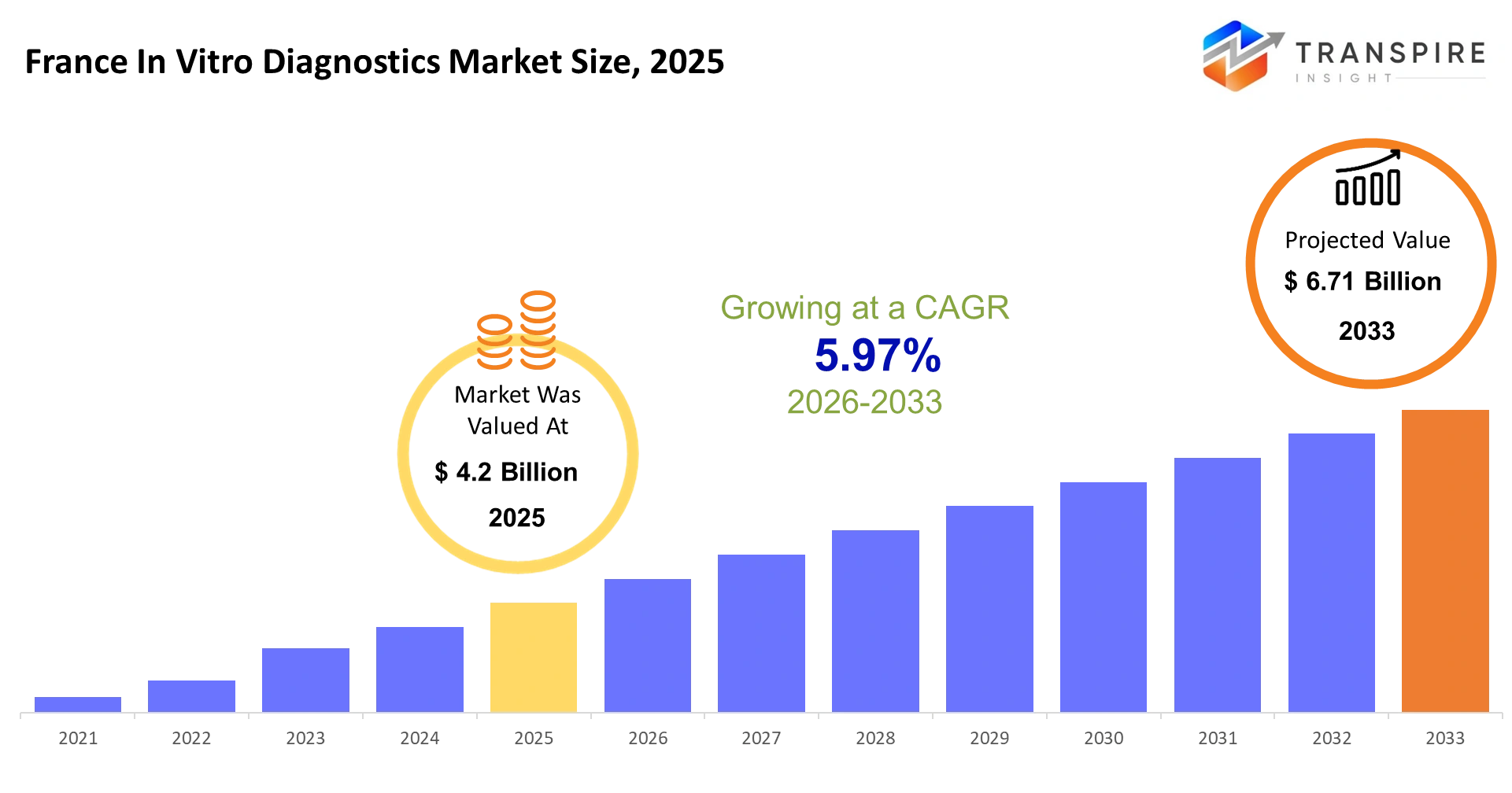
France In Vitro Diagnostics Market Size & Forecast:
- France In Vitro Diagnostics Market Size 2025: USD 4.2 Billion
- France In Vitro Diagnostics Market Size 2033: USD 6.71 Billion
- France In Vitro Diagnostics Market CAGR: 5.97%
- France In Vitro Diagnostics Market Segments: By Type (Reagents, Instruments, Software, Consumables, Others); By Application (Infectious Diseases, Oncology, Cardiology, Diabetes, Others); By End-User (Hospitals, Labs, Clinics, Others); By Technology (Immunoassay, Molecular Diagnostics, Hematology, Others)
To learn more about this report, Download Free Sample Report
France In Vitro Diagnostics Market Summary:
The France In Vitro Diagnostics Market size is estimated at USD 4.2 Billion in 2025 and is anticipated to reach USD 6.71 Billion by 2033, growing at a CAGR of 5.97% from 2026 to 2033.
The In Vitro Diagnostics Market in France will become an essential part of the healthcare sector because patient requirements are changing and testing methods are evolving to deliver results with greater speed and accuracy. People will want to receive test results from decentralized diagnostic systems which operate outside of conventional laboratory environments.
The combination of molecular testing advancements and automation technology will transform lab procedures by delivering greater accuracy and shorter testing times. The European Union will implement stricter regulations which will force manufacturers to meet new quality requirements and data standards in their operations. Digital systems will work together with data-based diagnostic methods to create new ways for doctors to understand and respond to medical test results.
What Has the Impact of Artificial Intelligence Been on the France In Vitro Diagnostics Market?
The France in vitro diagnostics market uses artificial intelligence to transform its operational and analytical processes which leads to new methods for stakeholders to examine both clinical and commercial information. AI systems in France's in vitro diagnostics market have become essential for market research because they can process extensive diagnostic and patient data in real time. The France in vitro diagnostics market uses artificial intelligence platforms to enhance data analysis which enables stakeholders to track changing disease testing patterns and laboratory operations with better accuracy.
Machine Learning and Predictive Analytics Now Enable The France In Vitro Diagnostics Market To Create New Forecasting Solutions. The tools help demand forecasting and market trends assessment because they can identify small changes in test volume and seasonal diagnostic pattern. Smart automation improves production efficiency by decreasing the need for human work during diagnostic kit manufacturing and laboratory procedures.
The France in vitro diagnostics market will create a data-driven system which functions as a responsive system through ongoing machine learning and intelligent automation development.
Key Market Trends & Insights:
- Artificial intelligence diagnostic platforms which use artificial intelligence are becoming more popular in France's in vitro diagnostics market.
- The platforms help labs achieve better disease detection results while decreasing their testing time by 20 to 30%.
- Smart laboratory systems will implement about 35% of workflow enhancements in 2025.
- Northern France controls 32% of the France in vitro diagnostics market because the region has many hospitals and modern laboratory facilities.
- Reagents and consumables lead the france in vitro diagnostics market which controls 45% of the market because diagnostic tests need these materials for their procedures.
- Molecular diagnostics has become the second largest market segment because the demand for genomic testing continues to grow.
- The France in vitro diagnostics market is primarily focused on infectious disease testing which accounts for approximately 38 of the total market share because hospitals need to monitor diseases at all times.
- The oncology diagnostics field is the fastest-growing sector because more people are choosing to undergo cancer screening tests.
- In 2025 hospitals will continue to dominate the France in vitro diagnostics market because they account for almost 50 of the market share.
- The diagnostic laboratories market segment will experience the most rapid growth as testing services are increasingly outsourced.
France In Vitro Diagnostics Market Segmentation
By Type:
The French in vitro diagnostics market will generate its main revenue from reagents and consumables which laboratories use throughout their testing procedures. Laboratories will adopt instruments at a consistent rate because they will replace their existing equipment with more precise tools. The growing importance of software will develop from advanced data management and reporting systems which enable automated testing processes and operational efficiency.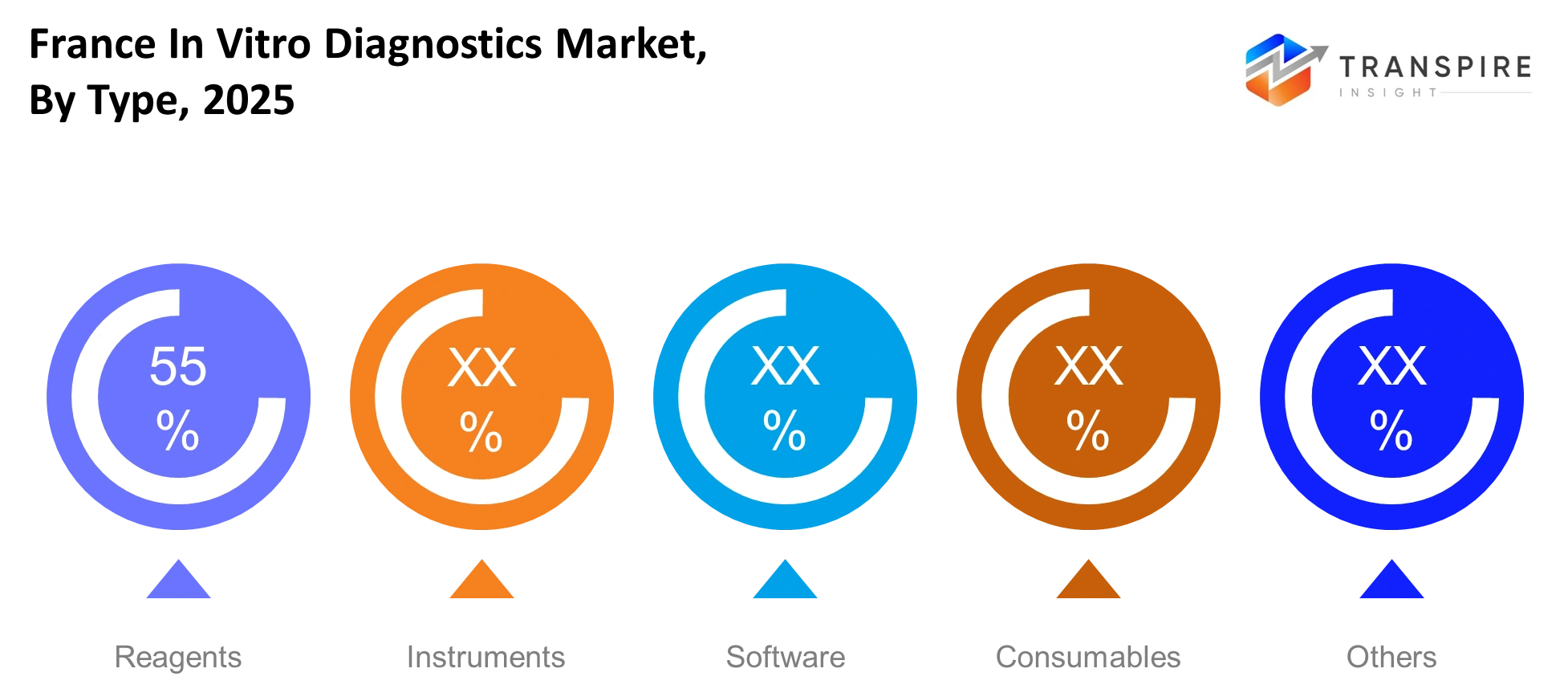
To learn more about this report, Download Free Sample Report
By Application:
The France in vitro diagnostics market will continue to use infectious disease testing because it supports ongoing patient monitoring requirements. The oncology field will experience growth because more patients want cancer screening tests which help identify cancer at an early stage. The demand for cardiology and diabetes testing will grow because more people develop lifestyle-related health issues which require doctors to conduct standard health examinations.
By End-User:
The French in vitro diagnostics market will receive most of its business from hospitals which handle large patient numbers while providing all necessary diagnostic services. The demand for testing services will lead to more diagnostic laboratories because hospitals will outsource their testing work to improve their operational efficiency and reduce expenses. The demand for quick diagnostic services will lead to gradual growth in clinics which provide easily accessible healthcare options for local communities.
By Technology:
The French in vitro diagnostics market will continue to use immunoassay technology because it provides reliable methods for detecting infections and hormonal disorders. The gene testing market together with precision medicine requirements will drive rapid growth for molecular diagnostics. Blood testing requires hematology systems as essential equipment, and new medical technologies will deliver speedier and more precise results.
What are the Main Challenges for the France In Vitro Diagnostics Market Growth?
The france in vitro diagnostics market will experience ongoing technical and operational challenges which testing systems face through their increasing complexity and data-driven nature. Manufacturers will face ongoing challenges in achieving stable product performance throughout various diagnostic situations. Laboratories which operate within the france in vitro diagnostics market will experience difficulties when they attempt to implement advanced systems because their current operations will face interruptions, and their processing times will increase.
The france in vitro diagnostics market will face manufacturing and commercialization obstacles because companies must follow demanding regulatory standards while achieving product validation needs. Organizations will spend more time and resources to achieve quality results which meet GMP requirements. The france in vitro diagnostics market will experience product launch delays because the lengthy testing and approval process will hinder new diagnostic solution market entry.
Smaller healthcare facilities which lack advanced diagnostic systems will face dual challenges from adoption issues and infrastructure deficiencies. Modern systems will face operational difficulties because the laboratory workforce remains insufficiently trained to operate them. The france in vitro diagnostics market will experience funding shortfalls which will hinder certain segments from adopting advanced technologies.
The france in vitro diagnostics market will face two main challenges from competitive market pressures and customer price sensitivity because people will start using new diagnostic technologies. The research needs of continuous innovation will compel companies to dedicate their resources toward developing new technologies while tightening regulatory changes and rapid technological shifts will increase long-term market uncertainty.
Regional Insights
The france in vitro diagnostics market will develop at different rates in various regions because of distinct healthcare systems and their ability to perform diagnostic tests. The advanced laboratories in urban areas together with greater funding resources will drive their adoption of new technologies. Medical centers in France will demonstrate better market performance through their implementation of automated testing systems whereas small medical facilities will depend on basic diagnostic equipment because of their inability to adopt new technologies.
The France in vitro diagnostics market will benefit from northern areas which possess both extensive hospital systems and research institutions. Southern regions will gradually improve as healthcare investments increase and access to modern diagnostic services expands. The France in vitro diagnostics market will see expanded testing services through partnerships between public healthcare institutions and private laboratory facilities, which will advance testing capabilities across multiple areas and improve diagnostic accuracy.
The rural areas of France will experience slower progress in their in vitro diagnostic services because they lack proper facilities and trained personnel. The situation will improve because mobile diagnostic units and remote testing solutions will provide essential support for gradual advancements. The digital health expansion will enable the France in vitro diagnostics market to achieve better operational results, which will make it easier for underserved regions to access timely and precise diagnostic services.
Recent Development News
In April 2026, bioMérieux announced the acquisition of a majority stake in a European molecular diagnostics startup to strengthen its infectious disease testing portfolio and expand its presence in France’s hospital laboratory segment.https://www.biomerieux.com
In March 2026, Roche announced the acquisition of a French digital pathology and AI diagnostics firm to enhance its in vitro diagnostics capabilities and accelerate personalized medicine solutions across France and Europe.https://www.roche.com
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 4.2 Billion |
|
Market size value in 2026 |
USD 4.47 Billion |
|
Revenue forecast in 2033 |
USD 6.71 Billion |
|
Growth rate |
CAGR of 5.97% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
France |
|
Key company profiled |
Roche, Abbott, Siemens Healthineers, Thermo Fisher, Danaher, Bio-Rad, Sysmex, Qiagen, Hologic, Ortho Clinical, Agilent, PerkinElmer, Mindray, BD, Beckman Coulter |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Reagents, Instruments, Software, Consumables, Others); By Application (Infectious Diseases, Oncology, Cardiology, Diabetes, Others); By End-User (Hospitals, Labs, Clinics, Others); By Technology (Immunoassay, Molecular Diagnostics, Hematology, Others) |
How Can New Companies Establish a Strong Foothold in the France In Vitro Diagnostics Market?
New companies entering the france in vitro diagnostics market must establish precise operational methods to achieve market presence between their competitive points. The france in vitro diagnostics market will reward players that solve real clinical challenges such as quicker disease identification and more efficient laboratory processes. Startups such as digital pathology innovators and AI-based diagnostic platforms like Owkin will demonstrate that machine learning combined with clinical data will enhance both diagnostic speed and accuracy in oncology and rare disease identification.
Strategic partnerships will play a key role in strengthening position within the france in vitro diagnostics market, especially collaborations with hospitals, research institutes, and established diagnostic firms. The france in vitro diagnostics market will also favor companies that integrate smart automation and cloud-based reporting systems to reduce operational delays in laboratories. Emerging players like molecular testing innovators will gain traction by aligning with healthcare providers that need scalable and cost-efficient diagnostic solutions.
The france in vitro diagnostics market requires technology differentiation as companies face competition from international industry leaders. Companies that develop AI-powered analytics and portable diagnostic tools and rapid testing kits will create stronger market presence. The france in vitro diagnostics market will keep evolving toward personalized data-based diagnostic methods which will create opportunities for agile startups that develop solutions for current clinical problems and efficient decision-making systems.
Key France In Vitro Diagnostics Market Company Insights
The France in vitro diagnostics market will experience strong competition because established global diagnostic firms operate in the market while new technology-driven startups enter. The market will experience competitive differentiation through testing innovation development and testing method execution and testing process automation. The France in vitro diagnostics market will demonstrate increasing testing requirements because companies need to lower testing expenses while they work to enhance precision and provide advanced diagnostic services to healthcare systems.
The France in vitro diagnostics market will grow through product innovation and digital solutions development because Roche Siemens Healthineers and Abbott Laboratories lead this market. The France in vitro diagnostics market will experience increasing impact from biotechnology companies such as bioMérieux which will drive progress in infectious disease and molecular testing solution development throughout Europe.
The France in vitro diagnostics market will develop through strategic partnerships and company acquisitions and AI-based diagnostic systems which will create new competitive dynamics. Companies will develop their laboratory automation systems and point-of-care testing systems to satisfy increasing consumer needs for rapid testing results. The France in vitro diagnostics market will experience heightened competition because new companies introduce specialized diagnostic products which compel established businesses to allocate additional resources for research and intelligent healthcare solutions.
Company List
- Roche
- Abbott
- Siemens Healthineers
- Thermo Fisher
- Danaher
- Bio-Rad
- Sysmex
- Qiagen
- Hologic
- Ortho Clinical
- Agilent
- PerkinElmer
- Mindray
- BD
- Beckman Coulter
What are the Key Use-Cases Driving the Growth of the France In Vitro Diagnostics Market?
The France in vitro diagnostics market will experience steady growth because clinical testing has become the primary method for detecting diseases in their early stages and for providing preventive healthcare. The medical field will obtain faster clinical results through the increasing adoption of blood tests and molecular tests and rapid diagnostic testing equipment. The France in vitro diagnostics market will experience growth due to the rising need for precise results which find application in infectious disease monitoring and chronic illness management because early diagnosis determines treatment success.
Oncology testing will serve as the main application area for the France in vitro diagnostics market because early cancer detection through biomarker analysis and genetic screening will improve survival rates. The France in vitro diagnostics market will experience increased usage in diabetes and cardiovascular monitoring because regular testing has become vital for ongoing healthcare of patients. Healthcare providers will expand these applications because they now prioritize preventive care as their main treatment approach.
Infectious disease surveillance will create another essential driver for the France in vitro diagnostics market because laboratories need high-throughput testing systems to handle outbreak situations and seasonal infection periods. The France in vitro diagnostics market will grow through point-of-care testing which clinics and emergency settings use to provide rapid diagnosis without requiring central laboratory systems. This transformation will enhance health system accessibility while it helps emergency teams reach crucial situations more efficiently.
France In Vitro Diagnostics Market Report Segmentation
By Type
- Reagents
- Instruments
- Software
- Consumables
- Others
By Application
- Infectious Diseases
- Oncology
- Cardiology
- Diabetes
- Others
By End-User
- Hospitals
- Labs
- Clinics
- Others
By Technology
- Immunoassay
- Molecular Diagnostics
- Hematology
- Others
Frequently Asked Questions
Find quick answers to common questions.
The Approximate France In Vitro Diagnostics Market size for the Market will be USD 6.71 Billion in 2033.
Key Segments for the France In Vitro Diagnostics Market are By Type (Reagents, Instruments, Software, Consumables, Others); By Application (Infectious Diseases, Oncology, Cardiology, Diabetes, Others); By End-User (Hospitals, Labs, Clinics, Others); By Technology (Immunoassay, Molecular Diagnostics, Hematology, Others).
Major France In Vitro Diagnostics Market Players are Roche, Abbott, Siemens Healthineers, Thermo Fisher, Danaher, Bio-Rad, Sysmex, Qiagen, Hologic, Ortho Clinical, Agilent, PerkinElmer, Mindray, BD, Beckman Coulter.
The France In Vitro Diagnostics Market size is USD 4.2 Billion in 2025.
The France In Vitro Diagnostics Market CAGR is 5.97%.
- Roche
- Abbott
- Siemens Healthineers
- Thermo Fisher
- Danaher
- Bio-Rad
- Sysmex
- Qiagen
- Hologic
- Ortho Clinical
- Agilent
- PerkinElmer
- Mindray
- BD
- Beckman Coulter
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


