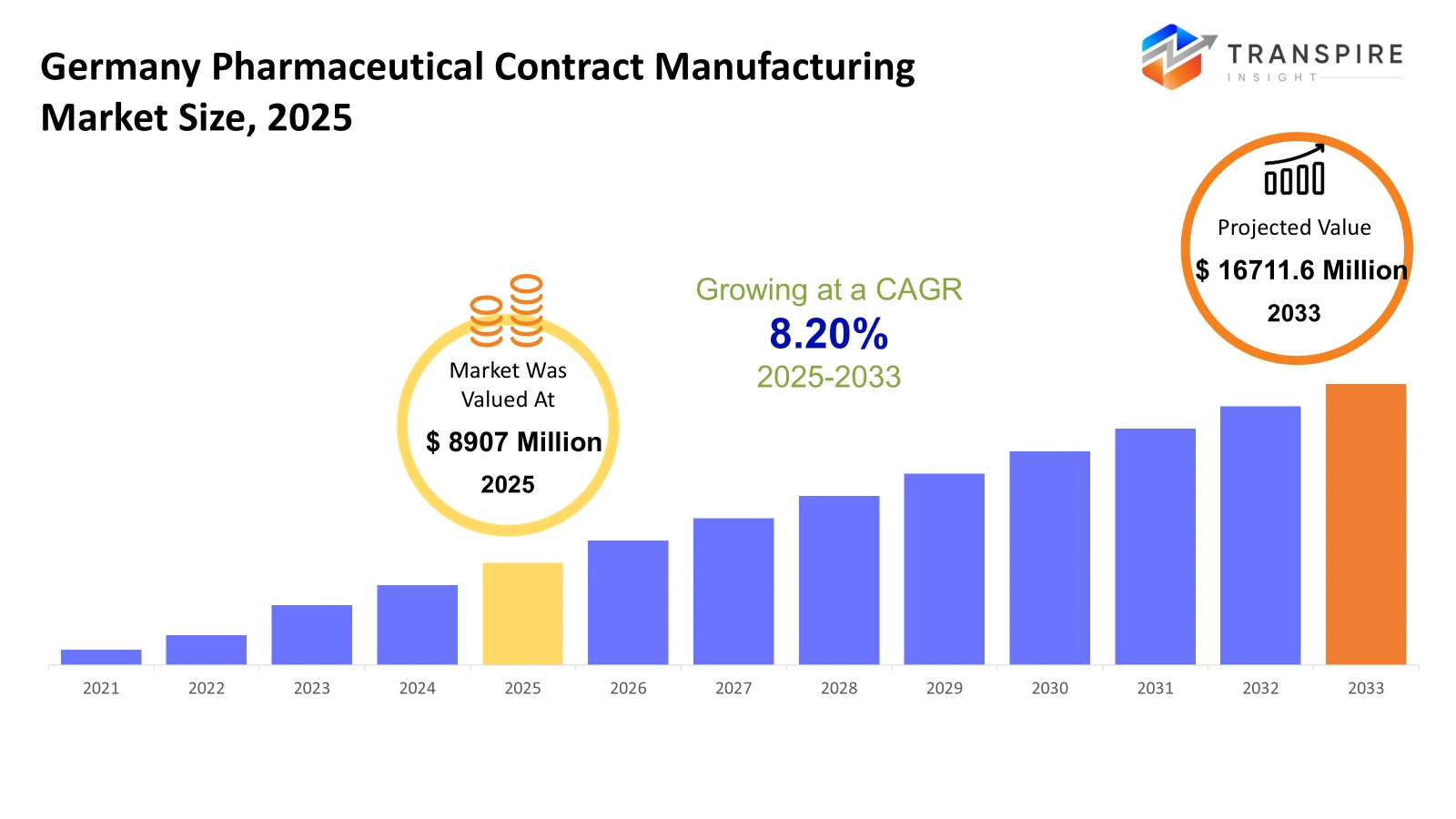
- Germany Pharmaceutical Contract Manufacturing Market Size 2025: USD 8907 Million
- Germany Pharmaceutical Contract Manufacturing Market Size 2033: USD 16711.6 Million
- Germany Pharmaceutical Contract Manufacturing Market CAGR: 8.20%
- Germany Pharmaceutical Contract Manufacturing Market Segments: by Service Type (API Manufacturing, Finished Dosage Form Manufacturing, Packaging Services), by Drug Type (Small Molecule Drugs, Biologics),by End User (Pharmaceutical Companies, Biotechnology Companies).
To learn more about this report, Download Free Sample Report
Germany Pharmaceutical Contract Manufacturing Market Summary:
TheGermany Pharmaceutical Contract Manufacturing Market size was estimated at USD 9628.5 Millionin 2026 and is anticipated to reach USD 16711.6 Million by 2033, growing at a CAGR of 8.20% from 2026 to 2033.
The pharmaceutical contract manufacturing market in Germany operates as a structured system which connects drug developers with specialized manufacturing partners for their research-based drug development needs. German pharmaceutical innovators will use flexible production methods which enable them to turn laboratory discoveries into commercial products without needing to operate extensive production equipment throughout their entire manufacturing process. Drug developers will search for partners who can produce complex formulations which include sterile injectables and biologics and small-batch specialty medicines as research pipelines grow and treatment options become more precise. The expectation of patients and healthcare providers for faster medicine delivery will create pressure on pharmaceutical companies to reduce their production timeframes while they continue to uphold their quality control protocols. Manufacturing partners will make substantial investments into their advanced facilities which feature automation and digital monitoring systems to achieve precise production tracking capabilities. These technologies enable manufacturers to achieve stable product quality while simultaneously decreasing material waste and handling complex therapies which demand precise environmental management.
The manufacturing industry in Germany and the European Union will undergo structural changes through the implementation of regulatory standards. The European Medicines Agency together with the Federal Institute for Drugs and Medical Devices will implement stricter regulations to control production procedures and product traceability and supply chain integrity, which will lead contract manufacturers to establish more effective compliance programs and complete documentation systems. Pharmaceutical companies will choose their partners based on two requirements, which include proof of reliable supply chains and environmental sustainability and capacity to support emerging treatment modalities that include gene therapies and advanced biologics. The adoption of digital manufacturing platforms with real-time quality control systems and modular production lines will lead to a gradual transformation of pharmaceutical drug production methods used in Germany. External manufacturing providers will need to develop advanced technical capabilities and operational efficiency because the manufacturing industry will establish new requirements for them. The pharmaceutical industry in the country will rely more on collaborative production systems because healthcare organizations need quicker access to medicines that meet safety and effectiveness standards, which makes contract manufacturers essential for developing and delivering upcoming therapeutic solutions.
Key Market Trends & Insights:
- The pharmaceutical industry experiences increased outsourcing requirements because drug developers face demands to lower their production expenses and expedite their product development processes. The Germany Pharmaceutical Contract Manufacturing Market sees multiple drug developers selecting third-party manufacturing companies to handle their product development processes which include formulation creation and packaging design and production execution. The partnership enables companies to use their resources effectively while delivering product quality that meets established standards across all treatment areas.
- The manufacturing sector for biologics and advanced therapeutic products continues to grow because researchers increasingly investigate biologics and cell-based treatments and complex injectable treatment options. Manufacturing companies throughout Germany establish sterile production facilities together with cutting-edge bioprocessing technology and tightly controlled research environments. The growing number of biologic drug pipelines results in constant work opportunities for the Germany Pharmaceutical Contract Manufacturing Market.
- The pharmaceutical industry in Germany operates under European regulatory standards which require businesses to establish complete systems for maintaining product quality and regulatory compliance. Contract manufacturing providers maintain detailed quality management systems, validated equipment, and continuous monitoring of production processes. International pharmaceutical companies build long-term production contracts in Germany because strong regulatory standards boost their confidence in German manufacturing services.
- Contract manufacturers expand their production capacity through facility upgrades and automated production lines and digital monitoring tools. Advanced manufacturing technologies enable companies to achieve higher production efficiency while decreasing their operational downtime. The current infrastructure enables pharmaceutical companies to conduct both small-batch clinical production and large-scale commercial supply activities.
- Supply chain stability and regional manufacturing preference: Global supply disruptions during recent years encouraged pharmaceutical companies to seek reliable European production partners. Germany offers strong infrastructure, skilled technical workforce, and dependable logistics networks. Regional manufacturing partnerships support faster distribution across European healthcare markets while strengthening supply continuity for essential pharmaceutical products.
Germany Pharmaceutical Contract Manufacturing Market Segmentation
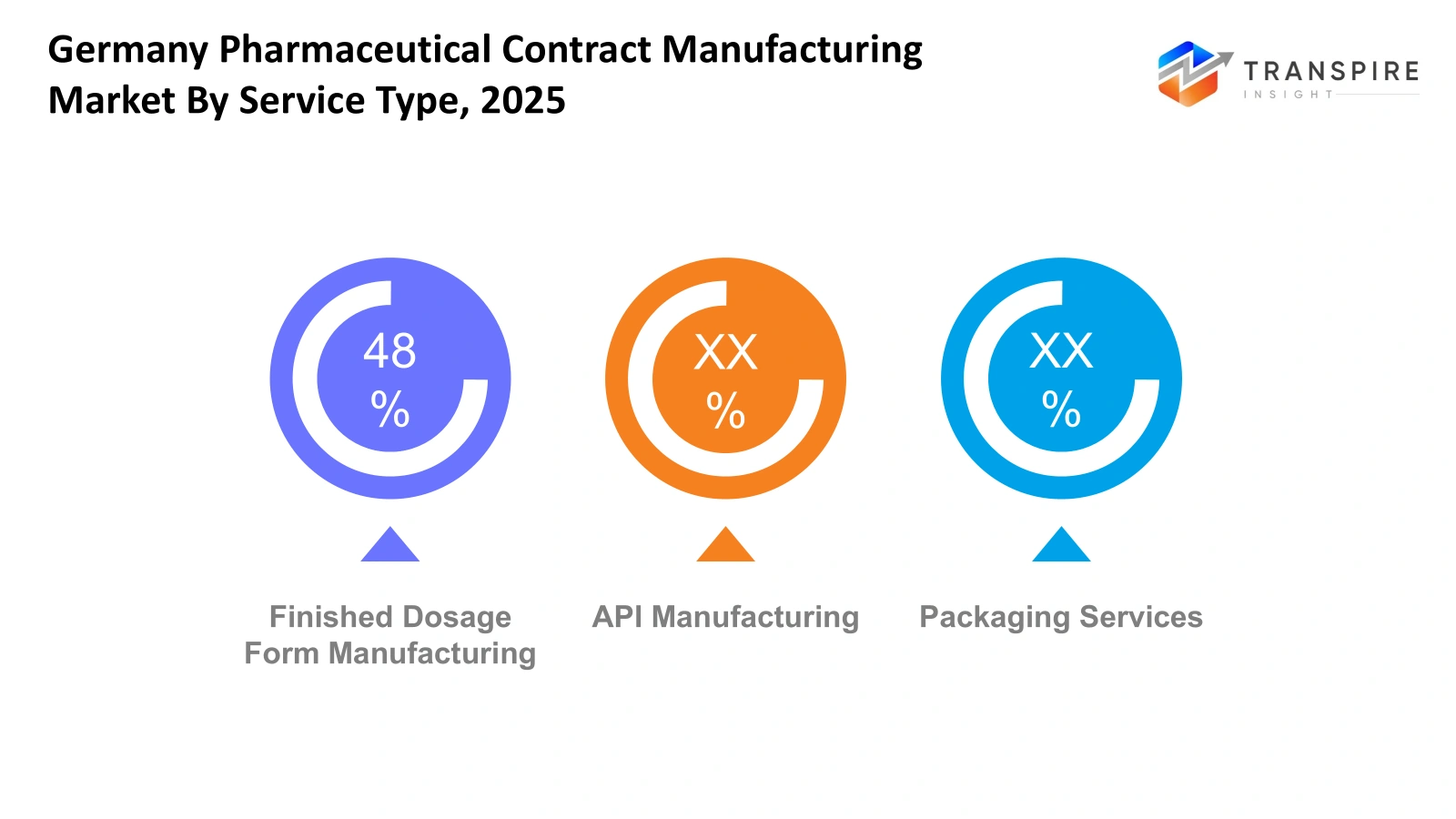
By Service Type :
- API Manufacturing: Germany Pharmaceutical Contract Manufacturing Market shows strong activity in API manufacturing as pharmaceutical production requires specialized facilities and strict regulatory compliance. Contract manufacturers will handle complex chemical synthesis, purification, and quality testing. German infrastructure, experienced workforce, and strong regulatory standards will support reliable API supply for global pharmaceutical production.
- Finished Dosage Form Manufacturing: Germany Pharmaceutical Contract Manufacturing Market will see steady demand for finished dosage form manufacturing as pharmaceutical firms depend on external partners to convert active ingredients into tablets, capsules, injectables, and other formats. Contract facilities will manage formulation development, scaling of production, and quality control to ensure consistent drug availability for domestic and international markets.
- Packaging Services: Germany Pharmaceutical Contract Manufacturing Market will also expand through packaging services that ensure drug safety, regulatory labeling, and efficient distribution. Contract providers will offer blister packaging, bottle filling, labeling, and serialization systems that meet strict European pharmaceutical standards. Reliable packaging operations will help pharmaceutical firms maintain product integrity during storage and transportation.
To learn more about this report, Download Free Sample Report
By Drug Type :
- Small Molecule Drugs: Germany Pharmaceutical Contract Manufacturing Market will continue to rely heavily on small molecule drug production because many widely used medicines still depend on chemical synthesis. Contract manufacturers will support large scale production, process optimization, and quality monitoring. Strong chemical manufacturing experience in Germany will support stable supply for therapeutic products across multiple disease areas.
- Biologics: Germany Pharmaceutical Contract Manufacturing Market will witness rising interest in biologics manufacturing as biotechnology based treatments expand in modern healthcare. Contract facilities will invest in advanced bioprocessing systems, cell culture technology, and sterile production environments. Growing research activity and demand for targeted therapies will strengthen biologics manufacturing services within the German pharmaceutical industry.
By End User :
- Pharmaceutical Companies: Germany Pharmaceutical Contract Manufacturing Market will experience significant participation from pharmaceutical companies that rely on contract manufacturers to manage production workloads. Outsourcing will allow pharmaceutical firms to focus on research, product development, and regulatory strategy while specialized manufacturers handle large scale production and compliance with strict quality standards.
- Biotechnology Companies: Germany Pharmaceutical Contract Manufacturing Market will gain increasing demand from biotechnology companies that require advanced manufacturing support for innovative therapies. Contract manufacturing partners will provide specialized equipment, controlled production environments, and regulatory expertise. Such collaboration will help biotechnology firms move treatments from laboratory development to commercial scale production efficiently.
Country Insights
- Germany pharmaceutical contract manufacturing market will continue to grow due to a strong pharmaceutical base and well-developed industrial systems. Advanced laboratories, strict regulatory systems, and a stable manufacturing environment support reliable drug production. Pharmaceutical companies often depend on contract partners for large-scale production, formulation work, and packaging services. High standards in manufacturing practices and careful quality monitoring support international demand. Strong logistics networks and skilled technical workers also help maintain smooth supply chains, making Germany a dependable location for pharmaceutical contract production and long-term industry collaboration.
- Germany Pharmaceutical Contract Manufacturing Market: Growth in the Germany pharmaceutical contract manufacturing market will also be influenced by the presence of experienced pharmaceutical firms and specialized service providers. Contract manufacturers support small and large drug developers that require additional production capacity. Research organizations and biotechnology firms depend on these partners to move laboratory discoveries toward commercial production. Clear regulatory frameworks and reliable certification processes support trust between manufacturers and pharmaceutical developers. Industrial clusters located near research centers strengthen collaboration, helping companies scale production while maintaining strict quality expectations.
- Demand within the Germany pharmaceutical contract manufacturing market will remain supported by the rising need for complex medicines, biologics, and advanced drug delivery systems. Pharmaceutical developers often choose external manufacturing partners to manage specialized production equipment and controlled environments. Germany offers strong infrastructure for sterile manufacturing, active ingredient production, and advanced formulation work. Export opportunities across Europe and other global regions strengthen production volumes. Stable economic conditions and a strong healthcare system also support continuous demand for high-quality pharmaceutical manufacturing services.
Recent Development News
Capacity expansion at existing German CDMO facilities.The company also announced capacity expansions at its Ravensburg and Langenargen facilities, aiming to increase pharmaceutical filling and packaging services for global drug manufacturers outsourcing production.
Growing investor interest in pharmaceutical contract manufacturing. Financial investors have increasingly targeted pharmaceutical contract manufacturers due to strong demand for outsourced production. Private-equity firms have pursued major deals and acquisitions in the sector, highlighting the strong growth outlook for CDMOs globally and in Europe.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 8907 Million |
|
Market size value in 2026 |
USD 9628.5 Million |
|
Revenue forecast in 2033 |
USD 16711.6 Million |
|
Growth rate |
CAGR of 8.20% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Germany |
|
Key company profiled |
Lonza Group, Catalent Inc., Recipharm AB, Boehringer Ingelheim, Siegfried Holding AG, Thermo Fisher Scientific (Patheon), Fareva Group, Samsung Biologics, WuXi AppTec, Piramal Pharma Solutions, CordenPharma, AbbVie Contract Manufacturing, Baxter BioPharma Solutions, Aenova Group, Delpharm. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Service Type (API Manufacturing, Finished Dosage Form Manufacturing, Packaging Services), by Drug Type (Small Molecule Drugs, Biologics),by End User (Pharmaceutical Companies, Biotechnology Companies). |
Key Germany Pharmaceutical Contract Manufacturing Company Insights
- The German Pharmaceutical Contract Manufacturing Market will experience active business operations from firms that deliver development manufacturing and packaging solutions to pharmaceutical manufacturers. Contract manufacturers will assist drug developers who require production flexibility without constructing additional facilities. The partnerships will control expenses while they assist in delivering products to various treatment areas at a faster rate.
- The Germany Pharmaceutical Contract Manufacturing Market will expand because manufacturing partners possess high technical capabilities. The facilities will establish operations for sterile production and biologics handling and advanced dosage form manufacturing while adhering to European quality standards. These partners will provide pharmaceutical companies with dependable production capabilities and necessary regulatory compliance and reliable supply chains which enable the companies to distribute medicines throughout Germany and other European regions.
Germany Pharmaceutical Contract Manufacturing Market Companies:
- Lonza Group
- Catalent Inc.
- Recipharm AB
- Boehringer Ingelheim
- Siegfried Holding AG
- Thermo Fisher Scientific (Patheon)
- Fareva Group
- Samsung Biologics
- WuXi AppTec
- Piramal Pharma Solutions
- CordenPharma
- AbbVie Contract Manufacturing
- Baxter BioPharma Solutions
- Aenova Group
- Delpharm
Germany Pharmaceutical Contract Manufacturing Market Segmentation
By Service Type
- API Manufacturing
- Finished Dosage Form Manufacturing
- Packaging Services
By Drug Type
- Small Molecule Drugs
- Biologics
By End User
- Pharmaceutical Companies
- Biotechnology Companies
Frequently Asked Questions
Find quick answers to common questions.
The approximate Germany Pharmaceutical Contract Manufacturing Market size for the market will be USD 16711.6 Million in 2033.
Key segments for the Germany Pharmaceutical Contract Manufacturing Market are by Service Type (API Manufacturing, Finished Dosage Form Manufacturing, Packaging Services), by Drug Type (Small Molecule Drugs, Biologics), by End User (Pharmaceutical Companies, Biotechnology Companies).
Major Germany Pharmaceutical Contract Manufacturing Market players are Lonza Group, Catalent Inc., Recipharm AB, Boehringer Ingelheim, Siegfried Holding AG, Thermo Fisher Scientific (Patheon), Fareva Group, Samsung Biologics, WuXi AppTec, Piramal Pharma Solutions, CordenPharma, AbbVie Contract Manufacturing, Baxter BioPharma Solutions, Aenova Group, Delpharm.
The Germany Pharmaceutical Contract Manufacturing Market size is USD 8907 Million in 2025.
The Germany Pharmaceutical Contract Manufacturing Market CAGR is 8.20%.
- Lonza Group
- Catalent Inc.
- Recipharm AB
- Boehringer Ingelheim
- Siegfried Holding AG
- Thermo Fisher Scientific (Patheon)
- Fareva Group
- Samsung Biologics
- WuXi AppTec
- Piramal Pharma Solutions
- CordenPharma
- AbbVie Contract Manufacturing
- Baxter BioPharma Solutions
- Aenova Group
- Delpharm
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


