Europe Pharmaceutical Manufacturing Market Size & Forecast:
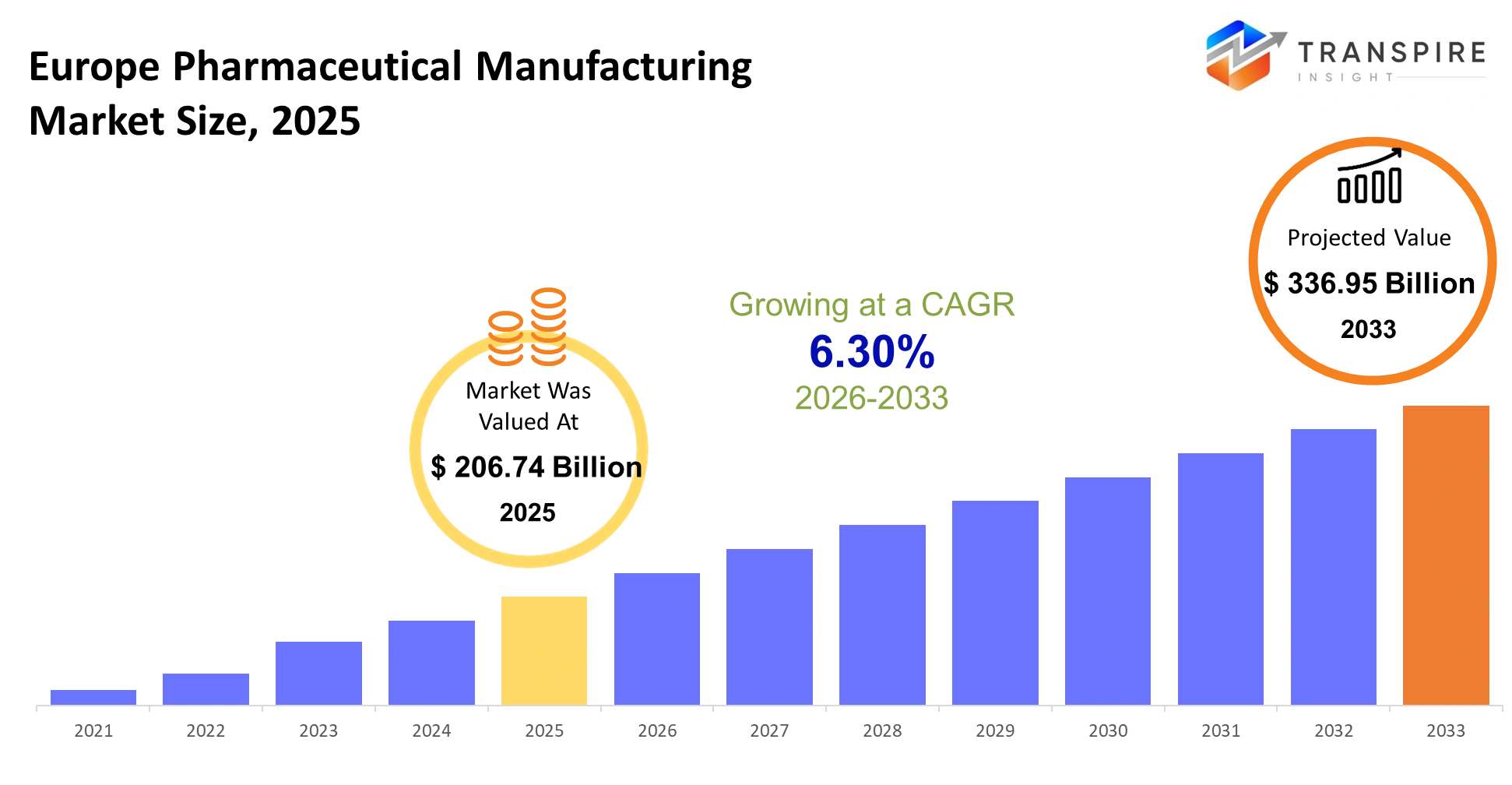
- Europe Pharmaceutical Manufacturing Market Size 2025: USD 206.74 Billion
- Europe Pharmaceutical Manufacturing Market Size 2033: USD 336.95 Billion
- Europe Pharmaceutical Manufacturing Market CAGR: 6.30%
- Europe Pharmaceutical Manufacturing Market Segments: By Type (API Manufacturing, Finished Dosage, Biologics Manufacturing, Others); By Application (Oncology, Cardiology, Neurology, Infectious Diseases, Others); By End-User (Pharma Companies, Biotech Firms, Others); By Process (Batch, Continuous, Others)
To learn more about this report, Download Free Sample Report
Europe Pharmaceutical Manufacturing Market Summary
The Europe Pharmaceutical Manufacturing Market was valued at USD 206.74 Billion 2025. It is forecast to reach USD 336.95 Billion by 2033. That is a CAGR of 6.30% over the period.
The pharmaceutical manufacturing market in Europe serves as the primary support for the healthcare system because it converts drug research into production capacity which meets the needs of hospitals, pharmacies, and export supply chains. Manufacturers are expanding their operations to include biologics and cell therapies and high-value injectables which need specialized facilities and stringent quality management systems. The market underwent a complete shift because European governments acknowledged the dangers of relying on foreign active pharmaceutical ingredient suppliers which resulted in the establishment of local manufacturing operations. The COVID-19 pandemic exposed supply bottlenecks which resulted in delayed raw material shipments and production interruptions, which led both regulators and drug companies to build new manufacturing facilities and adopt digital production technologies. The current trend leads to increased capital investment in contract manufacturing, automation systems, and sterile production facilities. Pharmaceutical companies are establishing long-term contracts with manufacturing partners who handle their complex production needs in order to shorten product development times and control their compliance expenses.
Key Market Insights
- Germany France and Switzerland together occupied more than 58% of the European pharmaceutical manufacturing market during 2025 which Western Europe controlled.
- Germany leads regional pharmaceutical production due to strong API manufacturing infrastructure, export capacity, and government-backed life sciences investments.
- The Central and Eastern European market represents the fastest-growing regional market, which will continue until 2030 because both operating costs and CDMO facility capacity are increasing.
- Pharmaceutical manufacturing in Ireland keeps increasing because multinational companies expand biologics production while the government offers tax incentives.
- The branded pharmaceutical manufacturing sector controlled 46% of the market share because European drug development for patented drugs showed strong commercial success.
- The generic drug manufacturing sector stands as the second-largest industry segment, which receives support from both healthcare cost-control measures and rising prescription rates.
- The biologics manufacturing sector shows the fastest growth rate until 2030 as monoclonal antibodies and biosimilars enter the commercial market.
- Pharmaceutical companies experienced major growth in contract manufacturing services because they needed to outsource production work for two main reasons.
- The production capacity for sterile injectable manufacturing grew quickly between 2022 and 2025 because both oncology and vaccine production needs increased.
- The European Pharmaceutical Manufacturing Market recognized oncology drug manufacturing as its leading segment which generated about 28% of total revenue during 2025.
What are the Key Drivers, Restraints, and Opportunities in the Europe Pharmaceutical Manufacturing Market?
The primary reason for market expansion in Europe Pharmaceutical Manufacturing Market exists because European manufacturers now prioritize local production after COVID-19 revealed their dependence on foreign active pharmaceutical ingredients and essential drug components. The governments in Germany France and Italy initiated funding programs which improved regulatory processes and provided incentives to expand domestic pharmaceutical manufacturing operations. Pharmaceutical companies increased their funding for biologics production facilities and sterile injectable manufacturing plants and automated production technologies to maintain their production operations. The transition resulted in higher capital expenditures for contract manufacturing and high-value drug production which generated permanent revenue streams for manufacturers who possess advanced compliance abilities and large-scale production capacities.
The market expansion process encounters its most substantial obstacle through the existing complex regulatory framework which governs all operations. Pharmaceutical manufacturers operating across Europe need to adhere to strict Good Manufacturing Practice standards together with country-specific pricing regulations and environmental protection rules and comprehensive validation protocols used in biologics production. The requirements result in longer facility construction periods and increased operational expenses which particularly impact smaller manufacturers and biotech companies. The incomplete regulatory harmonization process between European markets forces companies to either extend their expansion plans or delay their product launches which leads to decreased manufacturing capacity and extended revenue collection times.
The next growth phase will find its most significant potential through continuous manufacturing and AI-driven production analytics. Pharmaceutical companies develop smart factories that use predictive maintenance and digital twins and real-time quality monitoring to reduce batch failures and shorten production cycles. Ireland and Switzerland have become key centers for advanced manufacturing investments which focus on biologics and personalized medicine production.
What Has the Impact of Artificial Intelligence Been on the Europe Pharmaceutical Manufacturing Market?
European pharmaceutical manufacturing systems use artificial intelligence and digital technologies to achieve precise production results while reducing operational downtime and delivering fast solutions for regulatory compliance.The AI-powered manufacturing execution systems which drug manufacturers select for their facilities execute automated monitoring processes together with sterile production controls and quality assurance workflows which maintain biopharmaceuticals and injectable medications. The systems function to track sensor data from active operations which detects contamination threats and process deviations and equipment failures that could result in production interruptions.
The pharmaceutical industry uses machine learning models to improve their predictive maintenance operations. AI algorithms enable manufacturers to predict equipment failures by analyzing vibration patterns and temperature shifts and examining historical maintenance records of their reactors and filling lines and cold-chain storage systems. Businesses use predictive methods to decrease unexpected equipment shutdowns while increasing machine durability and maintaining operations for their essential therapeutic products.The European facilities utilize digital process analytics together with automated quality inspection systems to achieve operational efficiency improvements which result in double-digit reductions of their batch rejection rates.
Advanced analytics platforms enable regulatory compliance through their ability to automate documentation processes and production traceability systems while they track audit deviations. The primary barrier to integration remains because high integration expenses create a significant challenge. The fragmented infrastructure of legacy pharmaceutical plants in Europe makes AI implementation difficult and costly for small and mid-sized manufacturers.
Key Market Trends
- European pharmaceutical companies since 2021 started investing more in local API production after they realized their pandemic supply chain problems came from their heavy reliance on Asian imports.
- The period from 2022 to 2025 saw a major increase in biologics manufacturing capacity which resulted from the rising production of monoclonal antibodies and biosimilars in Germany and Switzerland.
- Contract manufacturing partnerships experienced rapid growth after mid-sized biotech companies chose to outsource their sterile injectable production work in order to save money on building new facilities and avoid delays from regulatory compliance.
- Pharmaceutical manufacturers implemented AI-based quality monitoring systems which helped them decrease product rejection rates while maintaining better inspection preparedness throughout their regulated manufacturing facilities.
- Ireland emerged as a prime location for major pharmaceutical companies to establish new operations after international businesses moved their valuable biologics production facilities to locations near European markets.
- The year 2023 marked the beginning of growing interest in continuous manufacturing technologies because regulators approved quicker batch testing methods and better manufacturing consistency.
- Companies including Roche and Novartis expanded their capacity to produce cell and gene therapies which enabled them to help customers develop personalized medicine products.
- Pharmaceutical plants that use large amounts of energy started automating their operations after European energy costs became unstable between 2022 and 2024.
- Western European pharmaceutical production facilities needed to upgrade their solvent recovery systems because environmental regulations required them to decrease their emission levels.
- Eastern European countries developed their manufacturing capability because international drug companies established production sites in these regions to take advantage of lower expenses while maintaining their drug development operations across multiple locations.
Europe Pharmaceutical Manufacturing Market Segmentation
By Type:
The market for finished dosage manufacturing currently leads the market because pharmaceutical companies expand their production of tablets and capsules and injectable products and sterile drugs throughout European markets. Advanced dosage facilities achieve high operational efficiency due to strong hospital requirements and export operations and tight government regulations which protect the public. The active pharmaceutical ingredient production process remains important because regions work to decrease their need for imported drug components which were disrupted by the global health emergency. The production of biologics has become the most rapidly expanding industry because monoclonal antibodies and biosimilars and cell therapies reach more advanced stages of commercial production.
The growth of biologics production results from increasing funding for high-value therapeutics which need special sterile facilities and sophisticated bioprocessing technology. Current manufacturers establish adaptable production facilities throughout Germany and Ireland and Switzerland to meet the needs of small-batch production and precision medicine development. The ongoing growth of biologics production facilities will lead to changes in how investors distribute their capital because they will focus on facilities that can manufacture intricate therapies while gaining quicker regulatory approvals. The manufacturing industry for finished dosage products will increase its automation budget to create more reliable production methods and decrease expenses related to regulatory compliance.
To learn more about this report, Download Free Sample Report
By Application:
The pharmaceutical industry depends on oncology manufacturing because cancer treatments which generate high market value need ongoing development for biologic, injectable, and precision medicine products. Major pharmaceutical corporations continue to build their manufacturing facilities for oncology drug development because they receive substantial reimbursement benefits and European countries increase their clinical trial operations. The production of cardiology and infectious disease treatments maintains constant demand because of the needs created by an aging population who require extended medical care. The production of neurology treatments has grown steadily because researchers develop Alzheimer’s disease and neurodegenerative disorder therapies into commercial-scale manufacturing processes.
The oncology manufacturing sector will maintain its market leadership position until the forecast period ends because personalized medicine and antibody-drug conjugates will become more widely used. The production capacity for infectious diseases continues to grow because vaccine manufacturing companies expanded their operations during the period from 2020 to 2023. Pharmaceutical developers are increasing spending on flexible manufacturing systems which enable them to produce multiple therapeutic categories from a single production facility. The European healthcare market will see more investment in neurology-focused manufacturers because of the growing number of approved advanced neurological therapies.
By End-User:
Pharmaceutical companies maintain the highest manufacturing output because multinational drug manufacturers operate their production facilities with vertical integration to handle multiple business operations while their existing regulatory systems permit their business activities. The large manufacturing companies maintain their expansion efforts through building new production sites which enables them to achieve better control over their supply network and safeguard their valuable therapeutic innovations. The fastest-growing segment for end-user markets belongs to biotech companies because they secure funding for the development of biologic treatments and gene therapy solutions and precision medicine research. Biotech companies with smaller operations now prefer to establish manufacturing contracts with external partners because this strategy helps them bring their products to market faster while it decreases their need for financial investment.
The need for biotechnology production drives contract manufacturing companies in Europe to adjust their operational methods. Manufacturing providers are investing in modular facilities which support single-use technologies and specialized biologics capabilities to enable their clients to develop new biotechnology products. Pharmaceutical companies are also increasing collaboration agreements with biotech developers to access innovative therapies and diversify product portfolios. The upcoming period will require companies to find appropriate manufacturing solutions which need specialized knowledge in handling complex manufacturing processes and understanding particular regulatory requirements.
By Process:
The current method used for pharmaceutical production is Batch manufacturing because European commercial drug facilities depend on traditional validation systems and regulatory procedures which follow batch processing methods. The manufacturing process of traditional batch processing allows pharmaceutical companies to produce large quantities of tablets capsules and sterile injectables which meet high-volume demand across various therapeutic fields. The share of continuous manufacturing remains small yet the demand for its implementation grows because pharmaceutical companies want to achieve faster production times while decreasing material waste and enhancing their operational efficiency. The regulatory bodies now support continuous manufacturing implementation by providing new guidelines and faster approval processes.
The development of continuous manufacturing depends on two main factors which are operational efficiency and digital process integration. Advanced manufacturing facilities which use real-time monitoring technology and artificial intelligence quality control systems can achieve two main benefits which include reduced downtime and faster product release processes. European pharmaceutical manufacturers will increase their investment in continuous production systems because energy expenses and compliance requirements continue to rise in Europe. The trend of automated process control system adoption and smart manufacturing platform usage will create new opportunities for equipment suppliers and technology developers.
What are the Key Use Cases Driving the Europe Pharmaceutical Manufacturing Market?
The main purpose of pharmaceutical manufacturing activities throughout Europe depends on oncology drug production as its primary use case. The demand for cancer therapies requires high-value biologics and sterile injectables together with precise dosage manufacturing which needs advanced production facilities and strict quality control systems to operate continuously.
Biotech companies and contract manufacturing organizations are experiencing rapid growth in their vaccine manufacturing and biosimilar production operations. European healthcare systems continue increasing procurement of biologics for infectious diseases, autoimmune disorders, and chronic care applications, pushing manufacturers to scale flexible production capacity and cold-chain infrastructure.
Cell and gene therapy manufacturing represents an emerging use case with strong long-term potential. The pharmaceutical industry of Germany Switzerland and Ireland is building specialized facilities to produce small-batch personalized medicine for targeted therapies and rare disease treatments.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 206.74 Billion |
|
Market size value in 2026 |
USD 219.77 Billion |
|
Revenue forecast in 2033 |
USD 336.95 Billion |
|
Growth rate |
CAGR of 6.30% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain, Rest of Europe) |
|
Key company profiled |
Pfizer, Novartis, Roche, Sanofi, GSK, AstraZeneca, Merck, Johnson & Johnson, Bayer, AbbVie, Eli Lilly, Teva, Boehringer Ingelheim, Takeda, Lonza |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (API Manufacturing, Finished Dosage, Biologics Manufacturing, Others); By Application (Oncology, Cardiology, Neurology, Infectious Diseases, Others); By End-User (Pharma Companies, Biotech Firms, Others); By Process (Batch, Continuous, Others) |
Which Regions are Driving the Europe Pharmaceutical Manufacturing Market Growth?
The pharmaceutical manufacturing sector exists in Western Europe because Germany, Switzerland, France, and Ireland operate effective regulatory systems and developed biologics infrastructure and extensive pharmaceutical research facilities. The region possesses established export routes together with fully automated manufacturing plants and strong partnerships among pharmaceutical developers and academic institutions and medical organizations. European Medicines Agency compliance standards require enterprises to create top-grade manufacturing facilities and digital quality assurance systems thus fostering ongoing investment activities. Western European countries provide stable political conditions and offer highly skilled employees which lead multinational pharmaceutical companies to build their sterile injectable and biologics production facilities in the region.
Northern Europe serves as the second most important regional contributor because its health care system receives government funding and its industrial policies remain stable and its biotechnology systems reach advanced development. The countries of Denmark, Sweden, and Finland dedicate their resources to developing precision medicine and biosimilars and sustainable pharmaceutical production technologies instead of establishing large-scale generic manufacturing operations. Northern European manufacturers prioritize specialized therapeutics while Western European manufacturers build their companies around standard production processes. Public-private partnerships and reimbursement systems that work reliably together enable the region to secure ongoing manufacturing investments while generating consistent income.
The period from 2022 to 2025 demonstrates that pharmaceutical manufacturers have achieved their most rapid growth in Central and Eastern Europe during this time. The combination of decreased operational expenses and improved infrastructure development initiatives has enabled Poland, Hungary, and the Czech Republic to attract new investment for their manufacturing businesses. Multiple international pharmaceutical companies moved some of their production operations to these countries as a strategy to create new supply routes while decreasing their reliance on expensive Western European manufacturing plants. The contract manufacturing industry and equipment manufacturing sector and biotechnology investment market will experience substantial growth opportunities until the year 2033.
Who are the Key Players in the Europe Pharmaceutical Manufacturing Market and How Do They Compete?
The Europe Pharmaceutical Manufacturing Market operates at a moderate level of consolidation because multinational pharmaceutical companies control the majority of high-value biologics and sterile injectables and patented drug production while smaller contract manufacturers serve specialized outsourcing markets. Companies now compete in three main areas which include their manufacturing technology ability to meet regulatory requirements and their ability to maintain operational efficiency during supply chain disruptions. Established manufacturers maintain their market presence through investments in automation technology and construction of new manufacturing facilities and establishment of advanced biologics production centers. CDMOs and biotechnology manufacturers implement new production strategies that enable them to deliver products more quickly while providing flexible options for small-scale manufacturing.
Roche operates its business as an oncology biologics and personalized medicine manufacturer, which establishes its unique market position against competitors who focus on producing high volumes of generic products. The advanced biologics facilities located in Switzerland and Germany operate precision therapy manufacturing processes that maintain their quality control measures through the use of digital production analytics. Novartis expands its cell and gene therapy manufacturing operations by making strategic research partnerships and targeted investments throughout Europe. Novartis possesses extensive proficiency in complex therapies, which enables the company to manufacture high-margin specialty drugs that have substantial regulatory restrictions.
Lonza competes through its dedicated contract manufacturing services that specialize in biologics, mRNA therapies, and small-scale commercial manufacturing. Biotechnology companies prefer to use flexible manufacturing systems which enable them to scale production quickly through commercializing their products without establishing their own manufacturing equipment.Sanofi establishes operational distinction through its vaccine production capabilities and its complete European distribution network operations which enable it to deliver pharmaceutical products at high volume. Bayer maintains its commitment to digital manufacturing system development while investing in environmentally friendly production technology solutions which will enhance its operational performance and decrease its European facility compliance expenses.
Company List
- Pfizer
- Novartis
- Roche
- Sanofi
- GSK
- AstraZeneca
- Merck
- Johnson & Johnson
- Bayer
- AbbVie
- Eli Lilly
- Teva
- Boehringer Ingelheim
- Takeda
- Lonza
Recent Development News
“In March 2026, Roche launched an NVIDIA-powered AI factory to accelerate therapeutics and diagnostics development across operations in Europe and the United States. The initiative expanded AI-enabled pharmaceutical manufacturing and data-driven drug development capabilities using more than 3,500 GPUs.https://www.nasdaq.com
In November 2025, Sanofi announced the inauguration of a new biomanufacturing unit at its Le Trait site in France. The expansion strengthens biologics fill-finish capacity for monoclonal antibody production and supports distribution across 44 countries. The investment improves sterile injectable throughput and reinforces localized European supply chain resilience.https://www.sanofi.fr
What Strategic Insights Define the Future of the Europe Pharmaceutical Manufacturing Market?
The Europe Pharmaceutical Manufacturing Market is moving toward a structurally more localized, technology-intensive production model which operates between two opposing forces that seek to decrease supply chain dependence and speed up biologics development. Manufacturing capacity has started to focus on digitally advanced facilities which provide organizations with the ability to produce different batch sizes while achieving faster regulatory approval processes and maintaining quality assurance throughout their operations. The upcoming 5 to 7 years will shift competitive advantage from scale to the ability of organizations to combine their compliance requirements with their continuous manufacturing operations and their advanced biologics development capabilities.
European states create a less noticeable risk because their different regulatory systems which govern GMP updates and pricing controls lead to production strategy fragmentation and increased costs for companies that need to meet cross-border regulations. The situation will delay optimization of multi-site manufacturing which will result in decreased efficiency benefits from consolidated operations across various regions.
Decentralized manufacturing hubs which support cell and gene therapy production through their presence in Ireland and Central Europe create a major opportunity because infrastructure investments will support personalized medicine development. Market participants should follow modular facility design and AI-enabled process control systems as their primary focus because these systems enable them to obtain high-margin biologics contracts while they operate under multiple regulatory systems.
Europe Pharmaceutical Manufacturing Market Report Segmentation
By Type
- API Manufacturing
- Finished Dosage
- Biologics Manufacturing
- Others
By Application
- Oncology
- Cardiology
- Neurology
- Infectious Diseases
- Others
By End-User
- Pharma Companies
- Biotech Firms
- Others
By Process
- Batch
- Continuous
- Others
Frequently Asked Questions
Find quick answers to common questions.
The Europe Pharmaceutical Manufacturing Market size is USD 336.95 Billion in 2033.
Key segments for the Europe Pharmaceutical Manufacturing Market are By Type (API Manufacturing, Finished Dosage, Biologics Manufacturing, Others); By Application (Oncology, Cardiology, Neurology, Infectious Diseases, Others); By End-User (Pharma Companies, Biotech Firms, Others); By Process (Batch, Continuous, Others).
Major Europe Pharmaceutical Manufacturing Market players are Pfizer, Novartis, Roche, Sanofi, GSK, AstraZeneca, Merck, Johnson & Johnson, Bayer, AbbVie, Eli Lilly, Teva, Boehringer Ingelheim, Takeda, Lonza
The Europe Pharmaceutical Manufacturing Market size is USD 206.74 Billion in 2025.
The Europe Pharmaceutical Manufacturing Market CAGR is 6.30% from 2026 to 2033.
- Pfizer
- Novartis
- Roche
- Sanofi
- GSK
- AstraZeneca
- Merck
- Johnson & Johnson
- Bayer
- AbbVie
- Eli Lilly
- Teva
- Boehringer Ingelheim
- Takeda
- Lonza
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033