Germany Biological Safety Testing Products And Services Market Size & Forecast:
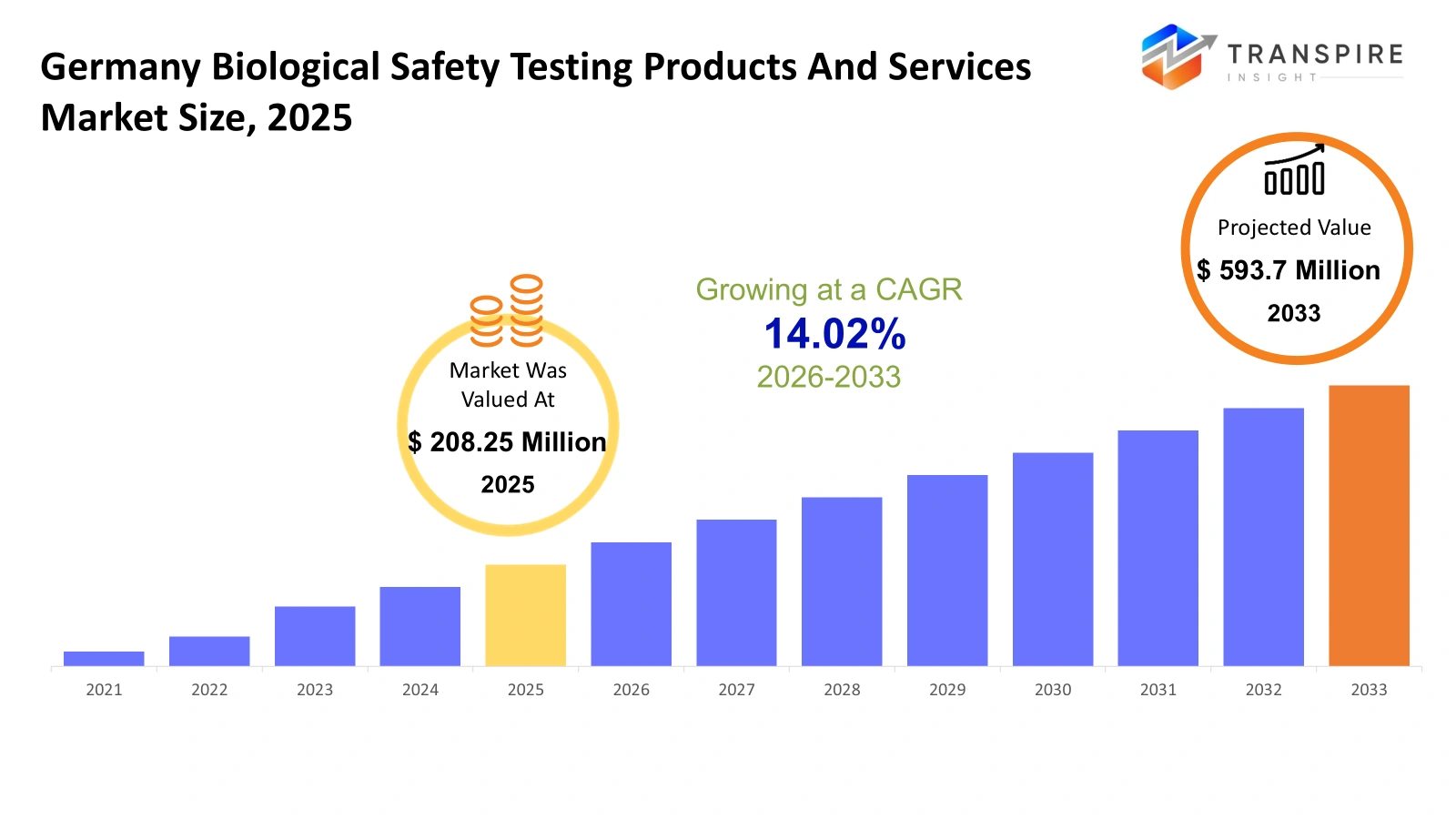
- Germany Biological Safety Testing Products And Services Market Size 2025: USD 208.25 Million
- Germany Biological Safety Testing Products And Services Market Size 2033: USD 593.7 Million
- Germany Biological Safety Testing Products And Services Market CAGR: 14.02%
- Germany Biological Safety Testing Products And Services Market Segments: by Product & Service (Instruments, Reagents & Kits, Services), Test Type (Sterility Testing, Bioburden Testing, Endotoxin Testing, Cell Line Authentication), Application (Vaccines & Therapeutics, Medical Devices, Blood & Blood Products), End User (Pharmaceutical & Biotechnology Companies, CROs, Academic Institutes
To learn more about this report, Download Free Sample Report
Germany Biological Safety Testing Products And Services Market Summary:
The Germany Biological Safety Testing Products And Services Market size is estimated at USD 208.25 Million in 2025 and is anticipated to reach USD 593.7 Million by 2033, growing at a CAGR of 14.02 % from 2026 to 2033. The Germany Biological Safety Testing Products and Services Market is an important segment of the life sciences and laboratory testing industry in Europe. This segment involves products and services that ensure the safety, purity, and quality of biological products such as biologics, vaccines, cell and gene therapies, tissue-based therapeutics, and other biological materials.
Key Market Trends & Insights:
- Strong Growth Driven by Bio pharma Expansion: The market is witnessing significant growth, and the main reason is the increased activities related to the development and manufacturing of biopharmaceuticals in Germany, which is a prominent life science hub in Europe.
- Adoption of Advanced Testing Technologies: The trend is shifting toward the adoption of next-generation sequencing, rapid microbiology tests, automation, and advanced molecular diagnostics in biological safety testing, which is proving to be the main reason behind the increased adoption of high-tech testing solutions in the market.
- Greater Need for Highly Sensitive Contaminant Detection: The trend is shifting toward the detection of microbial contaminants, viral adventitious agents, endotoxins, mycoplasma, and host cell proteins, which is due to the increased pressure from various regulatory bodies and biopharma industry players.
- Services Demand Is Expanding: The demand for outsourced safety testing services, particularly contract testing laboratories and service providers, is witnessing increased adoption due to the focus of biotech industry players on R&D and the outsourcing of validation and safety testing services.
Germany Biological Safety Testing Products And Services Market Segmentation
By Product & Services
Kits and Reagents- Kits and reagents serve as essential testing materials which laboratories need for their biological safety assessment work. This product category generates substantial revenue because laboratories continuously buy these items.
Instruments- The term instruments describes laboratory equipment which scientists need to perform biological safety testing activities. The equipment allows for accurate identification and examination of microbial contamination and biological impurities.
Services- The services segment provides testing and analytical services through specialized laboratories and contract research organizations (CROs) that operate in this field.
To learn more about this report, Download Free Sample Report
By Test Type
Endotoxin Tests- The tests detect bacterial endotoxins which serve as toxic substances that gram-negative bacteria release into their environment. The tests help evaluate injectable drugs and vaccines and medical devices because endotoxins trigger severe immune responses in humans.
Sterility Tests- The testing process verifies that pharmaceutical products contain no living microorganisms which include bacteria and fungi.
Cell Line Authentication & Characterization Tests- The tests authenticate cell line identity while determining its genetic composition and research quality used for biotechnology studies and drug development.
Bioburden Tests- Bioburden testing determines the count of live microorganisms that exist in a product or raw material before it undergoes sterilization.
Residual Host Contaminant Detection Tests- Biologics manufacturing uses host cells which include bacteria and mammalian cells to create proteins. The tests identify residual host cell proteins and host cell DNA that persist after the purification process.
By Application
Vaccine Development- The biological safety testing process of vaccine development establishes that vaccines must contain no microbial contamination to guarantee their safety for human medical applications.
Blood Products Testing- Plasma therapies and clotting factors which both originate from blood must complete extensive safety evaluations that test for multiple pathogens and harmful substances.
Cellular and Gene Therapy- The biological safety testing process for cell and gene therapies needs expert staff to assess the security and stability of genetically modified cells and viral vectors.
Tissue and Tissue-Related Products Testing- The safety tests of tissue products need to ensure that skin grafts and bone grafts and biomaterials do not contain any infectious agents.
Stem Cell Research- Stem cell research requires scientists to conduct multiple tests which assess cell quality and purity and genetic stability before scientists can use the cells in clinical applications.
By End Use
Pharmaceutical and Biotech Companies- The companies test biological safety during their drug discovery process and throughout their drug development work and their drug manufacturing process which makes them their largest end-user group.
Contract Research Organizations (CROs)- CROs provide pharmaceutical companies with outsourced research and testing services.
Academic and Research Institutes- Universities and research institutes perform biological safety testing in basic research and biotechnology development and the testing of new therapies.
Cell Banks and Biopharmaceutical Manufacturers- Cell banks store biological cell lines which companies use in their drug manufacturing and research activities.
Country Insights
Germany has many biopharmaceutical companies and biotechnology firms and CROs that heavily depend on biological safety testing. It has a strong biologics and vaccine development industry and cell and gene therapy research and development industry, which need strict validation for commercialization.
Germany has a highly developed research infrastructure, comprising research institutes and universities, which drive continuous innovation in biotechnology and pharmaceutical development. They are the drivers for the need for advanced technologies in the field of biological safety testing, including sterility testing, endotoxin testing, and microbial contaminant testing.
Germany has strict European regulations governing the biological industry, including safety testing during the development and manufacturing phases of drug development. This has increased the need for reliable biosafety testing solutions that can ensure the quality and safety of biologics, vaccines, and advanced therapies.
Germany is witnessing significant investments in cell therapy, gene therapy, and regenerative medicine, which is a highly specialized area that demands the highest level of biological safety testing services.
Recent Development News
Market expansion is being supported by tightening regulatory frameworks across the pharmaceutical, biotechnology, medical device, and food sectors. Biological safety testing is essential to confirm that products are free from contaminants, toxins, and potentially harmful biological substances before reaching the market.
Recent coverage of biotech sectors highlights growth in areas such as anti-aging biotech companies, showing broader industry momentum, though not specific to Germany alone, it signals increasing biotechnology activity that drives downstream safety testing needs.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 208.25 Million |
|
Market size value in 2026 |
USD 236.98 Million |
|
Revenue forecast in 2033 |
USD 593.7 Million |
|
Growth rate |
CAGR of 14.02% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Germany |
|
Key company profiled |
Thermo Fisher Scientific, Merck KGaA, Charles River Laboratories, SGS SA, Eurofins Scientific, Lonza Group, Sartorius AG, WuXi AppTec, BioReliance (Merck), Pace Analytical Services, Nelson Laboratories, BioAgilytix, Toxikon Corporation, Microbac Laboratories, Intertek Group. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Product & Service (Instruments, Reagents & Kits, Services), By Test Type (Sterility Testing, Bioburden Testing, Endotoxin Testing, Cell Line Authentication), By Application (Vaccines & Therapeutics, Medical Devices, Blood & Blood Products), By End User (Pharmaceutical & Biotechnology Companies, CROs, Academic Institutes) |
Key Germany Biological Safety Testing Products And Services Market Insights
Strict regulatory oversight from European health authorities requires extensive biological safety validation before drug commercialization. Compliance standards require sterility testing, endotoxin detection, and viral safety evaluation. Regulatory frameworks encourage high-quality testing procedures across pharmaceutical production facilities. Key Germany Biological Safety Testing Products And Services Market Insights highlight strong regulatory influence supporting market demand.
Competitive Landscape: Presence of international testing companies and specialized biotechnology laboratories creates strong market competition. Service providers focus on improving testing speed, reliability, and regulatory compliance support. Partnerships between pharmaceutical manufacturers and testing laboratories increase service demand. Key Germany Biological Safety Testing Products And Services Market Insights show competition driven by technology capability and laboratory expertise.
Future Opportunities: Expansion of personalized medicine, gene therapy research, and biologics manufacturing creates long-term growth potential. Increasing clinical research programs in Germany increase demand for biological safety validation. Investment in biotechnology infrastructure supports advanced testing services. Key Germany Biological Safety Testing Products And Services Market Insights indicate sustained market expansion supported by research growth and innovation.
Company List
- Thermo Fisher Scientific
- Merck KGaA
- Charles River Laboratories
- SGS SA, Eurofins Scientific
- Lonza Group
- Sartorius AG
- WuXi AppTec
- BioReliance (Merck)
- Pace Analytical Services
- Nelson Laboratories
- BioAgilytix
- Toxikon Corporation
- Microbac Laboratories
- Intertek Group.
Germany Biological Safety Testing Products And Services Market Report Segmentation
By Product & Services
- Kits and Reagents
- Instruments
- Services
By Test Type
- Endotoxin Tests
- Sterility Tests
- Cell Line Authentication & Characterization Tests
- Bioburden Tests
- Residual Host Contaminant Detection Tests
- Adventitious Agent Detection Tests
By Application
- Vaccine Development
- Blood Products Testing
- Cellular and Gene Therapy
- Tissue and Tissue-Related Products Testing
- Stem Cell Research
By End Use
- Pharmaceutical & Biotech
- Contract Research Organizations
- Academic & Research Institutes
- Cell Banks & Biopharmaceutical Manufacturers
Frequently Asked Questions
Find quick answers to common questions.
The approximate Germany Biological Safety Testing Products And Services Market size for the market will be USD 593.7 Million in 2033.
Key segments for the Germany Biological Safety Testing Products And Services Market: Product & Service (Instruments, Reagents & Kits, Services), Test Type (Sterility Testing, Bioburden Testing, Endotoxin Testing, Cell Line Authentication), Application (Vaccines & Therapeutics, Medical Devices, Blood & Blood Products), End User (Pharmaceutical & Biotechnology Companies, CROs, Academic Institutes.
Major players in the Germany Biological Safety Testing Products And Services Market are Thermo Fisher Scientific, Merck KGaA, Charles River Laboratories, SGS SA, Eurofins Scientific, Lonza Group, Sartorius AG, WuXi AppTec, BioReliance (Merck), Pace Analytical Services, Nelson Laboratories, BioAgilytix, Toxikon Corporation, Microbac Laboratories, Intertek Group.
The Germany Biological Safety Testing Products and Services Market size is USD 208.25 Million in 2025.
The Germany Biological Safety Testing Products and Services Market CAGR is 14.02%.
- Thermo Fisher Scientific
- Merck KGaA
- Charles River Laboratories
- SGS SA, Eurofins Scientific
- Lonza Group
- Sartorius AG
- WuXi AppTec
- BioReliance (Merck)
- Pace Analytical Services
- Nelson Laboratories
- BioAgilytix
- Toxikon Corporation
- Microbac Laboratories
- Intertek Group.
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


