North America Cardiac Pacemaker Market Size & Forecast:
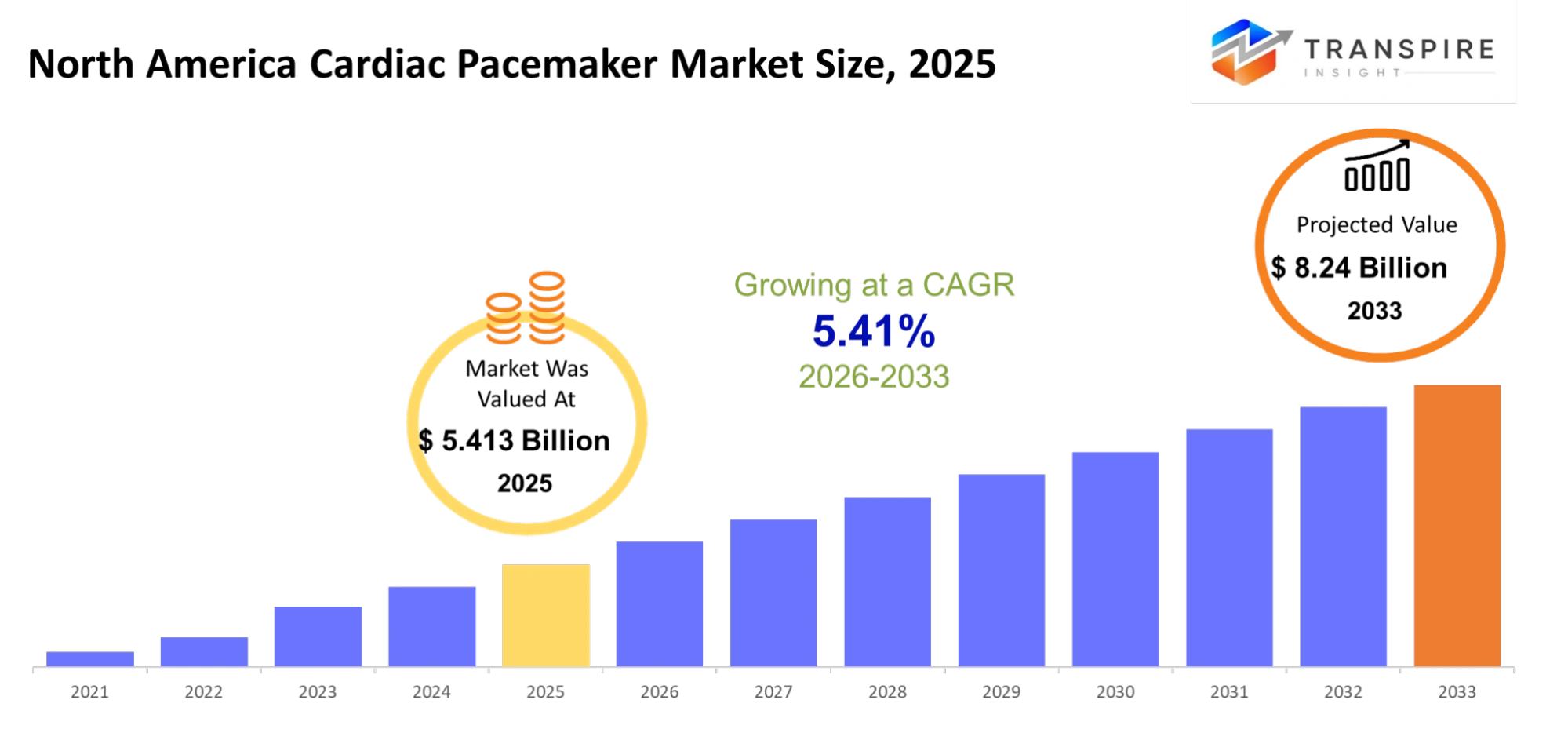
- North America Cardiac Pacemaker Market Size 2025: USD 5.413 Billion
- North America Cardiac Pacemaker Market Size 2033: USD 8.24 Billion
- North America Cardiac Pacemaker Market CAGR: 5.41%
- North America Cardiac Pacemaker Market Segments: By Product Type (Implantable Pacemakers, External Pacemakers), By Application (Arrhythmia, Heart Failure), By End-User (Hospitals, Cardiac Centers).
To learn more about this report, Download Free Sample Report
North America Cardiac Pacemaker Market Summary:
The North America Cardiac Pacemaker Market size is estimated at USD 5.413 Billion in 2025 and is anticipated to reach USD 8.24 Billion by 2033, growing at a CAGR of 5.41% from 2026 to 2033.
The cardiac pacemaker market in North America, which includes Canada, the United States and Mexico operates in the advanced medical device market, which will be shaped by precision engineering and patient-centered innovation. The growing need for medical equipment will stem from the increasing number of elderly people seeking treatments that require less invasive procedures and medical devices that last longer. Manufacturers will use AI to create diagnostic systems that enable remote patient monitoring while extending battery life, providing patients with uninterrupted care. The regulatory authorities will establish more stringent safety and cybersecurity requirements which will determine both product development processes and the duration of product approval procedures. Hospitals and outpatient centers will favor compact, wireless systems that reduce recovery time, while patients will expect seamless digital connectivity, which will determine the development and delivery methods for future pacing technologies.
Key Market Trends & Insights:
People will choose medical devices based on their growing understanding of treatment options because they want to use pacemakers which do not show and support their active lifestyles. The market will start to prefer leadless systems, which provide both better cosmetic results and fewer required follow-up appointments. Doctors will start using devices that enable patients to receive remote monitoring, as this system allows them to make immediate treatment adjustments while staying at home.
Device manufacturers need to enhance both battery life and product intelligence, as technological advancements are driving this requirement. Future pacemakers will use adaptive algorithms which deliver personalized cardiac support by responding to physiological changes in real-time. Digital health systems will connect with health systems through an expanded integration which enables patients to share their data with care providers, thus improving treatment accuracy and long-term health results.
The region's healthcare systems will adopt outpatient and ambulatory care models, which will create new requirements for developing and distributing medical devices. Hospitals will establish compact pacemakers as their standard device because these solutions help them decrease both operational expenses and time needed for medical treatments. The new system will help small clinics implement modern cardiac treatment methods by making advanced technologies more accessible.
The development of medical device regulatory systems will respond to the security needs associated with connected medical devices and the protection of sensitive data. Companies will need to adopt stricter security measures because authorities will establish new compliance requirements which will require them to spend resources on developing secure software and firmware updates. The system changes will build patient confidence because connected pacemakers will function securely within medical systems that increasingly use digital technology.
North America Cardiac Pacemaker Market Segmentation
By Product Type
Implantable Pacemakers- The medical community will select implantable pacemakers as their primary option because both doctors and patients value extended device durability which causes less interruption to everyday activities. The product will become more attractive because of its improved battery performance and development of leadless technology. External pacemakers will maintain their essential function in providing temporary heart support which occurs during emergency situations and when patients require stabilization after surgery inside medical facilities.
External Pacemakers- The development of external systems will include better systems which allow medical personnel to move equipment more easily while setting up their systems during emergency situations. Healthcare providers will increasingly rely on these devices for short-term rhythm management, particularly in intensive care units. The continuous research and development process will lead to new implantable devices which will deliver better detection capabilities while making operations simpler to perform.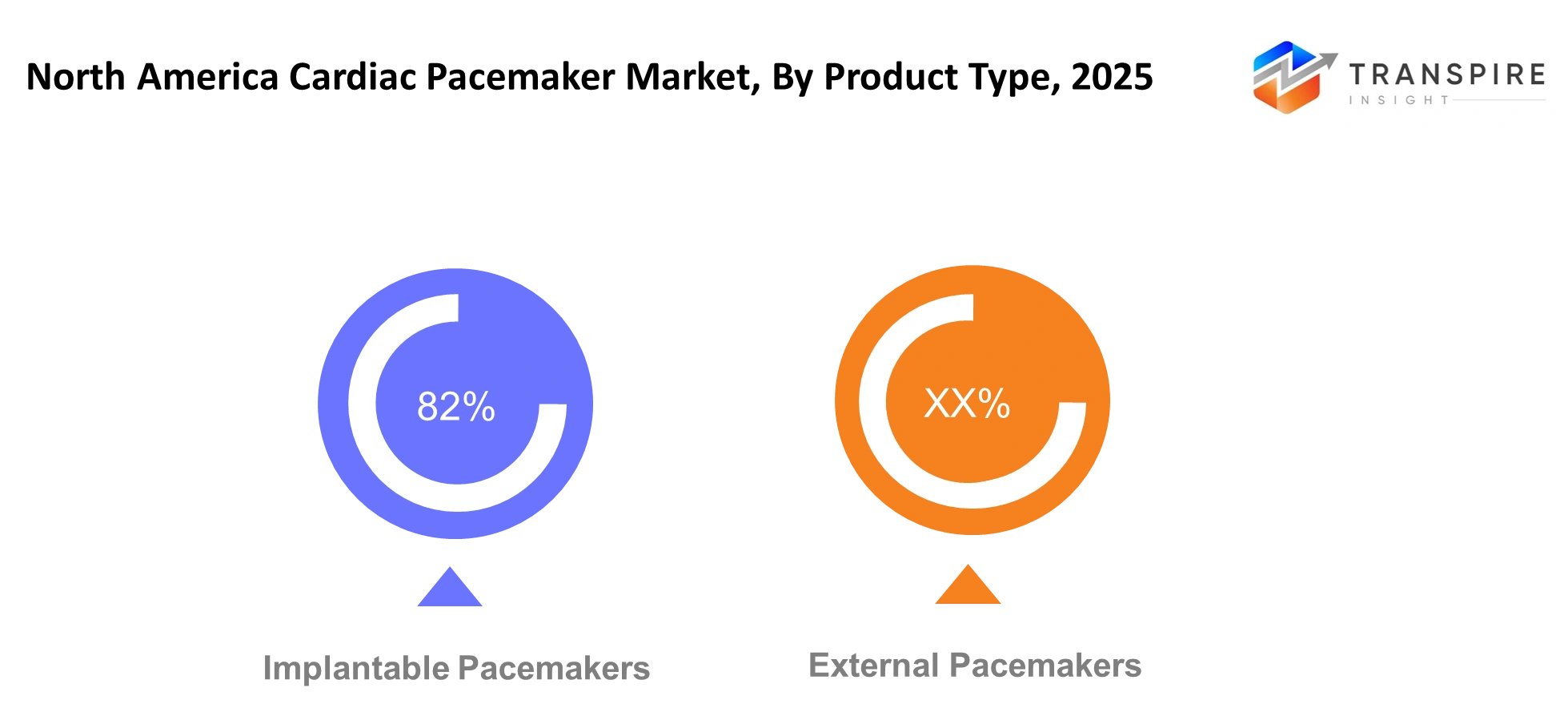
To learn more about this report, Download Free Sample Report
By Application
Arrhythmia- More healthcare facilities will use arrhythmia management systems because of their improved ability to detect abnormalities which enables timely treatment. Patients will seek timely intervention options that prevent complications and support normal daily activity. The devices will enable automatic adjustment to heart rhythm changes which will result in better rhythm control and decreased requirement for medical evaluations.
Heart Failure- Heart failure applications will gain attention as integrated pacing solutions evolve to support complex cardiac conditions. Devices will increasingly combine pacing with advanced therapeutic functions, improving coordination between heart chambers. The system will improve patient results while decreasing hospital readmissions for elderly patients who need ongoing heart treatment that adjusts to their needs.
By End-User
Hospitals- The advanced infrastructure of hospitals together with their specialized cardiac teams will enable hospitals to become the primary adopters of new technologies. The medical staff will conduct the implantation operations while they also provide complete postoperative treatment which guarantees successful outcomes. The organization will enhance its ability to deliver full pacemaker-based treatments through digital health integration throughout its various patient categories.
Cardiac Centers- Cardiac centers will expand their influence by focusing on specialized, high-volume procedures and personalized care pathways. The facilities will focus on achieving operational efficiency through implementation of quicker recovery processes and development of customized treatment schedules. The growth of outpatient care will transform cardiac centers into essential centers for both implantation procedures and ongoing patient observation which will enable easier access to cardiac care.
Regional Insights
The United States will drive regional technology implementation because of its advanced medical facilities and its early adoption of next-generation pacing solutions. Patients will increasingly demand devices that work with digital health systems to provide ongoing medical evaluation. The reimbursement systems will support hospitals that provide minimally invasive treatments by creating financial incentives to use leadless pacemakers which deliver faster recovery times and better patient results.
Canada will maintain a steady pace because its healthcare system receives funding from the government and it focuses on developing affordable medical solutions. Hospitals will select medical equipment that provides both extended operational capabilities and cost-effective solutions. Medical professionals will use remote patient monitoring to supervise patients who live in both rural and urban spaces because it enables them to effectively treat heart diseases while decreasing the demand for specialized medical centers.
The healthcare system in Mexico will experience slow development because of improved healthcare access and increasing public knowledge about heart disease. Urban areas will see private hospitals take the lead in implementing modern pacemaker systems. The development of medical facilities will create more treatment centers but patients will increasingly prefer advanced medical options because they provide better safety and lower treatment risks.
Cross-border partnerships and regulatory standards between North American countries will determine how markets will develop. Organizations will keep updating their security protocols and data protection methods for wireless devices. Healthcare organizations will create standardized methods for watching patients and handling medical devices which will result in uniform treatment standards. Clinical environments will adopt next-generation pacemaker technologies through this regional alignment, helping establish a more compatible framework.
Recent Development News
Advancements in Leadless and Smart Pacemaker Systems- Recent developments in the United States will highlight a clear shift toward leadless and intelligent pacing solutions. The introduction of dual-chamber leadless devices by companies enables patients to experience better safety and comfort because the devices eliminate wires and decrease surgical procedures. The adaptive algorithms and remote monitoring capabilities of new systems will enable physicians to continuously monitor cardiac performance while delivering customized therapy solutions.
Expansion of Remote Monitoring and Digital Cardiac Care- Canada will focus on strengthening remote cardiac care through digital integration. Healthcare providers will increasingly adopt pacemakers that connect with monitoring platforms which reduce the need for in-person follow-ups. The shift will improve access to remote areas that experience travel difficulties while providing continuous, data-driven patient monitoring for better disease management outcomes.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 5.413 Billion |
|
Market size value in 2026 |
USD 5.7 Billion |
|
Revenue forecast in 2033 |
USD 8.24 Billion |
|
Growth rate |
CAGR of 5.41% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Regional scope |
North America (Canada, The United States, and Mexico) |
|
Key company profiled |
Medtronic plc, Abbott Laboratories, Boston Scientific Corporation, Biotronik SE & Co. KG, LivaNova PLC, MicroPort Scientific Corporation, Lepu Medical Technology Co. Ltd., Osypka Medical GmbH, Shree Pacetronix Ltd., Medico S.p.A., Pacetronix India Pvt. Ltd., Oscor Inc., ZOLL Medical Corporation, Edwards Lifesciences Corporation, Siemens Healthineers AG. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Product Type (Implantable Pacemakers, External Pacemakers), By Application (Arrhythmia, Heart Failure), By End-User (Hospitals, Cardiac Centers). |
Key North America Cardiac Pacemaker Market Company Insights
The cardiac pacemakers market in North America, which includes Canada the US and Mexico will be impacted by the competitive activities of these companies. Medtronic Abbott, Boston Scientific and BIOTRONIK will operate as the primary market leaders in this sector. The companies would aim to expand their market presence by developing leadless pacemakers and smart pacemaker monitoring systems and forming strategic partnerships.
Company List
- Medtronic plc
- Abbott Laboratories
- Boston Scientific Corporation
- Biotronik SE & Co. KG
- LivaNova PLC
- MicroPort Scientific Corporation
- Lepu Medical Technology Co. Ltd.
- Osypka Medical GmbH
- Shree Pacetronix Ltd.
- Medico S.p.A.
- Pacetronix India Pvt. Ltd.
- Oscor Inc.
- ZOLL Medical Corporation
- Edwards Lifesciences Corporation
- Siemens Healthineers AG.
North America Cardiac Pacemaker Market Report Segmentation
By Product Type
- Implantable Pacemakers
- External Pacemakers
By Application
- Arrhythmia
- Heart Failure
By End-User
- Hospitals
- Cardiac Centers
Frequently Asked Questions
Find quick answers to common questions.
The Approximate North America Cardiac Pacemaker Market size for the Market will be USD 8.24 Billion in 2033.
Key segments for the North America Cardiac Pacemaker Market By Product Type (Implantable Pacemakers, External Pacemakers), By Application (Arrhythmia, Heart Failure), By End-User (Hospitals, Cardiac Centers).
Major Players in the North America Cardiac Pacemaker Market are Medtronic plc, Abbott Laboratories, Boston Scientific Corporation, Biotronik SE & Co. KG, LivaNova PLC, MicroPort Scientific Corporation, Lepu Medical Technology Co. Ltd., Osypka Medical GmbH, Shree Pacetronix Ltd., Medico S.p.A., Pacetronix India Pvt. Ltd., Oscor Inc., ZOLL Medical Corporation, Edwards Lifesciences Corporation, Siemens Healthineers AG.
The North America Cardiac Pacemaker Market size is USD 5.413 Billion in 2025.
The North America Cardiac Pacemaker Market CAGR is 5.41%.
- Medtronic plc
- Abbott Laboratories
- Boston Scientific Corporation
- Biotronik SE & Co. KG
- LivaNova PLC
- MicroPort Scientific Corporation
- Lepu Medical Technology Co. Ltd.
- Osypka Medical GmbH
- Shree Pacetronix Ltd.
- Medico S.p.A.
- Pacetronix India Pvt. Ltd.
- Oscor Inc.
- ZOLL Medical Corporation
- Edwards Lifesciences Corporation
- Siemens Healthineers AG
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033


