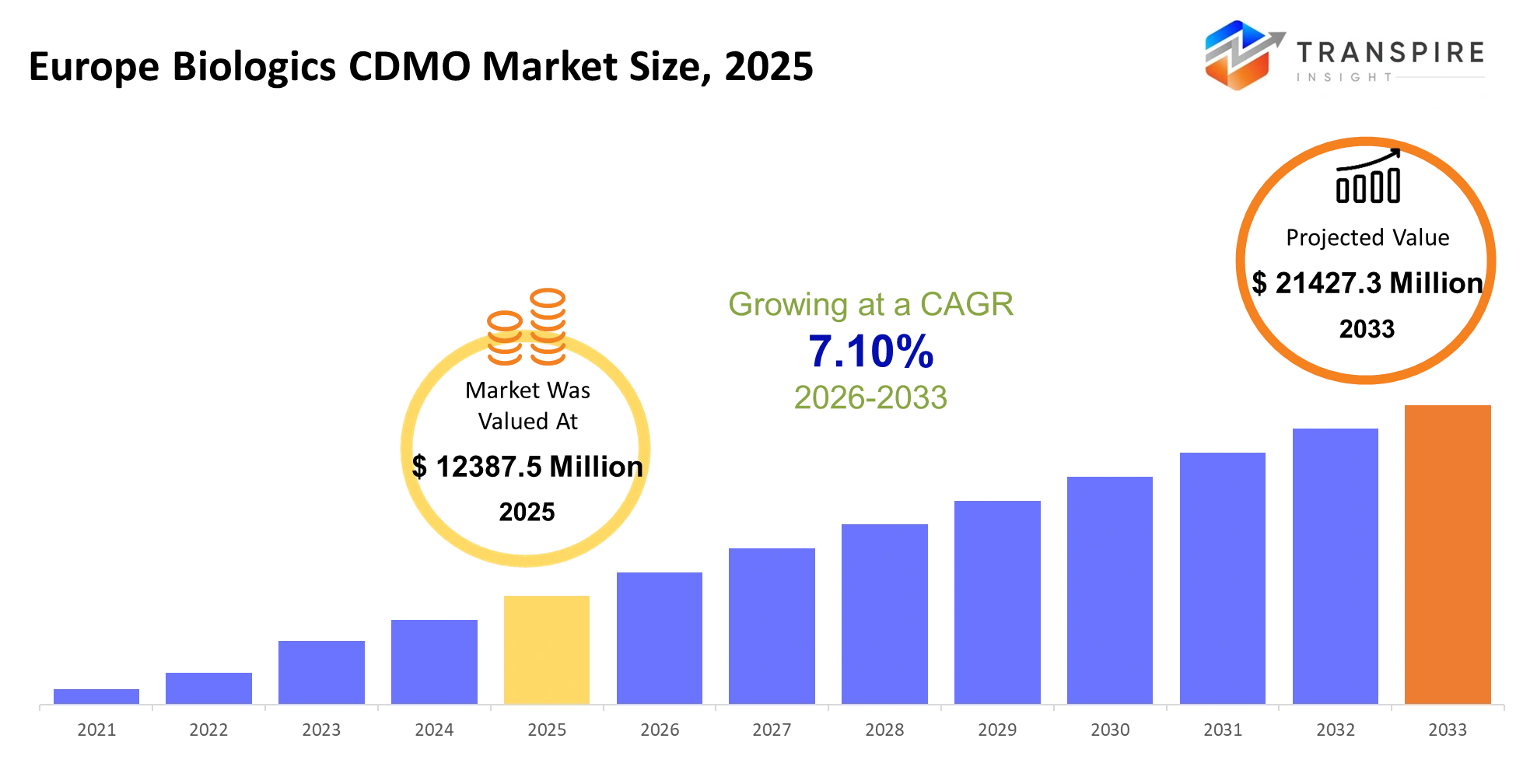
Europe Biologics CDMO Market Size & Forecast:
- Europe Biologics CDMO Market Size 2025: USD 12387.5 Million
- Europe Biologics CDMO Market Size 2033: USD 21427.3 Million
- Europe Biologics CDMO Market CAGR: 7.10%
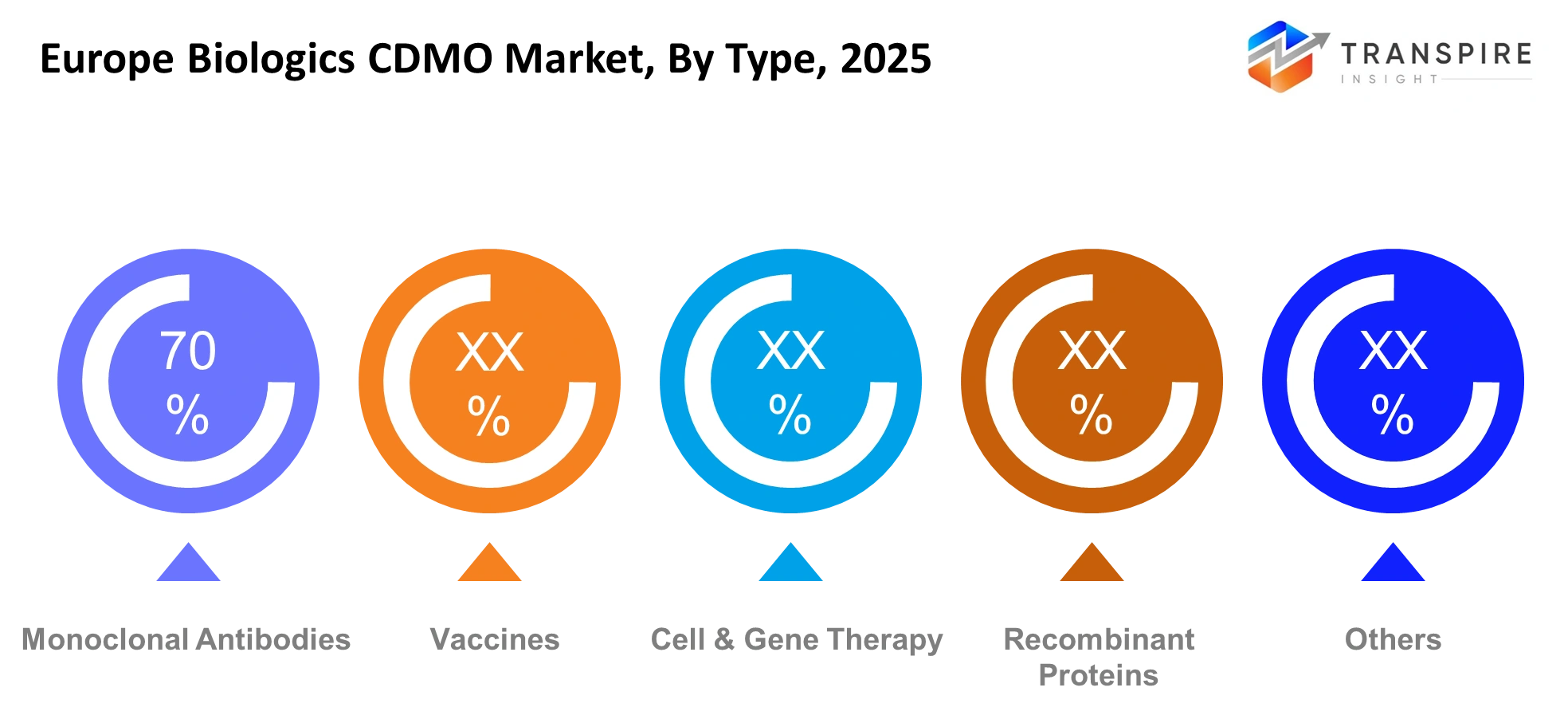
- Europe Biologics CDMO Market Segments: By Type (Monoclonal Antibodies, Vaccines, Cell & Gene Therapy, Recombinant Proteins, Others), By Application (Oncology, Infectious Diseases, Autoimmune Diseases, Others), By End-User (Pharma Companies, Biotech Firms, Others), By Service (Manufacturing, Development, Packaging, Others).
To learn more about this report, Download Free Sample Report
Europe Biologics CDMO Market Summary:
The Europe Biologics CDMO Market size is estimated at USD 12387.5 Million in 2025 and is anticipated to reach USD 21427.3 Million by 2033, growing at a CAGR of 7.10% from 2026 to 2033. The Europe Biologics CDMO market provides essential operational support to pharmaceutical and biotech companies by delivering outsourced development and manufacturing services for complex biologic drugs which include monoclonal antibodies and cell therapies and recombinant proteins. CDMOs enable drug developers to transition their production from clinical batches to commercial-scale operations because they provide all necessary facilities and regulatory systems and bioprocessing expertise without requiring billion-dollar investments.
The market has transitioned during the past five years from traditional large-batch biologics manufacturing to flexible production platforms which use single-use technologies and modular bioprocessing systems to create high-value products. The COVID-19 pandemic served as a significant catalyst which revealed Europe’s reliance on fragmented global supply chains while it sped up regional investments into domestic biologics manufacturing infrastructure development. Pharmaceutical companies now depend on CDMOs because their biotech pipelines become more specialized and time-sensitive to support development risk mitigation and commercialization speed and capital resource protection. The European biologics ecosystem experiences outsourcing growth which establishes permanent manufacturing relationships while developing new business models that generate consistent revenue streams.
Key Market Insights
- The European Biologics CDMO Market in 2025 experienced its highest market share through Western Europe which controlled 58% of the market because of its advanced biopharmaceutical manufacturing facilities.
- Germany holds the top position in regional biologics outsourcing because it invests heavily in GMP biologics production facilities and regulatory compliance capabilities.
- The United Kingdom maintains its position as a major biological development innovation center through its growing number of clinical-stage biotechnology companies.
- The Central and Eastern European region will remain the fastest expanding area until 2030 because of its lower operational expenses and planned business development projects.
- The Europe biologics outsourcing market for the year 2025 saw biologics manufacturing services control almost 46% of the market because of the need for production at commercial scale.
- Biotech companies use process development services as their major service which enables them to complete research activities between clinical and commercial stages at their quickest pace.
- The cell and gene therapy manufacturing sector will experience the most rapid expansion until 2030 because of rising demand for precision medicine and specialized contract development and manufacturing organizations.
- The bioprocessing industry widely adopted single-use technologies because they help facilities achieve their production goals while decreasing the chance of contamination.
- The production of monoclonal antibodies generated 42% of total revenue through its various applications because of strong oncology biologic product development.
- Biosimilar manufacturing emerged as a high-growth application segment as patent expirations increased competitive biologics development across Europe.
- The Europe Biologics CDMO Market saw large pharmaceutical companies achieve market dominance through strategic outsourcing models which enabled them to control over 51% of the total market.
What are the Key Drivers, Restraints, and Opportunities in the Europe Biologics CDMO Market?
The Europe Biologics CDMO Market experiences its most powerful movement through the increasing development of biologic drug pipelines which include monoclonal antibodies and biosimilars and cell therapies. Pharmaceutical companies increasingly outsource manufacturing because biologics production now requires highly specialized bioreactors, sterile fill-finish systems, and advanced regulatory compliance that demand multi-billion-euro capital investment. Major drug developers established long-term CDMO partnerships throughout Europe after the COVID-19 pandemic demonstrated that limited in-house manufacturing capabilities create commercial hazards. CDMOs gained higher revenue visibility through multi-year manufacturing contracts and capacity reservation agreements and integrated development services which included clinical trials and commercial production.
The market’s largest structural barrier is the shortage of qualified biologics manufacturing capacity combined with a limited pool of experienced bioprocessing talent. Building GMP-certified biologics facilities can take several years due to strict European regulatory validation, contamination control requirements, and technology installation timelines. The demand for upstream and downstream processing specialists currently exceeds the available workforce supply. The technology transfer projects experience delays because of this bottleneck which also prevents production growth and decreases potential revenue from late-stage biologics programs that need fast market entry.
The Europe Biologics CDMO Market will experience its next major growth phase through continuous bioprocessing and modular manufacturing systems. European governments and private investors are funding flexible biologics facilities which can quickly switch between different therapies and production levels. The combination of skilled scientific talent and favorable regulatory environments and strong pharmaceutical ecosystems makes Ireland and Belgium attractive for next-generation biologics hub investments. The technologies enable production to run without interruptions while increasing output productivity and helping CDMOs to deliver profitable services to their small biotech customers which creates new ongoing income opportunities in personalized medicine and orphan drug production.
What Has the Impact of Artificial Intelligence Been on the Europe Biologics CDMO Market?
Artificial intelligence and advanced digital technologies transform the Europe Biologics CDMO Market through their ability to enhance production accuracy and decrease operational defects and speed up the process of bringing biologics to market. The CDMO industry adopts AI manufacturing execution systems to track bioreactor environments while the systems handle automatic contamination detection and control both upstream and downstream production processes in real time. The systems use continuous monitoring to analyze multiple factors including temperature and oxygen concentration and cell density and nutrient consumption which enables them to sustain consistent biologics production while minimizing operational deviations.
Across biologics production facilities machine learning models enhance the effectiveness of predictive maintenance programs. Advanced analytics platforms enable the detection of equipment performance anomalies which helps operators to decrease unplanned downtime that occurs within critical GMP manufacturing facilities. European biologics manufacturers use digital twins and AI-based simulation tools to predict batch performance and enhance process scalability and decrease the time needed to transition from clinical to commercial production. The technologies enable organizations to achieve better facility utilization and decreased material waste and improved regulatory compliance through their ability to automatically track data throughout the organization.
The adoption of AI technology by organizations encounters a major problem because the biologics manufacturing industry stores its data in separate systems that use outdated technology and maintain proprietary control over their platforms. The implementation of AI technology in regulated GMP environments requires organizations to complete three processes which include system validation and cybersecurity protection measures and employee training programs. The need for these three processes results in higher costs for implementing AI systems and creates delays in deployment activities for smaller CDMO facilities.
Key Market Trends
- The pharmaceutical industry now establishes multi-year CDMO contracts to obtain manufacturing resources since their initial need to address pandemic-related biologics supply shortages caused delays in product development.
- The use of single-use bioprocessing systems has completely replaced traditional stainless-steel systems throughout many European facilities, which enables commercial operations to achieve 30% more time for production because they need less time for cleaning maintenance.
- Between 2022 and 2025 Lonza Group and Catalent established their European biologics expansion program to obtain profitable cell therapy contracts.
- The demand for biosimilars increased after major biologics lost their patent protections which led European pharmaceutical companies to implement cost-effective production through third-party manufacturing.
- CDMOs implemented AI-based process analytics solutions starting in 2023 to help them achieve two goals of decreasing batch failures and enhancing GMP compliance documentation precision.
- Mid-sized biotech companies preferred to outsource their late-stage biologics production because rising interest rates made building internal manufacturing facilities too expensive after 2022.
- European regulators established stronger supply chain resilience requirements which followed COVID-19, and their new rules promoted companies to build local biologics production facilities in Germany, Belgium, and Ireland.
- Since 2020, manufacturers have adopted continuous bioprocessing technologies because they want to increase production while decreasing operational waste.
- Major CDMOs in the biologics outsourcing market expanded their service capabilities through strategic acquisitions, which enabled them to offer complete solutions from clinical development to commercial manufacturing.
- Following 2021, cell and gene therapy manufacturing served as the most rapidly evolving service area because of rising approval rates for personalized biologic treatments throughout Europe.
Europe Biologics CDMO Market Segmentation
By Type
Biologics outsourcing practices remain predominantly controlled by monoclonal antibodies because oncology treatments and autoimmune disease therapies require extensive antibody manufacturing capacity. The successful commercial performance of antibody-based treatments led pharmaceutical companies to establish permanent biologics production contracts with specialized contract development and manufacturing organizations throughout Europe. The market for recombinant proteins and vaccines remains strong because clinical requirements and government-funded immunization initiatives continue to support their use. The cell and gene therapy manufacturing sector currently experiences its fastest growth rate because personalized treatment development requires manufacturers to establish unique production facilities which comply with intricate regulatory standards.
Biotech companies have increased their outsourcing work because they possess restricted production capabilities for their advanced therapy products. Future investment trends will direct funds toward three main areas which include modular cleanroom systems as well as flexible single-use bioprocessing equipment and facilities designed for high-containment operations which will enable small-scale precise treatment production. Companies that can produce antibodies at commercial levels while developing advanced therapies with flexible production methods will achieve better contract outcomes and higher-value biopharmaceutical products.
To learn more about this report, Download Free Sample Report
By Application
Biologics outsourcing services currently operate their biggest business sector through oncology applications because cancer drug development keeps growing through monoclonal antibodies, antibody-drug conjugates, and immunotherapy treatments. The pharmaceutical industry increasingly relies on outsourcing for oncology biologics manufacturing because this approach helps companies decrease their development time while obtaining access to advanced mammalian cell culture technology. Doctors maintain a strong preference for prescribing biologics as treatments for rheumatoid arthritis and psoriasis and inflammatory bowel disorders which supports the continued demand in the autoimmune disease therapy market.
The pandemic vaccine manufacturing investments combined with regional biologics supply chain restructuring activities have established stronger commercial momentum for infectious disease applications. The field of rare disease and orphan biologics programs will achieve substantial growth because regulatory incentives enable faster product approvals while companies can implement higher pricing strategies. The increasing clinical complexity found in therapeutic applications will prompt CDMOs to implement integrated development and manufacturing systems which utilize unified regulatory frameworks for managing process optimization and analytical testing and commercial production activities.
By End-User
The major pharmaceutical companies control the majority of outsourced biologics production because drug manufacturers now favor operating models that require fewer assets and permit them to produce their products through third-party manufacturers. The rising demand for capital expenditures to establish biologics facilities pushed pharmaceutical companies to pursue strategic outsourcing agreements instead of constructing their own manufacturing sites. Venture-backed biotech companies represent the fastest-growing user group because these startups lack experience in commercial-level production and they do not have certified facilities for good manufacturing practice. Clinical-stage companies of smaller size now rely more on combined CDMO solutions which enable them to handle their regulatory applications and process verification and technology handover work.
Early-stage biologics research and translational medicine projects at academic institutions and specialized research organizations create active market participation. The upcoming market analysis indicates that CDMOs will establish closer partnerships with emerging biotech firms which focus on developing cell therapies and gene editing and precision biologics. The service providers who can assist their clients from initial clinical development through to commercial product introduction will achieve higher customer loyalty while creating continuous revenue streams from multiple production phases.
By Service
The outsourcing industry mainly relies on manufacturing services because biopharmaceutical production needs particular bioreactors and contamination prevention systems and operational facilities that comply with Good Manufacturing Practice standards which require high costs for companies to build internally. The biologics industry generates its highest revenue through commercial manufacturing because it operates under long-term production contracts which guarantee ongoing supply of developed drugs. The market for development services remains important because pharmaceutical and biotech companies now prefer to hire outside professionals who specialize in cell line development and process optimization and analytical characterization.
The demand for packaging and fill-finish services increased when European regulators required companies to protect their supply networks and maintain the cold-chain system for temperature-sensitive biologics. Service delivery systems now use continuous manufacturing technologies together with automated quality monitoring systems to achieve better batch quality and decreased downtime in their operations. The ability of a business to compete in the future will depend on its capacity to deliver integrated service solutions that unite research and development and manufacturing and packaging and regulatory assistance through one system which enables faster drug development.
What are the Key Use Cases Driving the Europe Biologics CDMO Market?
The primary application which drives European markets to adopt biologics CDMO services centers on commercial production of monoclonal antibody treatments. The oncology drug development process requires companies to outsource their operations because antibody therapies demand specialized mammalian cell culture systems and large-scale bioreactors which must follow strict Good Manufacturing Practices.
The production of vaccines and manufacturing of biosimilars have become new fields of expansion for mid-sized biotechnology companies and regional pharmaceutical businesses. European health systems now create supportive environments for biosimilar treatments which enable cost savings through enhanced process development and specialized CDMO facilities that handle both development and fill-finish duties.
Cell and gene therapy manufacturing has established itself as an essential industrial process for companies developing treatments that target rare diseases and personalized medicine. Developers of advanced therapies now require cleanroom facilities that operate as modular units together with automated systems for producing small quantities of specialized biologic products which need to be developed for quick clinical testing.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 12387.5 Million |
|
Market size value in 2026 |
USD 13254.8 Million |
|
Revenue forecast in 2033 |
USD 21427.3 Million |
|
Growth rate |
CAGR of 7.10% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Europe (Germany, United Kingdom, France, Italy, Spain and Rest of Europe) |
|
Key company profiled |
Lonza, Catalent, Samsung Biologics, WuXi Biologics, Thermo Fisher, Boehringer Ingelheim, Fujifilm Diosynth, Recipharm, Aenova, Piramal Pharma, Rentschler Biopharma, AGC Biologics, KBI Biopharma, Abzena, Cytovance. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Monoclonal Antibodies, Vaccines, Cell & Gene Therapy, Recombinant Proteins, Others), By Application (Oncology, Infectious Diseases, Autoimmune Diseases, Others), By End-User (Pharma Companies, Biotech Firms, Others), By Service (Manufacturing, Development, Packaging, Others). |
Which Regions are Driving the Europe Biologics CDMO Market Growth?
Western Europe maintains its position as the primary market for biologics CDMO services because the region possesses both advanced pharmaceutical manufacturing facilities and effective regulatory systems and extensive biotechnology research networks. The three countries of Germany, Switzerland, and Belgium create strong biologics development systems through their combination of manufacturing facilities and research institutes and clinical trial organizations. The European Medicines Agency enforces stringent compliance requirements which lead pharmaceutical companies to select CDMO partners who possess expertise in handling intricate biologics validation processes. The combination of established cold-chain logistics networks and bioprocessing experts and active venture capital funding establishes Western Europe as the primary force in both commercial biologics production and advanced therapy manufacturing.
Northern Europe develops its own biologics outsourcing system because market stability in the region depends more on operational efficiency and pharmaceutical investments and healthcare systems that drive innovation than on manufacturing capacity. The government provides ongoing support for Denmark and Sweden's biologics research ecosystems which enable academic institutions and pharmaceutical manufacturers to work together. CDMOs in the region now focus their operations on specific biologics markets that include recombinant proteins and metabolic disease treatments and precision biologics development. Pharmaceutical companies establish multi-year manufacturing contracts with stable reimbursement systems and predictable regulatory processes because these systems present less operational risk than unstable global outsourcing locations.
Who are the Key Players in the Europe Biologics CDMO Market and How Do They Compete?
The Europe Biologics CDMO Market exhibits moderate market consolidation which results in multinational CDMOs dominating most commercial biologics manufacturing operations while regional companies operate in specific treatment markets. Competitors now fight for advantage through their technological abilities and their capacity to complete regulatory procedures and their ability to provide complete service solutions instead of competing over manufacturing expenses. Established companies use three main methods to maintain their market position which include establishing long-term pharmaceutical collaborations and developing advanced therapy facilities and building complete biologics development systems. Emerging CDMOs build their market positions by providing flexible small-batch production capabilities and fast clinical development processes and specialized cell and gene therapy services which larger facilities struggle to deliver effectively.
The Lonza Group establishes its unique market position through its ability to provide high-capacity mammalian cell culture production and its complete biologics development services which cover everything from preclinical research to commercial manufacturing. The company expands its modular biologics production facilities throughout Europe to meet the increasing demand for both personalized medicine and advanced antibody production. Catalent uses its advanced delivery systems and sterile fill-finish production capabilities to help pharmaceutical companies combine their entire production process into a single external production system. The company boosts customer loyalty and business expansion through its funding of biologics packaging automated systems and facilities which handle high-potency product manufacturing operations.
WuXi Biologics uses its rapid development capabilities and flexible production systems to assist European biotechnology companies in their clinical-stage product development and market entry processes.
Company List
- Lonza
- Catalent
- Samsung Biologics
- WuXi Biologics
- Thermo Fisher
- Boehringer Ingelheim
- Fujifilm Diosynth
- Recipharm
- Aenova
- Piramal Pharma
- Rentschler Biopharma
- AGC Biologics
- KBI Biopharma
- Abzena
- Cytovance
Recent Development News
In May 2026, Lonza Reaffirms 2026 CDMO Growth Outlook Amid Strong Biologics Demand: Swiss biologics CDMO major Lonza confirmed sustained outsourcing demand from pharmaceutical and biotech companies in Q1 2026. The company stated that its integrated biologics operations continued to gain momentum despite global geopolitical uncertainty and shifting U.S. investment strategies. The update reinforces Europe’s strong positioning in high-value biologics manufacturing.
Source: https://www.wsj.com
In January 2026, Lonza Forecasts Double-Digit 2026 Growth for Core CDMO Business: Lonza projected 11–12% sales growth for 2026 and highlighted strong biologics manufacturing performance driven by mammalian, bioconjugate, and drug product platforms. The company also reported continued expansion in advanced synthesis capabilities, strengthening Europe’s role in global biologics outsourcing.
Source: https://www.reuters.com
What Strategic Insights Define the Future of the Europe Biologics CDMO Market?
The Europe Biologics CDMO Market is currently transitioning towards specialized manufacturing networks which rely on modern technology to produce advanced therapies through flexible manufacturing processes and collaborative research development initiatives. The primary force driving this transition is the rapid shift from blockbuster biologics toward smaller patient-population therapies that require agile manufacturing, faster clinical scaling, and tighter regulatory coordination. The period from five to seven years ahead will establish digital process optimization and modular facility design as essential elements for businesses to gain competitive advantages while they develop their capacity to produce cell and gene therapies using lower production volumes.
The distribution of essential raw materials for cell culture media and single-use bioprocessing components and viral vector production represents an unrecognized danger which needs to be addressed. A disruption within a small group of specialized suppliers could create significant production bottlenecks despite strong market demand. Central and Eastern Europe have become significant biologics investment hubs because their operating expenses remain low while their biotechnology programs continue to expand. Companies entering the market should prioritize hybrid manufacturing strategies that combine large-scale biologics capacity with flexible small-batch infrastructure capable of adapting to next-generation precision therapies.
Europe Biologics CDMO Market Report Segmentation
By Type
- Monoclonal Antibodies
- Vaccines
- Cell & Gene Therapy
- Recombinant Proteins
- Others
By Application
- Oncology
- Infectious Diseases
- Autoimmune Diseases
- Others
By End-User
- Pharma Companies
- Biotech Firms
- Others
By Service
- Manufacturing
- Development
- Packaging
- Others
Frequently Asked Questions
Find quick answers to common questions.
The approximate Europe Biologics CDMO Market size for the market will be USD 21427.3 Million in 2033.
The key segments of the Europe Biologics CDMO Market are By Type (Monoclonal Antibodies, Vaccines, Cell & Gene Therapy, Recombinant Proteins, Others), By Application (Oncology, Infectious Diseases, Autoimmune Diseases, Others), By End-User (Pharma Companies, Biotech Firms, Others), By Service (Manufacturing, Development, Packaging, Others).
Major players in the Europe Biologics CDMO Market are Lonza, Catalent, Samsung Biologics, WuXi Biologics, Thermo Fisher, Boehringer Ingelheim, Fujifilm Diosynth, Recipharm, Aenova, Piramal Pharma, Rentschler Biopharma, AGC Biologics, KBI Biopharma, Abzena, Cytovance.
The current market size of the Europe Biologics CDMO Market is USD 12387.5 Million in 2025.
The Europe Biologics CDMO Market CAGR is 7.10%.
- Lonza
- Catalent
- Samsung Biologics
- WuXi Biologics
- Thermo Fisher
- Boehringer Ingelheim
- Fujifilm Diosynth
- Recipharm
- Aenova
- Piramal Pharma
- Rentschler Biopharma
- AGC Biologics
- KBI Biopharma
- Abzena
- Cytovance
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


