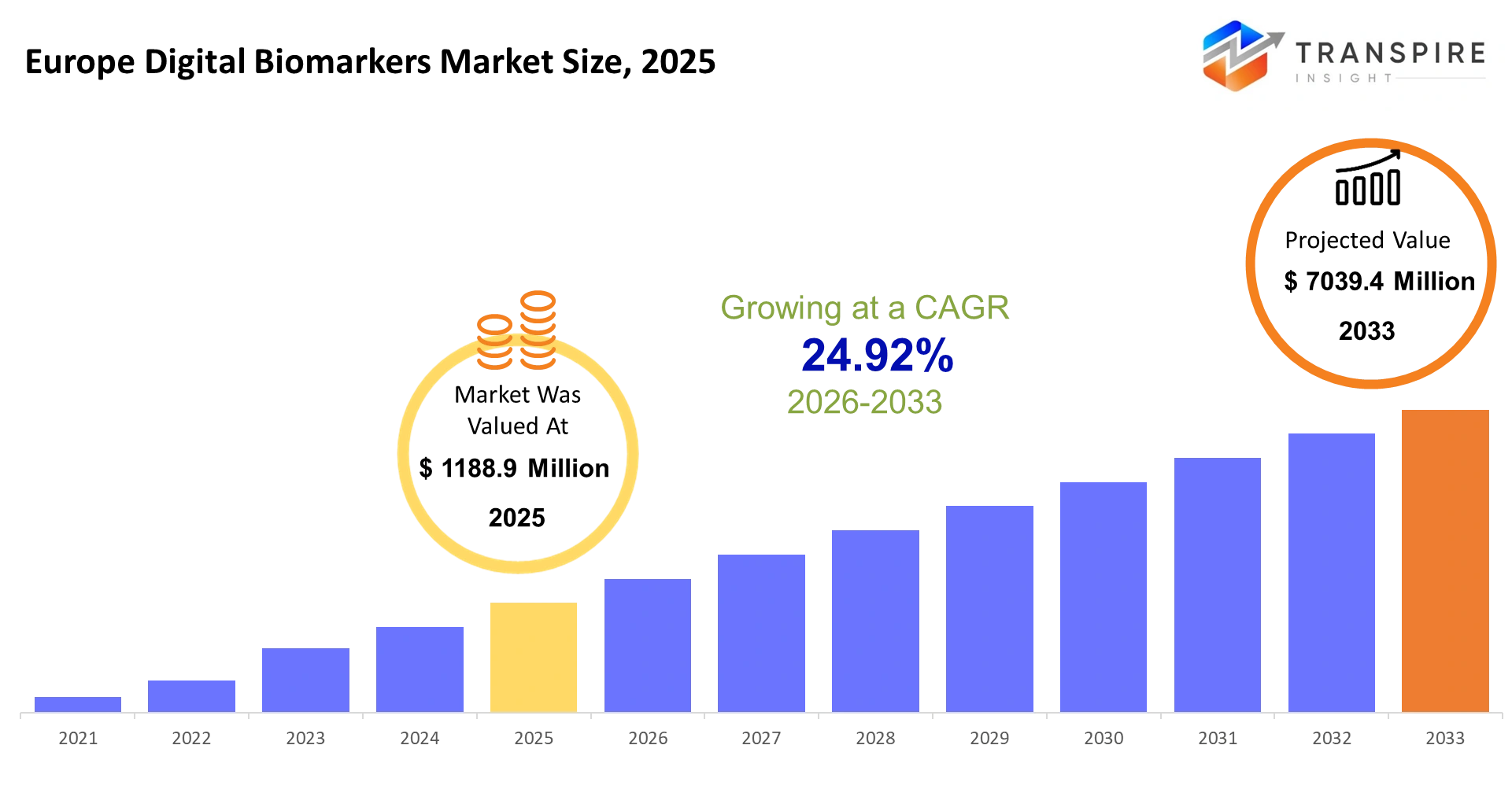
Europe Digital Biomarkers Market Size & Forecast:
- Europe Digital Biomarkers Market Size 2025: USD 1188.9 Million
- Europe Digital Biomarkers Market Size 2033: USD 7039.4 Million
- Europe Digital Biomarkers Market CAGR: 24.92%
- Europe Digital Biomarkers Market Segments: By Type (Wearable Biomarkers, Mobile-based Biomarkers, Sensor-based Biomarkers, Software-based Biomarkers, AI-based Biomarkers, Others); By Application (Clinical Trials, Diagnostics, Remote Monitoring, Drug Development, Disease Management, Others); By End-User (Pharma, Healthcare Providers, Research Institutes, Biotech, Hospitals, Others); By Technology (AI, IoT, Mobile Apps, Cloud Platforms, Data Analytics, Others)
To learn more about this report, Download Free Sample Report
Europe Digital Biomarkers Market Summary:
The Europe Digital Biomarkers Market size is estimated at USD 1188.9 Million in 2025 and is anticipated to reach USD 7039.4 Million by 2033, growing at a CAGR of 24.92% from 2026 to 2033. The european digital biomarkers market within the healthcare technology industry will revolve around data collected from smartphone devices, wearables, and smart sensor systems to provide real-time analysis of the human body. The market will evolve from traditional clinical environments, where patients will require constant monitoring without intrusive procedures compatible with their lifestyles. There will be a need for personalized information rather than mere measurements, compelling firms to enhance their algorithmic approaches and context-based analytical techniques. European regulatory authorities will impose stringent validation guidelines, balancing accuracy and innovation. With improvements in artificial intelligence and edge computing, this market will closely align with preventive medicine and telemedicine goals.
What Has the Impact of Artificial Intelligence Been on the Europe Digital Biomarkers Market?
Artificial intelligence has rapidly reshaped the europe digital biomarkers market by transforming how health data is captured, interpreted, and applied in real-world settings. AI in europe digital biomarkers market environments has strengthened market research capabilities by processing vast volumes of patient-generated data from wearables and mobile platforms. Advanced machine learning models now identify subtle behavioral and physiological patterns, enabling more precise data analysis and supporting evidence-based decision-making for healthcare providers and industry stakeholders.
Artificial intelligence in europe digital biomarkers market operations has also improved demand forecasting and trend prediction. Predictive analytics tools will anticipate patient needs, disease progression patterns, and therapy responses with greater accuracy. This shift will allow companies to align product development with emerging market trends while enhancing personalization. Smart automation further streamlines data collection and validation processes, reducing manual intervention and minimizing errors, which directly improves production efficiency and accelerates innovation cycles.
Beyond analytics, AI will optimize supply chains and operational costs across the europe digital biomarkers market by enabling real-time monitoring and intelligent resource allocation. Companies leveraging AI-driven systems will gain a competitive advantage through faster deployment, scalable solutions, and tailored patient insights. As digital healthcare adoption expands, AI will continue to drive innovation, making the europe digital biomarkers market more responsive, efficient, and aligned with personalized medicine expectations.
Key Market Trends & Insights:
- Western Europe dominates the europe digital biomarkers market with over 45% market share in 2025, driven by advanced healthcare infrastructure and strong regulatory frameworks.
- Eastern Europe is the fastest-growing region, expected to expand at a CAGR above 20% during 2025–2030 due to rising digital health investments.
- Wearable-based biomarkers dominate with nearly 50% share in 2025, supported by growing adoption of smartwatches and fitness trackers.
- Mobile-based applications hold the second-largest share, driven by increasing smartphone penetration and health app usage.
- Sensor-integrated platforms are the fastest-growing segment, projected to grow rapidly through 2030 due to continuous monitoring capabilities.
- Chronic disease management dominates with over 40% share, as demand for remote monitoring solutions continues to rise across Europe.
- Mental health monitoring is the fastest-growing application, fueled by increasing awareness and AI-based behavioral tracking tools.
Europe Digital Biomarkers Market Segmentation
Europe Digital Biomarkers Market:
Europe digital biomarkers market will reflect a shift toward continuous health tracking supported by connected devices and advanced analytics. Healthcare systems will integrate digital signals into routine care, allowing real-time insights instead of episodic clinical observations. Demand will grow for solutions that deliver accuracy, ease of use, and meaningful interpretation of patient data. Regulatory updates across European countries will shape validation standards and ensure clinical-grade reliability.
Technology progress will push this market toward predictive and preventive care models. Data from wearables, mobile platforms, and sensors will support early detection and better disease understanding. Industry players will invest in scalable platforms that align with digital health ecosystems. Market dynamics will remain influenced by consumer expectations for convenience, privacy protection, and personalized health insights.
By Type
Wearable biomarkers will lead adoption as smart devices capture continuous physiological signals such as heart rate and activity levels. Mobile-based biomarkers will expand through smartphone integration, offering accessible and cost-effective monitoring solutions. Sensor-based biomarkers will strengthen clinical accuracy by collecting precise environmental and biological data. Software-based biomarkers will transform raw inputs into structured health insights using analytical tools.
AI-based biomarkers will enhance interpretation by identifying patterns that traditional methods may overlook. These solutions will support deeper analysis and improve reliability in health tracking. Other categories will include hybrid approaches that combine multiple data sources. Growth across types will depend on usability, data accuracy, and integration with healthcare systems.
By Application
Clinical trials will use digital biomarkers to collect real-time patient data, improving study efficiency and reducing dependency on site visits. Diagnostics will benefit from early detection capabilities driven by continuous monitoring and data analysis. Remote monitoring will expand as healthcare providers seek scalable solutions for chronic condition management. Drug development will gain from precise patient insights, enabling better outcome tracking.
Disease management will remain a key application, with ongoing tracking supporting long-term treatment plans. Digital tools will allow timely interventions and improved patient engagement. Other applications will include wellness tracking and preventive healthcare strategies. Adoption across applications will depend on accuracy, regulatory acceptance, and integration with clinical workflows.
To learn more about this report, Download Free Sample Report
By End-User
Pharma companies will adopt digital biomarkers to improve clinical trial design and drug evaluation processes. Healthcare providers will use these tools for patient monitoring, diagnostics, and treatment optimization. Research institutes will explore data-driven insights to support innovation and medical discoveries. Biotech firms will integrate biomarker data into product development pipelines.
Hospitals will implement digital monitoring systems to enhance patient care and reduce readmission rates. Other end-users will include wellness platforms and digital health startups. Adoption across end-users will depend on infrastructure readiness and data management capabilities. Market participation will grow as organizations seek efficient and scalable healthcare solutions.
By Technology
AI will drive advanced data interpretation, enabling pattern recognition and predictive health insights. IoT will support seamless connectivity between devices, ensuring continuous data flow across platforms. Mobile apps will act as user interfaces, allowing easy access to health information and engagement tools. Cloud platforms will enable secure data storage and real-time access across healthcare systems.
Data analytics will transform collected information into actionable insights for decision-making. These tools will support both clinical and operational improvements. Other technologies will include emerging frameworks that enhance data security and interoperability. Technology adoption will remain central to scaling digital biomarker solutions across Europe.
What are the Main Challenges for the Europe Digital Biomarkers Market Growth?
The europe digital biomarkers market faces several technical and operational challenges that continue to limit scalability and consistent performance. Data accuracy remains a key concern, as variations in wearable devices and sensor calibration can affect reliability. Integration of multiple data streams into unified platforms often requires complex architectures, slowing deployment across healthcare systems. These challenges in europe digital biomarkers market environments also include interoperability issues, where different technologies struggle to communicate effectively, creating operational inefficiencies.
Manufacturing and commercialization barriers further restrict progress in the europe digital biomarkers market. Companies must comply with strict European regulatory frameworks, which demand clinical validation and data security assurance before product approval. This process increases development costs and extends time-to-market, creating pressure on smaller firms. Market limitations also arise from the need for standardized protocols, as inconsistent validation methods can delay commercialization and reduce stakeholder confidence.
Adoption challenges continue to affect the europe digital biomarkers market, particularly in regions with limited digital health infrastructure. Healthcare providers often face a shortage of skilled professionals capable of managing advanced data systems, which slows implementation. Funding gaps and budget constraints in emerging European markets also restrict large-scale deployment. These growth barriers highlight the need for improved training, infrastructure investment, and streamlined integration to support broader adoption.
Competitive pressure from alternative diagnostic technologies and evolving regulatory expectations introduces further risks to the europe digital biomarkers market. Traditional clinical tools still hold strong market positions, creating pricing pressure and slowing transition toward digital solutions. At the same time, rapid technological advancements may outpace regulatory updates, adding uncertainty and increasing compliance risks for market participants.
Country Insights
Europe digital biomarkers market will reflect steady progress supported by connected health devices and data-driven care models. Healthcare systems will adopt continuous monitoring to replace episodic observation, improving early detection and treatment planning. Consumer demand will shift toward simple, accurate, and non-invasive solutions that fit daily routines. Regulatory alignment across European regions will strengthen trust and support clinical validation of digital tools.
Technology adoption will increase as AI, cloud platforms, and IoT improve data collection and interpretation. Companies will invest in scalable systems that connect devices with healthcare providers for real-time insights. Market direction will remain influenced by data privacy expectations and secure infrastructure needs. Industry participants will focus on reliability and integration to maintain long-term adoption.
Western Europe will dominate due to strong healthcare infrastructure, higher digital adoption, and supportive regulatory frameworks. Countries such as Germany, France, and the UK will lead in implementation of wearable and AI-based monitoring systems. Investment in digital health platforms and public-private partnerships will further strengthen regional leadership. Demand will remain high for advanced patient monitoring solutions.
Eastern Europe will emerge as a fast-growing region due to rising healthcare digitization and increasing awareness of remote monitoring solutions. Governments will support digital transformation through funding and policy initiatives. Infrastructure development and mobile health adoption will improve access to digital biomarker tools. Growth potential will remain strong as technology becomes more affordable and widely available.
Recent Development News
In March 2026, Kaia Health announced acquisition. The company was acquired in a deal valued at around $285 million, marking one of the largest digital health M&A transactions in Europe and strengthening its position in digital therapeutics and biomarker-driven care.https://www.galengrowth.com/europe-digital-health-funding-q1-2026-analysis/ (Source)
In March 2026, Gleamer announced acquisition. The AI-based medical imaging firm was acquired in a transaction valued at approximately $267 million, highlighting growing consolidation in AI-enabled diagnostics closely linked to digital biomarker development.https://www.galengrowth.com/europe-digital-health-funding-q1-2026-analysis/ (Source)
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 1188.9 Million |
|
Market size value in 2026 |
USD 1482.6 Million |
|
Revenue forecast in 2033 |
USD 7039.4 Million |
|
Growth rate |
CAGR of 24.92% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain, Rest of Europe) |
|
Key company profiled |
Roche, Novartis, Pfizer, IBM, Apple, Google, Medtronic, Philips, Siemens Healthineers, Biogen, Sanofi, AstraZeneca, Oracle, SAP, Cognizant |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Wearable Biomarkers, Mobile-based Biomarkers, Sensor-based Biomarkers, Software-based Biomarkers, AI-based Biomarkers, Others); By Application (Clinical Trials, Diagnostics, Remote Monitoring, Drug Development, Disease Management, Others); By End-User (Pharma, Healthcare Providers, Research Institutes, Biotech, Hospitals, Others); By Technology (AI, IoT, Mobile Apps, Cloud Platforms, Data Analytics, Others) |
How Can New Companies Establish a Strong Foothold in the Europe Digital Biomarkers Market?
New companies can secure early traction in the europe digital biomarkers market by targeting focused clinical use cases where measurable outcomes matter, such as remote cardiac monitoring or early neurological assessment. Precision-led niche targeting will help align products with clear healthcare pain points, including delayed diagnosis and fragmented patient data. In the europe digital biomarkers market, firms that build solutions around validated endpoints and real-world evidence will gain faster clinical acceptance and support ongoing market growth.
Innovation strategies will depend on combining machine learning with high-quality sensor data to deliver predictive analytics rather than basic tracking. Technology differentiation will come from interoperable platforms that integrate with hospital systems and electronic health records. In the europe digital biomarkers market, companies that prioritize data security, GDPR compliance, and explainable AI models will build stronger credibility with providers and regulators.
Partnerships will play a critical role in scaling. Collaborations with pharmaceutical companies, CROs, and academic institutions will accelerate validation and commercialization. Emerging players such as Koneksa and Huma show how co-development models and platform-based ecosystems can drive adoption. These approaches will strengthen positioning within the europe digital biomarkers market by aligning innovation with clinical and commercial needs.
Long-term success will depend on solving operational challenges like data fragmentation and patient engagement. Companies that offer seamless user experiences, real-time insights, and scalable cloud infrastructure will stand out. As industry trends shift toward personalized and preventive care, new entrants will need to combine technical strength with practical healthcare impact to compete effectively.
Key Europe Digital Biomarkers Market Company Insights
into europe digital biomarkers market will depend on clear focus on specific healthcare gaps such as remote monitoring and early-stage diagnosis. Strong clinical validation will support acceptance among healthcare providers and regulators. Data accuracy and ease of use will remain key factors influencing adoption across hospitals and research settings.
Innovation will center on AI-driven analytics and connected devices that will deliver real-time insights. Secure data handling and compliance with European standards will build trust and long-term value. Startups such as Huma and Koneksa highlight how targeted solutions and scalable platforms will strengthen market entry strategies.
Competitive insights:
Competition in europe digital biomarkers market will remain strong as both established firms and new entrants focus on advanced solutions. Strategic partnerships with pharmaceutical companies and healthcare institutions will improve product reach and validation. Cost efficiency and flexible platforms will support differentiation in crowded segments.
Larger companies will expand capabilities through acquisitions and integration of advanced analytics technologies. Continuous investment in cloud systems and machine learning will enhance performance and scalability. Competitive advantage will depend on speed of deployment, accuracy of insights, and ability to meet evolving healthcare requirements.
Company List
- Roche,
- Novartis,
- Pfizer,
- IBM, Apple
- Google,
- Medtronic,
- Philips,
- Siemens Healthineers,
- Biogen,
- Sanofi,
- AstraZeneca,
- Oracle,
- SAP,
- Cognizant
What are the Key Use-Cases Driving the Growth of the Europe Digital Biomarkers Market?
The europe digital biomarkers market is gaining momentum through practical healthcare use-cases that focus on continuous patient monitoring and early intervention. Remote patient monitoring remains a primary driver, where wearable devices track vital signs such as heart rate, sleep patterns, and activity levels. These insights allow healthcare providers to detect abnormalities early and reduce hospital visits. The europe digital biomarkers market benefits from this shift toward preventive care, which improves patient outcomes and lowers system costs.
Another key use-case in the europe digital biomarkers market lies in clinical trials and drug development. Digital biomarkers enable real-time data collection outside clinical settings, improving trial accuracy and patient compliance. Pharmaceutical companies use these tools to monitor treatment responses and adjust protocols faster. This approach reduces trial timelines and enhances decision-making, supporting faster innovation and increasing adoption across the industry.
Chronic disease management is also driving demand within the europe digital biomarkers market, especially for conditions such as diabetes, cardiovascular disorders, and mental health issues. Continuous tracking supports personalized treatment plans and better patient engagement. At the same time, consumer health applications are expanding, where individuals use mobile apps and wearables to manage wellness and fitness goals.
Emerging opportunities are developing through AI-powered analytics and integration with smart healthcare systems. The europe digital biomarkers market will scale further as technology enables predictive insights and seamless data sharing across platforms. This will support personalized medicine and strengthen long-term growth potential.
Europe Digital Biomarkers Market Report Segmentation
By Type
- Wearable Biomarkers
- Mobile-based Biomarkers
- Sensor-based Biomarkers
- Software-based Biomarkers
- AI-based Biomarkers
- Others
By Application
- Clinical Trials
- Diagnostics
- Remote Monitoring
- Drug Development
- Disease Management
- Others
By End-User
- Pharma
- Healthcare Providers
- Research
- Institutes
- Biotech
- Hospitals
- Others
By Technology
- AI
- IoT
- Mobile Apps
- Cloud Platforms
- Data Analytics
- Others
Frequently Asked Questions
Find quick answers to common questions.
The Approximate Europe Digital Biomarkers Market size for the Market will be USD 7039.4 Million in 2033.
Key segments for the Europe Digital Biomarkers Market are By Type (Wearable Biomarkers, Mobile-based Biomarkers, Sensor-based Biomarkers, Software-based Biomarkers, AI-based Biomarkers, Others); By Application (Clinical Trials, Diagnostics, Remote Monitoring, Drug Development, Disease Management, Others); By End-User (Pharma, Healthcare Providers, Research Institutes, Biotech, Hospitals, Others); By Technology (AI, IoT, Mobile Apps, Cloud Platforms, Data Analytics, Others).
Major Europe Digital Biomarkers Market players are Roche, Novartis, Pfizer, IBM, Apple, Google, Medtronic, Philips, Siemens Healthineers, Biogen, Sanofi, AstraZeneca, Oracle, SAP, Cognizant.
The Europe Digital Biomarkers Market size is USD 1188.9 Million in 2025.
The Europe Digital Biomarkers Market CAGR is 24.92%.
- Roche,
- Novartis,
- Pfizer,
- IBM, Apple
- Google,
- Medtronic,
- Philips,
- Siemens Healthineers,
- Biogen,
- Sanofi,
- AstraZeneca,
- Oracle,
- SAP,
- Cognizant
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


