Asia Pacific AI in Predictive Toxicology Market Size & Forecast:
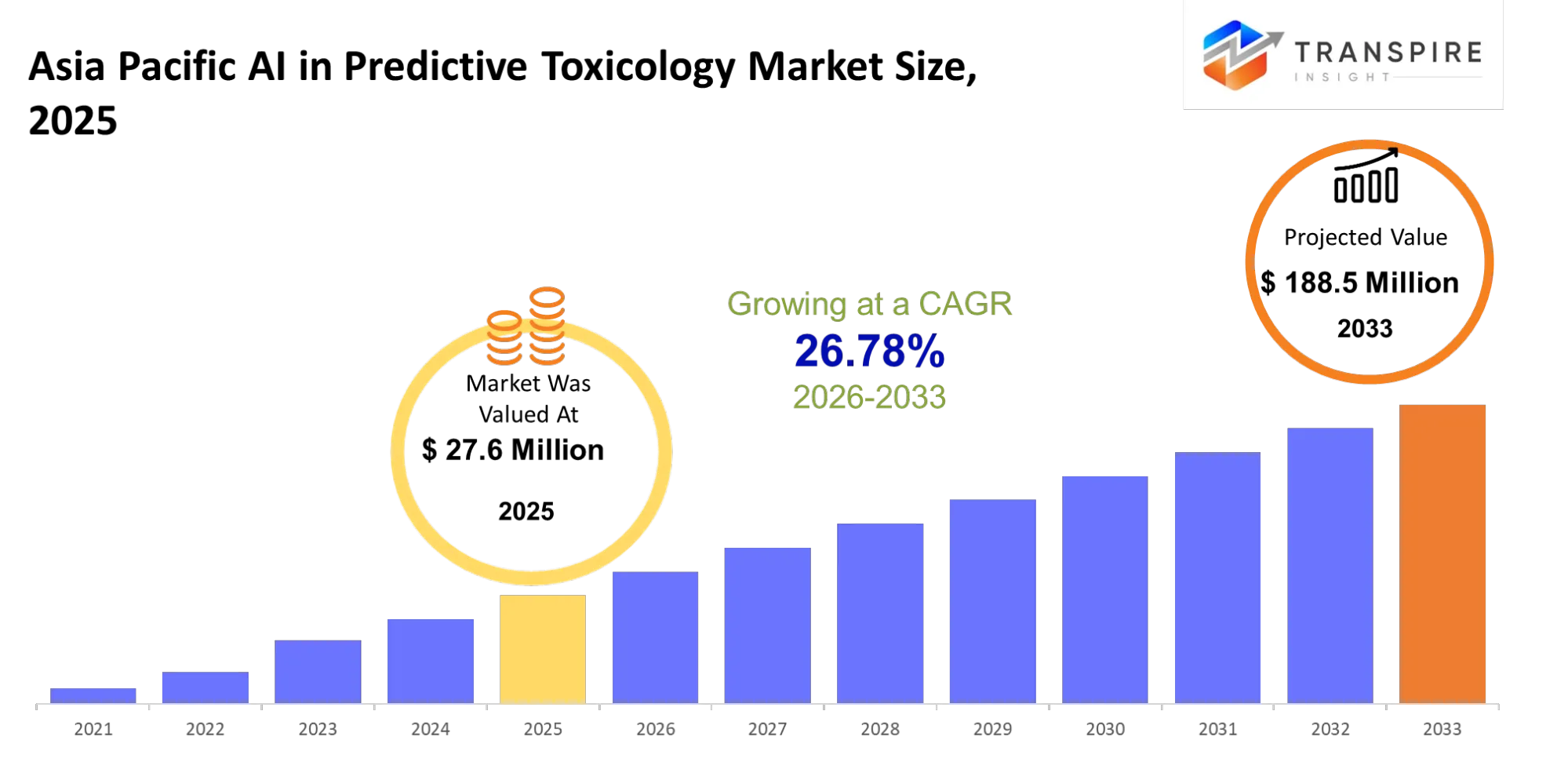
- Asia Pacific AI in Predictive Toxicology Market Size 2025: USD 27.6 Million
- Asia Pacific AI in Predictive Toxicology Market Size 2033: USD 188.5 Million
- Asia Pacific AI in Predictive Toxicology Market CAGR: 26.78%
- Asia Pacific AI in Predictive Toxicology Market Segments: By Type (Machine Learning Models, Deep Learning Platforms, AI Software, Data Analytics Tools, Others); By Application (Drug Safety Testing, Chemical Toxicity Prediction, Environmental Risk Assessment, Food Safety, Cosmetics Testing, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Government Agencies, CROs, Others); By Deployment (Cloud, On-premise, Hybrid, Others)
To learn more about this report, Download Free Sample Report
Asia Pacific AI in Predictive Toxicology Market Summary
The Asia Pacific AI in Predictive Toxicology Market was valued at USD 27.6 Million in 2025. It is forecast to reach USD 188.5 Million by 2033. That is a CAGR of 26.78% over the period.
Pharmaceutical companies and chemical manufacturers and regulatory laboratories use the Asia Pacific AI in Predictive Toxicology market to detect potential toxicity risks during the early stages of product development. The platforms use biological and chemical and genomic data to create predictions about compound behavior which helps scientists save resources before they conduct lab testing or clinical-stage validation. The process helps organizations to reduce research time while decreasing failed projects which enables them to reach faster project decisions.
The market has evolved during the last five years from its previous reliance on rule-based computational toxicology tools toward machine learning predictive platforms which analyze multisonic data and actual experimental results. The major trigger resulted from countries including China and Japan and South Korea implementing stricter chemical safety regulations and pharmaceutical compliance standards which collided with pandemic emergency requirements for faster drug development. Companies started to spend money on digital toxicology systems which help them decrease their need for time-consuming animal testing and manual screening methods. Companies use AI-based toxicity prediction to enhance their research and development efficiency because development costs keep rising which leads to wider adoption of AI in their business operations and higher commercial investments throughout the region.
Key Market Insights
- China maintains market leadership in the Asia Pacific AI for Predictive Toxicology Market because its market share will reach 34% by 2025 through its active implementation of biotech digitalization projects.
- Japan operates as the second-largest market because its pharmaceutical sector uses artificial intelligence and its computational toxicology facilities provide strong support.
- India will maintain its status as the fastest-growing regional market until 2032 because its drug discovery outsourcing sector will generate annual growth rates exceeding 20%.
- South Korea demonstrates strong market expansion through its support of artificial intelligence life sciences research and its implementation of modernized regulatory systems.
- Software platforms dominate, capturing approximately 48% market share in 2025, which reflects enterprise demand for predictive toxicology automation tools that they provide.
- The second-largest segment of cloud-based predictive toxicology services exists because it requires lower infrastructure expenses and provides flexible system deployment options.
- The fastest-growing segment between now and 2030 will be AI-driven multiomics analytics platforms because precision medicine research continues to grow.
- Pharmaceutical companies use drug discovery applications which account for 42% of their resources because they need to predict toxicity at earlier stages to decrease their clinical trial failure rates.
- The market research shows that pharmaceutical and biotechnology companies hold 51% of the market share because they make significant financial commitments to develop predictive toxicology processes.
- Deep learning toxicity prediction models support product innovation which enables compound screening processes to achieve 20–30% improved accuracy during testing.
What are the Key Drivers, Restraints, and Opportunities in the Asia Pacific AI in Predictive Toxicology Market?
The strongest force accelerating the Asia Pacific AI in Predictive Toxicology market exists because chemical safety assessment regulations have become stricter while ethical standards mandate reduced animal testing. Regulatory agencies throughout Japan, South Korea, and China have implemented more rigorous validation standards for both pharmaceutical compounds and industrial chemicals during the last three years. Machine learning models that use toxicogenomic and bioactivity datasets show significant progress because AI predictions now achieve higher accuracy. The combination of these two factors compels pharmaceutical and biotechnology companies to start using predictive toxicology platforms during their early development stages which leads to increased software licensing revenue and more enterprise-scale platform subscriptions.
Access to standardized high-quality toxicology datasets remains the most significant structural obstacle that organizations face throughout the market. Organizations need to create predictive AI models which demand extensive validated biological datasets that support data sharing. Many research institutions and smaller pharmaceutical companies in Asia Pacific continue to use their outdated systems which results in their inability to access needed biological datasets. The procedure requires several years because it needs cross-border regulatory frameworks to reach agreement on data sharing and databases that need complete alignment with the project specifications. The process of data sharing agreements between organizations needs multiple years because it requires cross-border regulatory agreements to achieve complete data sharing and database standardization. The process of data sharing agreements between organizations needs multiple years because it requires cross-border regulatory agreements to achieve complete data sharing and database standardization.
Federated learning platforms which enable cross-institutional toxicology modeling work present a significant business growth opportunity. Singapore and other countries establish secure biomedical data ecosystems which permit organizations to develop artificial intelligence models through collaborative work without sharing their sensitive data. The new framework will enable more regions to participate in activities which will boost the rate of industry adoption.
What Has the Impact of Artificial Intelligence Been on the Asia Pacific AI in Predictive Toxicology Market?
Artificial intelligence transforms predictive toxicology processes throughout Asia Pacific by automating intricate screening methods and risk-assessment procedures which used to take laboratory tests several weeks to complete. Advanced machine learning systems now analyze chemical structure libraries and omics datasets and historical toxicological records to establish automated compound prioritization which identifies high-risk candidates before expensive in vitro or in vivo tests start. AI-assisted screening at pharmaceutical R&D centers across Japan, South Korea and Singapore enables 25 to 40 percent faster early-stage toxicity assessment which allows development pipelines to advance viable compounds more quickly.
Predictive capabilities have become particularly useful for predicting negative biological interactions. Deep learning models analyze molecular behavior patterns to estimate hepatotoxicity, cardiotoxicity, and genotoxicity risks with increasing precision, allowing developers to optimize candidate selection earlier. The organization achieved operational efficiency improvements through its dual achievements of reduced late-stage development failures and decreased testing expenses, which simultaneously enhanced its capacity to submit regulatory documents through improved computational evidence packages.
Current limitations remain significant. Model accuracy suffers from two main issues which include high costs needed to fully integrate legacy laboratory systems and restricted access to toxicology datasets that cover different regions. Many AI platforms still struggle to generalize across novel compound classes, which means human expert validation remains essential before critical development decisions are made.
Key Market Trends
- Pharmaceutical companies in Japan and South Korea have redirected approximately 30% of their initial toxicity testing budgets toward AI-based digital screening methods since 2022.
- China's regulatory bodies established new alternative testing methods in 2023 which required developers to use combined computer-based testing methods instead of traditional animal testing.
- Between 2023 and 2025 Certara and Schrödinger established new regional alliances to support their local toxicology model implementation efforts.
- Since 2021 toxicogenomics integration has become more common as buyers search for platforms that can analyze multiomics data instead of single endpoint toxicity models.
- Indian contract research organizations have expanded their AI toxicology platform funding by more than 20% each year since 2024 to fulfill growing needs for pharmaceutical evaluation services.
- The ability to explain a model's functions became essential for organizations after 2023 because regulatory requirements now demand companies to show their algorithms work through transparent decision-making processes which they use when submitting safety data for testing.
- The pandemic demonstrated that organizations needed distant work capabilities which led them to select cloud-based toxicology solutions because their existing on-premises systems could not handle their research operations at multiple sites.
- The 2025 business environment saw vendors change their operational patterns because they stopped selling independent software products and began offering unified toxicology-as-a-service solutions which their pharmaceutical clients could access through subscription agreements.
- Since 2024 Singapore biomedical research projects have increased their use of federated learning because this technology allows institutions to develop predictive models together while keeping their proprietary data secure.
Asia Pacific AI in Predictive Toxicology Market Segmentation
By Type :
The machine learning models for predictive toxicology use their ability to detect patterns in extensive biological and chemical databases. The models enable businesses to begin toxicity assessment through their early-stage screening process which requires less traditional testing. The system will achieve better precision through future structured data which will be used for its training and validation process.
Deep learning platforms AI software and data analytics tools improve prediction capabilities through their ability to process complex unstructured data. The tools enable researchers to analyze toxic effects from different compounds at an increased speed. The research and development process relies on additional supporting technologies which help improve results to achieve more trustworthy toxicity predictions.
By Application :
AI systems predict harmful reactions which drug safety testing uses before clinical trials begin. The process decreases risk while it speeds up product development. Chemical toxicity prediction enables early detection of dangerous substances which helps create safer products. The applications of these technologies enhance efficiency in safety assessment procedures that organizations use across various sectors.
AI tools assist environmental risk assessment by analyzing how pollutants affect ecosystems. Predictive systems detect harmful ingredients in food safety and cosmetics testing. The applications extend into various safety sectors which use early toxicity detection to improve decision making and regulatory compliance.
To learn more about this report, Download Free Sample Report
By End-User :
The pharmaceutical industry uses predictive toxicology tools to enhance their drug development processes by selecting better candidates and decreasing the rate of drug development failures. Biotech companies use these systems to accelerate their product development process while maintaining safety standards for their products. Research institutes depend on AI models to conduct both academic studies and experimental research that examines chemical and biological safety.
Government agencies use predictive toxicology to support public safety regulations and monitoring standards. CROs offer their clients outsourced testing services through the use of AI tools. Other end-users also adopt these systems to improve accuracy in toxicity analysis and reduce dependence on traditional laboratory methods.
By Deployment :
The cloud-based system enables users to access predictive toxicology tools while processing data in real time and collaborating with teams from multiple locations. The system provides users with scalable storage options and rapid computing capabilities that enable them to manage extensive datasets in toxicity prediction and analysis tasks.
Organizations that handle sensitive research data choose on-premise systems because these systems create secure environments. Hybrid deployment combines both cloud and on-premise models, allowing balanced performance, security, and accessibility. The predictive toxicology operations process needs this approach to establish efficient workflow management.
What are the Key Use Cases Driving the Asia Pacific AI in Predictive Toxicology Market?
The Asia Pacific AI in Predictive Toxicology market shows its primary application through drug discovery, which serves as the main use case. Pharmaceutical developers use predictive models to determine toxic compounds during preclinical screening, which helps them identify potential candidates faster while decreasing their expense of late-stage project failures. The application creates its greatest demand because it delivers direct benefits to R&D operations through improved research efficiency and shortened product development cycles.
Chemical safety assessment and cosmetic ingredient validation now use adjacent applications which have started to become popular. AI platforms enable industrial chemical manufacturers to achieve compliance with regional regulations which have become more demanding in Japan and South Korea, while cosmetic companies turn to computational screening for better alignment with alternative testing standards and lower their need for animal testing.
The medical field has developed personalized toxicology modeling systems, which help doctors create precise treatment plans. Singapore and China academic institutes and government-funded biomedical research centers conduct AI testing through patient genomic response analysis, while their initial platforms use predictive risk assessment methods to study complex biologics and advanced cell therapies.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 27.6 Million |
|
Market size value in 2026 |
USD 35.8 Million |
|
Revenue forecast in 2033 |
USD 188.5 Million |
|
Growth rate |
CAGR of 26.78% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
Asia Pacific (China, India, Japan, South Korea, Australia, Rest of Asia Pacific) |
|
Key company profiled |
IBM, Google, Microsoft, NVIDIA, Amazon, Schrödinger, Insilico Medicine, BenevolentAI, Exscientia, Certara, Dassault Systèmes, Oracle, SAP, DeepMind, Atomwise |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Machine Learning Models, Deep Learning Platforms, AI Software, Data Analytics Tools, Others); By Application (Drug Safety Testing, Chemical Toxicity Prediction, Environmental Risk Assessment, Food Safety, Cosmetics Testing, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Government Agencies, CROs, Others); By Deployment (Cloud, On-premise, Hybrid, Others) |
Which Regions are Driving the Asia Pacific AI in Predictive Toxicology Market Growth?
China establishes itself as the leading nation for AI development in Predictive Toxicology across the Asia Pacific region because it invests heavily in biotechnology while its government develops digital health systems and pharmaceutical industry upgrades. The development of computational toxicology tools received increasing approval from regulatory bodies which provided developers with better options to use AI-driven safety assessment systems in their research processes. The nation possesses an extensive network of artificial intelligence startups together with pharmaceutical production companies and genomic research organizations and cloud computing service providers. The platform enables users to train models at high speed while accessing extensive data resources which results in quicker time to market for predictive toxicology solutions compared to other areas within the region.
Japan holds the second-largest position, but its strength comes from consistency rather than scale-driven expansion. Japan relies on its established pharmaceutical research and development systems and its consistent regulatory operations and its business commitment to creating computational biology systems which have existed for extended periods. Major pharmaceutical companies continue upgrading toxicology assessment frameworks through measured digital transformation strategies. The company establishes a stable business model through its approach which generates ongoing revenue streams from software and service sales, making Japan an essential market player for the regional economic sector.
India has become the fastest-growing market because of its recent growth in contract research services and pharmaceutical outsourcing and AI-based biotechnology incubation programs. The implementation of digital health projects supported by the government together with heightened funding for life sciences research infrastructure has resulted in increased adoption since 2024. Companies from both domestic markets and international markets use AI toxicology tools to enhance their preclinical testing processes while fulfilling international regulatory requirements. The current market situation provides investors and new companies with various opportunities to establish platform partnerships and implement localized business models which will drive their commercial development until 2033.
Who are the Key Players in the Asia Pacific AI in Predictive Toxicology Market and How Do They Compete?
The Asia Pacific AI in Predictive Toxicology market shows a moderately fragmented competitive environment because the industry competes through advanced technology and superior data quality instead of using price as a competitive factor. Established computational biology software firms maintain their market positions through platform improvements and unified modeling functions while new AI-native companies use faster model development and unique toxicology systems to compete against them. The main competitive factor for companies lies in their ability to deliver accurate algorithms that provide understandable results for regulatory purposes while working with existing pharmaceutical research and development processes.
Partnerships between different regions become essential because organizations need local data access and knowledge about regional regulations to achieve successful implementation. Schrödinger uses physics-based simulation together with AI molecular modeling to provide pharmaceutical users enhanced predictive accuracy for their compound screening processes. The company achieves its main competitive edge through its ability to embed toxicology knowledge into its molecular design processes which enables faster progress between discovery teams. The company has developed its Asian presence through partnerships with local biotech companies and academic research centers. Certara establishes its market position through its regulatory-grade biosimulation platforms which match submission criteria, thus offering value to businesses that focus on compliance-based product development.
Insilico Medicine develops new artificial intelligence models which predict early toxic effects through its proprietary generative biology systems that enhance candidate testing efficiency. The company plans to grow by establishing partnerships with Asian pharmaceutical firms which need to accelerate their preclinical research processes. Exscientia uses automated processes to create its high-throughput design-to-validation system while BenevolentAI uses knowledge graphs to perform toxicology analysis which detects compound-risk relationships that standard predictive systems fail to identify.
Company List
- IBM
- Microsoft
- NVIDIA
- Amazon
- Schrödinger
- Insilico Medicine
- BenevolentAI
- Exscientia
- Certara
- Dassault Systèmes
- Oracle
- SAP
- DeepMind
- Atomwise
Recent Development News
In April 2026, DeepCyte launched its AI-driven single-cell toxicology platform backed by $1.5 million in seed funding. The company introduced tools designed to predict and explain drug toxicity at single-cell resolution, supporting more precise preclinical safety evaluation in drug discovery workflows across global markets, including Asia-Pacific biotech pipelines.
Source https://www.prnewswire.com/
In April 2026, Roche entered into a definitive agreement to acquire PathAI, an AI-powered pathology and diagnostics company. The acquisition strengthens Roche’s AI-driven predictive and diagnostic capabilities, accelerating biomarker discovery and clinical translation workflows that are increasingly used in toxicology-adjacent safety and drug development applications. Source https://www.roche.com/
What Strategic Insights Define the Future of the Asia Pacific AI in Predictive Toxicology Market?
The Asia Pacific AI in Predictive Toxicology market will develop complete computational safety systems which will connect different safety assessment components into unified systems for use in both pharmaceutical and chemical development processes during the upcoming five to seven years. The current structural transformation results from three technological factors which include new restrictions on non-animal testing, escalating expenses of drug development, and the development of multimodal AI systems which can process both genomic and chemical and biological information at the same time. The market will increasingly reward platforms that combine predictive accuracy with regulatory-grade explainability.
The market faces a concealed danger because it depends on a small group of proprietary toxicology dataset owners who control most of the existing datasets. Market concentration will limit access to validated regional datasets which will create financial obstacles for smaller developers who require these datasets to support their innovation efforts.
The federated toxicology modeling across Asia's biomedical research clusters offers significant potential for development according to researchers from Singapore and South Korea who work in secure cross-institution data collaboration frameworks. The best current market strategy requires participants to invest in explainable AI frameworks while developing partnerships for regional validation because these factors will determine regulatory trust which separates commercial platforms from technical solutions that cannot be monetized.
Asia Pacific AI in Predictive Toxicology Market Report Segmentation
By Type
- Machine Learning Models
- Deep Learning Platforms
- AI Software
- Data Analytics Tools
By Application
- Drug Safety Testing
- Chemical Toxicity Prediction
- Environmental Risk Assessment
- Food Safety
- Cosmetics Testing
By End-User
- Pharma Companies
- Biotech Firms
- Research Institutes
- Government Agencies
- CROs
By Deployment
- Cloud
- On-premise
- Hybrid
Frequently Asked Questions
Find quick answers to common questions.
The Asia Pacific AI in Predictive Toxicology Market size is USD 188.5 Million in 2033.
Key segments for the Asia Pacific AI in Predictive Toxicology Market are By Type (Machine Learning Models, Deep Learning Platforms, AI Software, Data Analytics Tools, Others); By Application (Drug Safety Testing, Chemical Toxicity Prediction, Environmental Risk Assessment, Food Safety, Cosmetics Testing, Others); By End-User (Pharma Companies, Biotech Firms, Research Institutes, Government Agencies, CROs, Others); By Deployment (Cloud, On-premise, Hybrid, Others).
Major Asia Pacific AI in Predictive Toxicology Market players are IBM, Google, Microsoft, NVIDIA, Amazon, Schrödinger, Insilico Medicine, BenevolentAI, Exscientia, Certara, Dassault Systèmes, Oracle, SAP, DeepMind, Atomwise.
The Asia Pacific AI in Predictive Toxicology Market size is USD 27.6 Million in 2025.
The Asia Pacific AI in Predictive Toxicology Market CAGR is 26.78% from 2026 to 2033.
- IBM
- Microsoft
- NVIDIA
- Amazon
- Schrödinger
- Insilico Medicine
- BenevolentAI
- Exscientia
- Certara
- Dassault Systèmes
- Oracle
- SAP
- DeepMind
- Atomwise
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033