Japan Rhabdoid Oncology Market Size & Forecast:
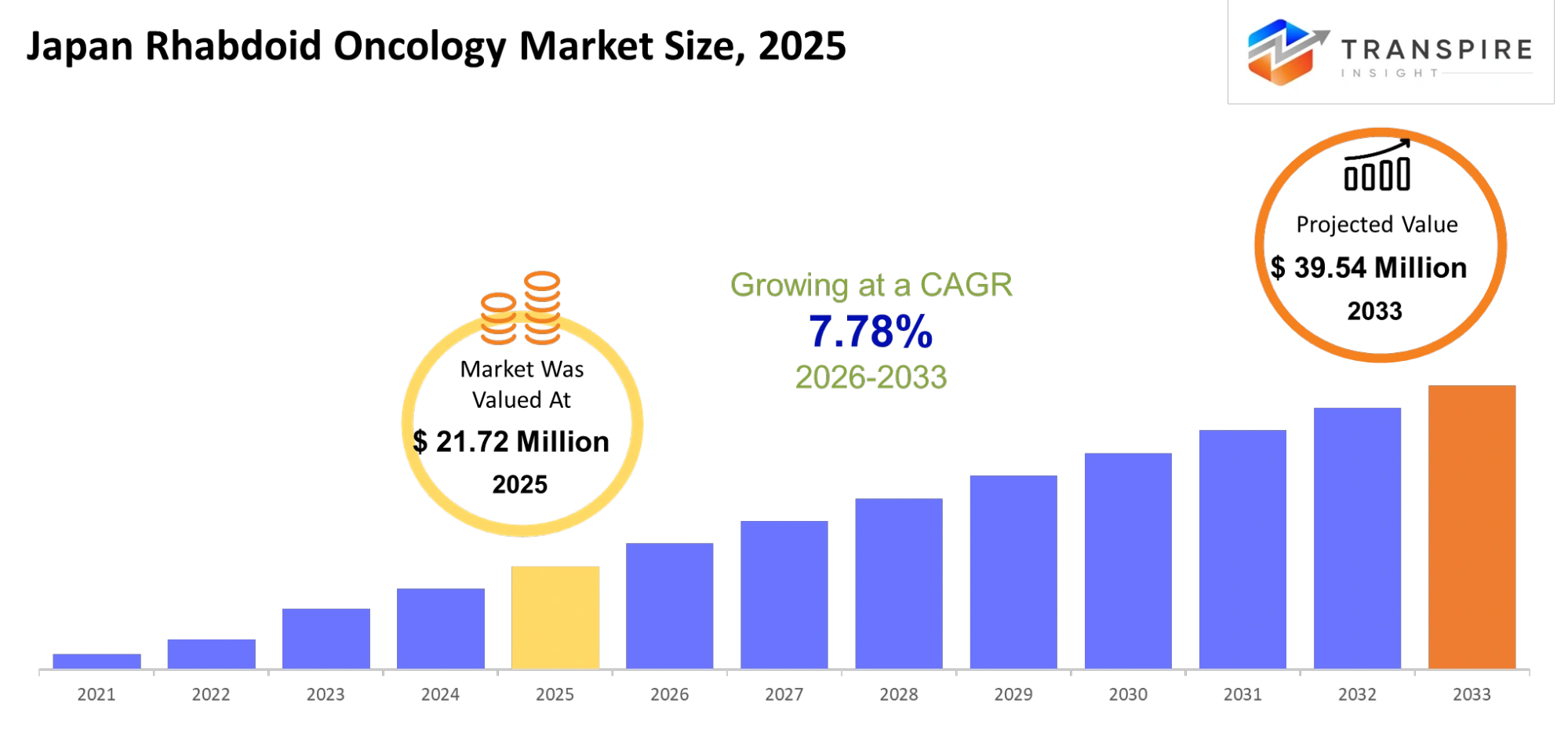
- Japan Rhabdoid Oncology Market Size 2025: USD 21.72 Million
- Japan Rhabdoid Oncology Market Size 2033: USD 39.54 Million
- Japan Rhabdoid Oncology Market CAGR: 7.78%
- Japan Rhabdoid Oncology Market Segments: By Type (Chemotherapy, Targeted Therapy, Immunotherapy, Combination Therapy, Precision Medicine, Others); By Application (Cancer Treatment, Pediatric Oncology, Rare Tumor Treatment, Clinical Trials, Oncology Research, Others); By End-User (Hospitals, Cancer Centers, Clinics, Research Institutes, Specialty Oncology Centers, Academic Institutions, Others); By Distribution (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Specialty Pharmacies, Others)
To learn more about this report, Download Free Sample Report
Japan Rhabdoid Oncology Market Summary
The Japan Rhabdoid Oncology Market was valued at USD 21.72 Million in 2025. It is forecast to reach USD 39.54 Million by 2033. That is a CAGR of 7.78% over the period.
The Japanese market for rhabdoid oncology research identifies and treats rare tumors which display extreme aggressive behavior toward patients who suffer from malignant rhabdoid tumors and INI1-deficient cancers particularly in children and young adults. The system enables hospitals to perform molecular tests for diagnostic purposes while selecting treatment options and providing patients with access to specialized cancer treatments. The market underwent a fundamental transformation during the last five years because it shifted from traditional histopathology methods and general chemotherapy approaches to precise medical techniques which use genomic data and next-generation sequencing methods.
The first permanent digital pathology system in Japanese hospitals became operational after the nationwide implementation of Japan's cancer genomic medicine system and hospitals received increased funding for complete genomic analysis. The process of identifying patients with specific needs has become more precise which helps more people join targeted treatment programs while pharmaceutical companies invest in developing medications for uncommon tumors and hospitals build their diagnostic capabilities.
Key Market Insights
- The Japan Rhabdoid Oncology Market is shifting toward precision oncology because genomic sequencing has become standard for diagnosing and treating rare pediatric tumors.
- The Kanto region controls more than 35% of the Japan Rhabdoid Oncology Market because Tokyo contains most of the leading cancer research hospitals and biotechnology centers.
- The Kansai region shows the fastest growth rate because oncology research partnerships are increasing and clinical trials will expand from 2024 to 2030.
- The Japan Rhabdoid Oncology Market is dominated by molecular diagnostics which holds almost 40% market share because next-generation sequencing technologies are in high demand.
- The second largest market segment consists of therapeutic oncology drugs which receive support from both orphan drug approvals and targeted treatment development programs.
- The fastest expanding medical field currently shows clinical genomics services as its most rapidly growing segment, which will continue until 2030 because of expanding precision medicine programs.
- The Japan Rhabdoid Oncology Market shows pediatric rhabdoid tumor treatment as its most important treatment method because this disease type has extremely severe symptoms.
- The hospital-based genomic research programs and clinical trial expansion drive the most rapid growth of rare cancer research applications.
- The major treatment and diagnostic centers for cancer research exist in hospitals and specialized cancer treatment facilities, which together control more than 50% of the market.
- The end-user category of research institutes experiences rapid growth because funding for rare oncology studies has increased.
What are the Key Drivers, Restraints, and Opportunities in the Japan Rhabdoid Oncology Market?
Next-generation sequencing technology now enables hospitals to diagnose rare cancers through its implementation in their medical facilities. This transformation occurred after sequencing costs decreased and Japan established its precision medicine framework through cancer genomics research programs. Clinicians now use this method to detect rhabdoid tumor mutations which enables them to select suitable therapies and clinical trials for their patients with greater efficiency. The process has resulted in better treatment start times at tertiary hospitals which leads to more resources spent on molecular diagnostics and rare oncology medications thus boosting revenue for both testing and treatment operations.
The research process faces its main challenge because researchers need to treat complex patients who make up a very small group, which prevents them from conducting extensive clinical studies and brings delays to the development of new drugs. Rhabdoid tumors remain rare and often pediatric, making data collection slow and statistically constrained. The approval process for new treatments in Japan takes extended periods because of regulatory validation requirements. The pharmaceutical industry faces two major challenges because reimbursement processes take too long and pharmaceutical firms cannot predict their financial returns. These problems stop companies from entering the market quickly while their scientific research progresses rapidly.
AI-assisted digital pathology presents a major opportunity for hospital networks based in Tokyo and Osaka. This technology enables faster tumor classification using imaging and genomic correlation models, reducing diagnostic turnaround time. Major university hospitals are allocating funds toward developing AI-powered rare cancer registries which will connect to real-world evidence databases. The existing infrastructure will enable researchers to build scalable clinical trial matching systems which will help them develop precise medication solutions at a faster pace.
What Has the Impact of Artificial Intelligence Been on the Japan Rhabdoid Oncology Market?
The combination of artificial intelligence with advanced digital technologies is transforming the clinical and research operations of Japan's rare rhabdoid oncology system. This transformation enables faster tumor detection and classification while providing treatment path matching. The implementation of AI-powered digital pathology systems at hospitals allows staff to perform automatic slide analysis which decreases their diagnostic duties while enabling better detection of rhabdoid tumor indicators. The automation system allows oncology centers with high patient volumes to process cases more efficiently according to their operational needs which operate mainly in Tokyo and Osaka because they experience high demand for rare cancer cases.
Machine learning predictive models assess tumor progression risk while creating patient response forecasts for targeted therapies which use genomic and clinical datasets. These tools enable clinicians to determine which treatments should occur at which points in time while they help improve study participant selection methods which remain vital for treating ultra-rare cancers that have extremely limited patient populations. The hospitals experience operational improvements because their diagnostic processes become faster while their resource management systems achieve better efficiency which results in more oncology services being delivered at a quicker rate and less time needed before starting treatments.
The process of adoption faces obstacles because hospitals lack access to sufficient high-quality training datasets which they need to study the extremely rare rhabdoid tumors that occur in Japan. The lack of data limits model performance while research progress towards clinical validation faces slowdowns. The expensive costs required to integrate AI systems with existing hospital IT systems prevent hospitals from implementing technology outside their research facilities.
Key Market Trends
- The adoption of genomic sequencing started to rise dramatically after 2022 which resulted in a shift from histology-based diagnosis methods toward mutation-based classification procedures for diagnosing rhabdoid tumors.
- Digital pathology systems expanded across Japanese cancer hospitals which resulted in shorter manual slide review times and better diagnostic results for uncommon cancer cases.
- The introduction of AI-based patient matching systems at hospitals which combined genomic data with national cancer databases led to better recruitment results for clinical trials after 2023.
- The development of orphan drugs gained speed as Takeda and other companies increased their funding for oncology treatments that focused on rhabdoid tumor mutations in rare pediatric diseases.
- The Japanese regulatory system introduced new pathways that enabled conditional drug approval processes after 2021 which permitted patients to access experimental treatments through ongoing real-world evidence research.
- The hospital networks began to use centralized rare cancer data systems which allowed better cooperation between Tokyo University Hospital and nearby oncology research facilities.
- The use of companion diagnostics became more popular because it helped doctors choose targeted therapies instead of using chemotherapy for aggressive rhabdoid cases.
- The implementation of AI technology into pathology operations at Japanese cancer treatment centers led to a 30 to 40 percent decrease in time needed for diagnostic results.
- The pharmaceutical industry moved its collaborative research model from conducting independent research to creating joint research initiatives with academic institutions that aimed to fast-track treatments for ultra-rare cancers.
Japan Rhabdoid Oncology Market Segmentation
By Type:
Japan uses chemotherapy as its main treatment method for rhabdoid oncological cases because doctors have used it successfully for many years to treat severe pediatric cases and uncommon tumors. Hospitals depend on existing multi-drug chemotherapy protocols because they lack effective targeted and immunotherapy treatments for certain rhabdoid cancer subtypes. Doctors continue to use chemotherapy despite its toxic effects because they believe it will help patients survive their immediate health crisis.
Genomic profiling has become standard practice in tertiary oncology centers which has led to faster growth in targeted therapy and precision medicine segments. Drug developers at Novartis now develop therapies for specific mutations which decreases their need for traditional cytotoxic treatment methods. The forecasting period will see precision medicine grow at a fast pace which will create new personalized treatment methods and increase the need for companion diagnostics and biomarker-based drug development.

To learn more about this report, Download Free Sample Report
By Application:
Pediatric oncology remains the most critical application area due to the high incidence of rhabdoid tumors in infants and young children in Japan. The combination of urgent treatment needs and the shortage of available treatment options leads hospitals to implement established pediatric oncology treatment protocols. The establishment of dedicated pediatric cancer treatment centers has become essential for both patient care and medical research activities.
The development of genomic datasets and the growth of national cancer registries are driving rapid expansion of both rare tumor treatments and oncology research initiatives. University hospitals are experiencing increased clinical trial activity because AI-based patient matching systems lead to better trial enrollment results in Tokyo and Osaka. Research-driven applications will develop drug innovation pipelines which will speed up the process of turning experimental therapies into approved treatments.
By End-User:
The Japanese rhabdoid oncology treatment system operates through hospitals and specialized cancer centers which possess advanced diagnostic equipment and critical care facilities and medical professionals who specialize in oncology. The institutions handle most of their diagnosed cases because metropolitan areas contain their primary referral systems. The healthcare system maintains centralized treatment methods which receive government support to deliver cancer treatments thus establishing their authority over cancer treatment operations.
Research institutes and academic institutions are emerging as high-growth end-users as they expand rare cancer genomic studies and clinical trial collaborations. University hospitals increasingly form partnerships with pharmaceutical companies to accelerate the development of new therapies. The integrated data platforms will experience higher demand during the forecast period because academic-medical partnerships will drive new treatment developments.
By Distribution:
The primary distribution channel for rhabdoid tumor treatment exists through hospital pharmacies because the treatment requires patients to receive chemotherapy and specialized oncology drugs during their hospital stay. The primary location for medication distribution in tertiary care hospitals exists because the facility needs to maintain complete control over medication distribution and ensure proper dosage administration. The system restricts possibilities to use external retail distribution systems.
The importance of specialty pharmacies has been increasing because targeted therapies and precision medicine drugs need specific handling and patient-focused distribution solutions. Regulations restrict retail and online pharmacies from distributing high-risk oncology drugs which results in operational limitations for these businesses. The future distribution system will establish greater control through hospital-based pharmacy networks while specialty systems will provide support for advanced biologics and customized treatment programs.
What are the Key Use Cases Driving the Japan Rhabdoid Oncology Market?
The Japan Rhabdoid Oncology Market experiences its main demand through pediatric rhabdoid tumor treatment which needs hospitals to use intensive chemotherapy together with early genomic diagnosis for their treatment of fast-developing cases. The use case remains the most important one because cancer centers need to start treatment right away while they keep observing patients throughout their treatment process.
The university hospitals and oncology research institutes now use genomic profiling to match patients with clinical trials which leads to better access to experimental medicine and more customized treatment for patients at tertiary care facilities.
The new applications now include AI-based pathology systems which automatically classify tumors together with national rare cancer registries that operate across multiple countries. The applications are currently restricted to top academic medical centers in Tokyo and Osaka but their availability will increase throughout the forecast period as digital infrastructure and precision medicine systems continue to develop.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 21.72 Million |
|
Market size value in 2026 |
USD 23.41 Million |
|
Revenue forecast in 2033 |
USD 39.54 Million |
|
Growth rate |
CAGR of 7.78% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 - 2024 |
|
Forecast period |
2026 - 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Japan |
|
Key company profiled |
Roche, Novartis, Pfizer, Bristol Myers Squibb, Merck, AstraZeneca, GSK, Sanofi, Takeda, AbbVie, Amgen, Eli Lilly, Johnson & Johnson, Bayer, Teva |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Type (Chemotherapy, Targeted Therapy, Immunotherapy, Combination Therapy, Precision Medicine, Others); By Application (Cancer Treatment, Pediatric Oncology, Rare Tumor Treatment, Clinical Trials, Oncology Research, Others); By End-User (Hospitals, Cancer Centers, Clinics, Research Institutes, Specialty Oncology Centers, Academic Institutions, Others); By Distribution (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Specialty Pharmacies, Others) |
Which Regions are Driving the Japan Rhabdoid Oncology Market Growth?
The Japan Rhabdoid Oncology Market in the Kanto region operates as its leading market because the region has multiple national cancer institutes and advanced pediatric oncology hospitals and government-backed precision medicine programs. Tokyo functions as the main center for rare tumor diagnosis and genomic sequencing because of its combined hospital system and advanced laboratory facilities. National health funding programs establish continuous financial support which enables healthcare providers to implement early diagnostic methods while boosting their clinical research efforts. The system enables fast conversion of molecular discoveries into therapeutic solutions which confirms Kanto's ongoing leadership status.
The Kansai region operates as a stable secondary market because Osaka and Kyoto maintain their existing university hospital systems which produce ongoing oncology research. The academic-medical system in Kansai enables research to progress at a steady pace through its existing distributed network structure while Kanto relies on its centralized research approach for progress. The pharmaceutical partnership between Takeda and regional hospitals creates a research framework that enables ongoing rare cancer studies in the area. The stable research environment of Kansai enables healthcare providers to solve treatment challenges while participating in clinical trials.
The Chubu region has become the fastest-growing area because Nagoya established advanced cancer care centers and dedicated more resources to molecular diagnostics facilities. The new hospital partnerships with national research programs have provided better access to genomic testing for rare tumors. The government-backed healthcare modernization programs which began in 2023 led to a faster implementation of digital pathology systems. The market growth will create more possibilities for new companies which develop diagnostic technologies and precision oncology solutions between now and 2033.
Who are the Key Players in the Japan Rhabdoid Oncology Market and How Do They Compete?
The Japan rhabdoid oncology market shows moderate consolidation because global oncology specialists and rare disease experts combat each other through precise diagnostic tests and specific treatment methods instead of competing on their product prices. Incumbents protect their market share by establishing partnerships with hospitals and running their early access programs across Japanese university hospitals. The competition directly competes through its ability to conduct genomic profiling and implement companion diagnostic systems while accelerating clinical trial enrollment processes for pediatric rare tumors which affect restricted patient groups because scientific advancement needs to be completed rapidly.
Roche uses its diagnostic testing and treatment solution which connects genomic testing platforms to targeted oncology drugs for faster treatment identification in Japanese tertiary hospitals. Novartis develops precision oncology products while creating new clinical trial networks that enable early-phase studies throughout Tokyo. Takeda uses its domestic market advantage to establish strong partnerships with hospitals while developing its pediatric oncology programs according to Japan’s orphan drug regulatory system which helps patients access treatments more quickly and improves regulatory collaboration.
Pfizer expands its operations through two immunotherapy pipelines which it developed through partnerships with academic institutions studying rare tumors. Bayer uses its regional clinical partnerships in Japan to develop new biomarker-based products for market introduction. The company uses its immuno-oncology knowledge and its combination treatment studies for harsh cancer types to enhance its market position. The companies create more intense market competition by establishing partnerships and conducting clinical research which enables them to complete trials more quickly while building complete precision medicine systems.
Company List
- Roche
- Novartis
- Pfizer
- Bristol Myers Squibb
- Merck
- AstraZeneca
- GSK
- Sanofi
- Takeda
- AbbVie
- Amgen
- Eli Lilly
- Johnson & Johnson
- Bayer
- Teva
Recent Development News
In April 2026, Nuvation Bio announced acquisition of Japan rights to safusidenib from Daiichi Sankyo. The agreement granted Nuvation Bio full global development and commercialization control of the investigational oncology therapy, enabling expansion of clinical programs into Japan, including studies targeting IDH1-mutant glioma. This strengthens Japan’s access to next-generation precision oncology pipelines relevant to rare CNS tumors such as rhabdoid-related malignancies. Sourcehttps://investors.nuvationbio.com/
In November 2025, NEXT Oncology entered a joint venture partnership with Kansai Medical University to expand Phase I oncology clinical trial capabilities in Japan. The collaboration established a global early-phase drug development platform in Osaka, increasing Japan’s capacity to enroll patients in first-in-human cancer trials, including rare and aggressive CNS tumor categories such as atypical teratoid/rhabdoid tumors.Source https://nextoncology.com/
What Strategic Insights Define the Future of the Japan Rhabdoid Oncology Market?
The Japan Rhabdoid Oncology Market will move toward complete integration of precision oncology systems during the next five to seven years because genomic diagnostics and AI-assisted pathology and targeted therapeutics will function as a single treatment system. National rare cancer initiatives drive this shift while hospitals increasingly adopt molecular-first treatment protocols which use mutation-level decision-making as their primary decision method instead of traditional chemotherapy. The platform will generate value through its diagnostic and therapy optimization combination which will create better results than traditional drug development approaches.
The Japanese tertiary hospitals depend too much on centralized genomic datasets which creates a hidden risk that will restrict model application across different settings while making it harder to implement solutions throughout the country. The system becomes vulnerable to disruptions because data centers depend on specific interoperability standards which might be affected by funding changes.
The development of AI-linked rare cancer registries at regional centers such as Chubu represents a fresh market opportunity because digital pathology implementation in that area is increasing. The datasets will play a vital role in advancing orphan drug trials and producing real-world evidence. Market participants should focus on building partnerships with hospital networks because this approach will enable them to obtain genomic data access at an early stage while establishing their presence in the complete diagnostic-to-therapy system.
Japan Rhabdoid Oncology Market Report Segmentation
By Type
- Chemotherapy
- Targeted Therapy
- Immunotherapy
- Combination Therapy
- Precision Medicine
By Application
- Cancer Treatment
- Pediatric Oncology
- Rare Tumor Treatment
- Clinical Trials
- Oncology Research
By End-User
- Hospitals
- Cancer Centers
- Clinics
- Research Institutes
- Specialty Oncology Centers
- Academic Institutions
By Distribution
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Specialty Pharmacies
Frequently Asked Questions
Find quick answers to common questions.
The Japan Rhabdoid Oncology Market size is USD 39.54 Million in 2033.
Key segments for the Japan Rhabdoid Oncology Market are By Type (Chemotherapy, Targeted Therapy, Immunotherapy, Combination Therapy, Precision Medicine, Others); By Application (Cancer Treatment, Pediatric Oncology, Rare Tumor Treatment, Clinical Trials, Oncology Research, Others); By End-User (Hospitals, Cancer Centers, Clinics, Research Institutes, Specialty Oncology Centers, Academic Institutions, Others); By Distribution (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies, Specialty Pharmacies, Others).
Major Japan Facioscapulohumeral Muscular Dystrophy Market players are Roche, Novartis, Pfizer, Bristol Myers Squibb, Merck, AstraZeneca, GSK, Sanofi, Takeda, AbbVie, Amgen, Eli Lilly, Johnson & Johnson, Bayer, Teva.
The Japan Rhabdoid Oncology Market size is USD 21.72 Million in 2025.
The Japan Rhabdoid Oncology Market CAGR is 7.78% from 2026 to 2033.
- Roche
- Novartis
- Pfizer
- Bristol Myers Squibb
- Merck
- AstraZeneca
- GSK
- Sanofi
- Takeda
- AbbVie
- Amgen
- Eli Lilly
- Johnson & Johnson
- Bayer
- Teva
Recently Published Reports
-
Apr 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033