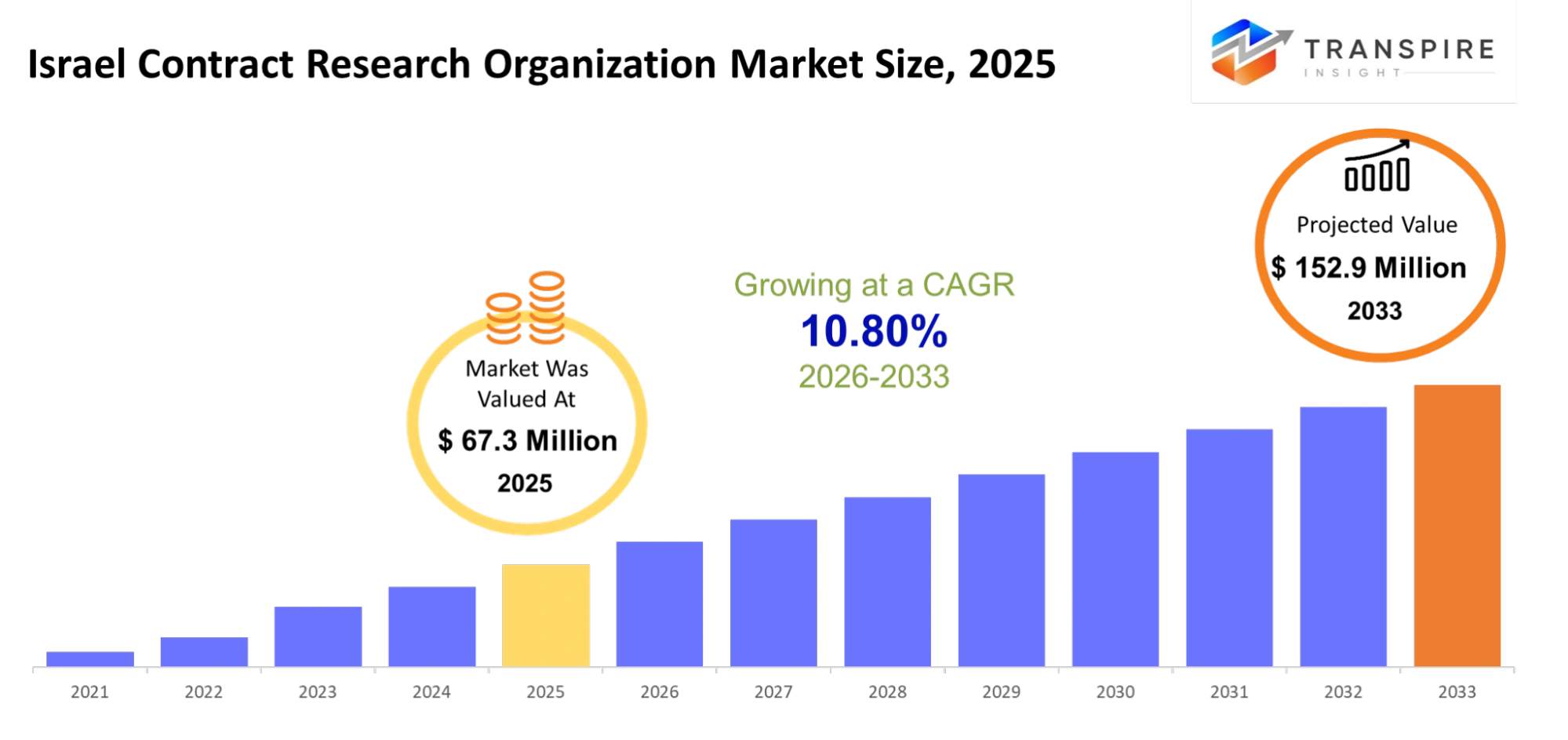
Israel Contract Research Organization Market Size & Forecast:
- Israel Contract Research Organization Market Size 2025: USD 67.3 Million
- Israel Contract Research Organization Market Size 2033: USD 152.9 Million
- Israel Contract Research Organization Market CAGR: 10.80%
- Israel Contract Research Organization Market Segments: By Service Type (Clinical Research Services, Preclinical Services, Laboratory Services), By Therapeutic Area (Oncology, Cardiovascular Diseases, Infectious Diseases, Neurology), By End-User (Pharmaceutical Companies, Biotechnology Companies, Medical Device Companies).
To learn more about this report, Download Free Sample Report
Israel Contract Research Organization Market Summary:
The Israel Contract Research Organization Market size is estimated at USD 67.3 Million in 2025 and is anticipated to reach USD 152.9 Million by 2033, growing at a CAGR of 10.80% from 2026 to 2033.
The life sciences services market in Israel will develop a dedicated support structure that focuses on three main areas which include outsourced research and clinical trials and regulatory consulting services. Pharmaceutical and biotech firms will increasingly depend on local expertise to navigate Israel’s strict data integrity standards and fast-moving approval pathways. Digital health tools will drive research demand toward trial models that use adaptable technology and flexible testing procedures. Advanced analytics and decentralized trial methods will reshape how studies are conducted, while regulators will tighten oversight on patient data usage. The service providers must modify their operations to meet scientific requirements which need precise execution and fast results and compliance with established standards.
Key Market Trends & Insights:
The contract research organization market in Israel will progress toward decentralized clinical trial operations which will rely on remote monitoring technology and wearable monitoring devices. The sponsors will choose hybrid testing methods which enable them to verify data accuracy while decreasing the number of patient appointments. The upcoming changes will enhance recruitment diversity and participant retention rates which will specifically benefit communities that have not been included in standard research methods used at research facilities throughout Israel.
Artificial intelligence will become more important for developing trial designs which help match patients and perform predictive analytics. Local providers will create data science teams to improve their operational processes while decreasing research project durations. The use of automated data management systems will decrease human mistakes which will lead to quicker business choices. The new technologies will change work processes and increase the requirement for particular technical knowledge.
The regulatory framework will impose stricter requirements about protecting patient information and maintaining cybersecurity while disclosing data. Israeli authorities will maintain their efforts to improve digital health regulations which will affect how organizations handle confidential medical data. The need for compliance will become a strategic advantage which will drive companies to implement secure systems and standardized reporting frameworks that fulfill both local and international legal obligations.
Customers will demand service packages that combine clinical research services with regulatory consulting and real-world evidence generation. Biotech startups will look for partners who can grow their operations together with their business. The need for these services will result in partnerships and mergers between companies which will develop specific expertise that enables organizations to provide customized solutions for challenging drug development projects.
Israel Contract Research Organization Market Segmentation
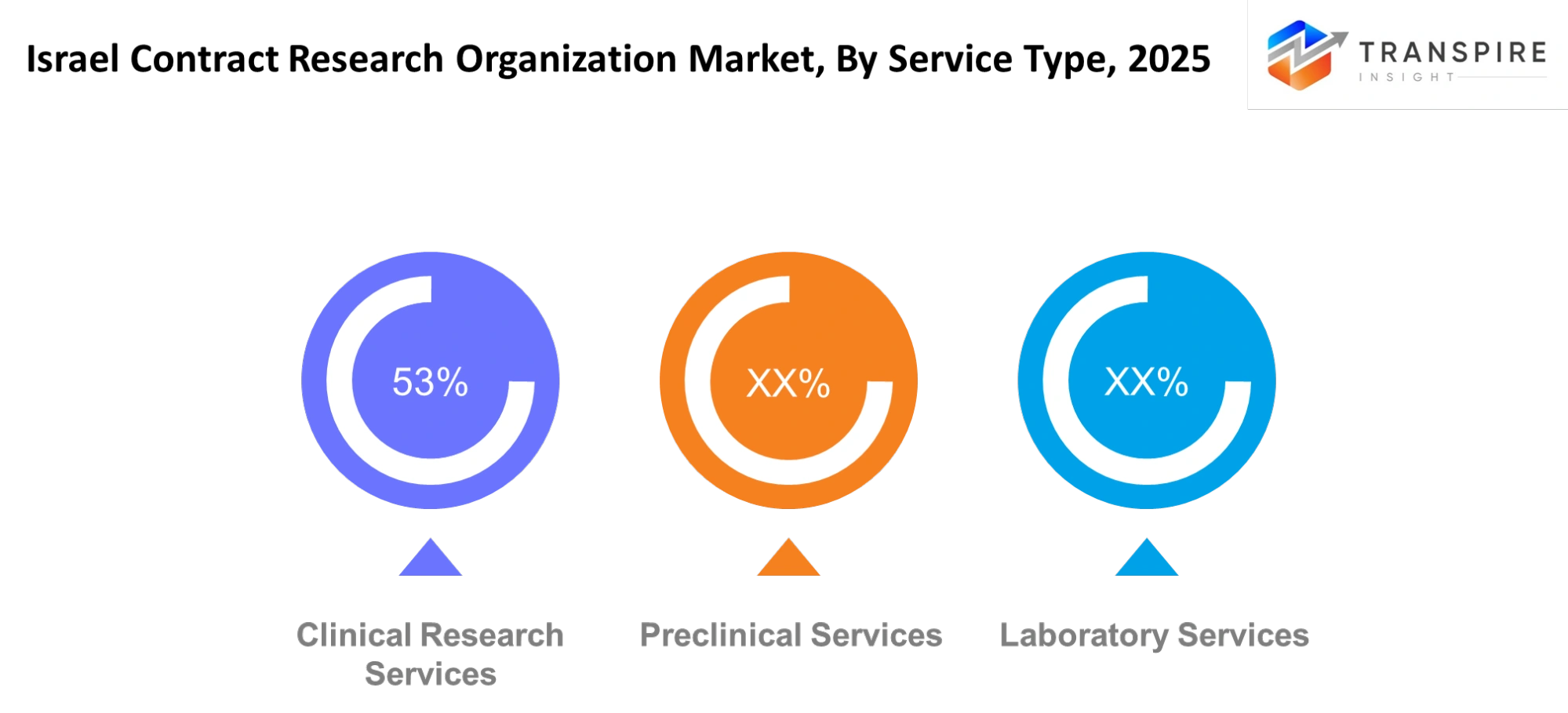
By Service Type
Clinical Research Services- The research services for clinical studies will serve as the main service which sponsors will use to delegate their trial management and site coordination and patient recruitment tasks. The adoption of decentralized and hybrid trial methods will become more common among study providers because these methods enable more people to take part and they achieve quicker study enrollment. The need for real-time data monitoring together with adaptive trial designs and effective regulatory documentation assistance will also increase.
Preclinical Services- The preclinical services will conduct testing at the early stage which includes testing toxic substances and studying how drugs move through the body and testing their effectiveness by using advanced laboratory models. Organizations will use computational biology and simulation tools to create predictions about future outcomes which will happen before they start human testing. The approach will help research teams to reduce development risks while they select research candidates for clinical trials in their competitive research programs.
Laboratory Services- The laboratory services will provide their support to clinical and preclinical work through their ability to analyze samples and test biomarkers and validate diagnostic tests. The growing use of precision medicine will create a need for both specialized assays and genomic profiling testing. The laboratories will implement automation systems together with digital tracking systems to achieve accurate results at fast speeds while meeting their strict quality requirements.
To learn more about this report, Download Free Sample Report
By Therapeutic Area
Oncology- The research field of oncology studies will experience growth because of increasing cancer patient numbers and advancements in targeted therapy research. The research organizations will conduct complicated trials that require testing immunotherapy treatments and customized therapy methods. The study design will use both advanced biomarker identification methods and genetic sequencing techniques which demand specialized skills for managing confidential data and individual patient treatment results.
Cardiovascular Diseases- Cardiovascular disease research will study patient results over extended periods while gathering data from actual medical settings. The medical staff will conduct research studies that investigate how to prevent diseases and how to use medical devices and different types of medications. The use of remote monitoring technologies will increase because they enable doctors to monitor patients continuously while remote monitoring improves the accuracy of data collected during extensive research periods.
Infectious Diseases- The study of infectious diseases will continue to be essential because of worldwide health problems and new disease-causing pathogens. Organizations will handle the research process for vaccine development and antiviral research and emergency response study programs. Organizations need to establish fast and flexible operational methods that create testing procedures that can adjust to sudden disease outbreaks and changing health emergency requirements.
Neurology- The field of neurology research will expand because scientists will start studying Alzheimer’s disease and Parkinson’s disease more intensely. The clinical studies will need lengthy monitoring times combined with sophisticated imaging methods. Organizations will spend money on digital tools for cognitive assessment development and systems that monitor patients to identify minor neurological changes which will help them evaluate brain function more effectively.
By End-User
Pharmaceutical Companies- In order to proceed with their complex development, pharmaceutical companies will rely on external research partners. The company will use outsourcing to decrease its workload while increasing project completion speed. The companies will search for partners who can provide complete testing and marketing research solutions which will maintain efficiency and compliance with regulations throughout the entire process.
Biotechnology Companies- Biotechnology companies will require specialized research organizations to develop new treatments because they lack research capabilities. The organization will choose to build flexible partnerships which will adapt to their project expansion. The organization will focus on scientific research partnerships together with fast testing methods and advanced technologies which enable them to make significant scientific breakthroughs and develop specialized medical treatments.
Medical Device Companies- Medical device companies need customized research solutions which will help them confirm their products safety and performance standards and user experience testing. Clinical trials will include both medical testing and equipment testing activities. Regulatory submissions will be supported by organizations which produce accurate data while modifying study protocols to meet new requirements from device advancements and user performance standards.
Country Insights
Israel will improve its research-oriented national status through its extensive network of educational institutions and medical facilities and innovation centers. The academic and industrial sectors will develop better research programs through their collaborative efforts. Global sponsors will come to this environment because they want to find high-quality data which they can obtain quickly from complicated clinical development programs.
The government will use tax breaks and funding initiatives to motivate international companies to invest in research services within the country. The regulatory authorities will conduct strict supervision while they work to shorten approval processes. The system will establish dependable research methods which protect patient safety and enable scientific progress throughout the nation.
The country’s diverse population will provide valuable opportunities for conducting varied clinical studies. The presence of advanced medical systems together with electronic healthcare records will enable better patient recruitment and data collection processes. Israel will become an attractive research site because its advantages will lead to better trial outcomes which sponsors need for precise research methods.
The company will use technological integration as its main element which includes digital health applications and artificial intelligence systems and remote patient monitoring. Local organizations will invest in these capabilities to improve trial efficiency. The organization will use its innovative capabilities to comply with worldwide research standards while maintaining its scientific and operational excellence.
Recent Development News
Expansion of Decentralized Trial Models- Current research shows that decentralized clinical trials and hybrid clinical trials will become the main clinical trial method because remote monitoring together with digital tools will decrease the need for physical research locations. The new system enables research organizations in Israel to conduct studies more efficiently while improving patient access to clinical trials and enhancing participant rates in their research work.
Integration of Artificial Intelligence in Research Operations- All clinical development activities will experience AI implementation but especially patient recruitment and data analysis work. Organizations will increasingly rely on predictive analytics to improve trial outcomes and reduce delays. The advancement will establish Israel as a leading research center that offers advanced technology and strong data analysis capabilities.
Rising Strategic Collaborations and Market Consolidation- The industry will experience more partnerships between companies which will result in companies acquiring other businesses to achieve better competitive positions. The larger companies will acquire smaller specialized organizations to create complete service packages. The market will undergo changes because the demand for efficient research solutions increases which will drive companies to innovate and develop new products.
|
Report Metrics |
Details |
|
Market size value in 2025 |
USD 67.3 Million |
|
Market size value in 2026 |
USD 74.57 Million |
|
Revenue forecast in 2033 |
USD 152.9 Million |
|
Growth rate |
CAGR of 10.80% from 2026 to 2033 |
|
Base year |
2025 |
|
Historical data |
2021 – 2024 |
|
Forecast period |
2026 – 2033 |
|
Report coverage |
Revenue forecast, competitive landscape, growth factors, and trends |
|
Country scope |
Israel |
|
Key company profiled |
IQVIA Holdings Inc., Labcorp Drug Development, ICON plc, Parexel International Corporation, Charles River Laboratories International Inc., Syneos Health Inc., PPD Inc. (Thermo Fisher Scientific), Medpace Holdings Inc., WuXi AppTec Co. Ltd., PRA Health Sciences Inc., SGS S.A., Covance Inc., Clinipace Inc., Worldwide Clinical Trials Inc., KCR S.A. |
|
Customization scope |
Free report customization (country, regional & segment scope). Avail customized purchase options to meet your exact research needs. |
|
Report Segmentation |
By Service Type (Clinical Research Services, Preclinical Services, Laboratory Services), By Therapeutic Area (Oncology, Cardiovascular Diseases, Infectious Diseases, Neurology), By End-User (Pharmaceutical Companies, Biotechnology Companies, Medical Device Companies). |
Key Israel Contract Research Organization Market Company Insights
The main businesses in the Israeli marine construction industry will concentrate their efforts on three specific fields which are specialized engineering and coastal infrastructure development and offshore project execution. The local companies will work together with international construction companies to complete complex energy and port development projects. The ability of a business to succeed in the market will depend on its technical skills and its ability to meet environmental regulations and its capacity to implement modern construction methods for effective project execution..
Company List
- IQVIA Holdings Inc.
- Labcorp Drug Development
- ICON plc
- Parexel International Corporation
- Charles River Laboratories International Inc.
- Syneos Health Inc.
- PPD Inc. (Thermo Fisher Scientific)
- Medpace Holdings Inc.
- WuXi AppTec Co. Ltd.
- PRA Health Sciences Inc.
- SGS S.A.
- Covance Inc.
- Clinipace Inc.
- Worldwide Clinical Trials Inc.
- KCR S.A
Israel Contract Research Organization Market Report Segmentation
By Service Type
- Clinical Research Services
- Preclinical Services
- Laboratory Services
By Therapeutic Area
- Oncology
- Cardiovascular Diseases
- Infectious Diseases
- Neurology
By End-User
- Pharmaceutical Companies
- Biotechnology Companies
- Medical Device Companies
Frequently Asked Questions
Find quick answers to common questions.
The approximate Israel Contract Research Organization Market size for the market will be USD 152.9 Million in 2033.
Key segments for the Israel Contract Research Organization Market By Service Type (Clinical Research Services, Preclinical Services, Laboratory Services), By Therapeutic Area (Oncology, Cardiovascular Diseases, Infectious Diseases, Neurology), By End-User (Pharmaceutical Companies, Biotechnology Companies, Medical Device Companies).
Major players in the Israel Contract Research Organization Market are IQVIA Holdings Inc., Labcorp Drug Development, ICON plc, Parexel International Corporation, Charles River Laboratories International Inc., Syneos Health Inc., PPD Inc. (Thermo Fisher Scientific), Medpace Holdings Inc., WuXi AppTec Co. Ltd., PRA Health Sciences Inc., SGS S.A., Covance Inc., Clinipace Inc., Worldwide Clinical Trials Inc., KCR S.A.
The Israel Contract Research Organization Market size is USD 67.3 Million in 2025.
The Israel Contract Research Organization Market CAGR is 10.80%.
- IQVIA Holdings Inc.
- Labcorp Drug Development
- ICON plc
- Parexel International Corporation
- Charles River Laboratories International Inc.
- Syneos Health Inc.
- PPD Inc. (Thermo Fisher Scientific)
- Medpace Holdings Inc.
- WuXi AppTec Co. Ltd.
- PRA Health Sciences Inc.
- SGS S.A.
- Covance Inc.
- Clinipace Inc.
- Worldwide Clinical Trials Inc.
- KCR S.A
Recently Published Reports
-
May 2026
Biosimilars Market
Biosimilars Market By Product Type (Monoclonal Antibodies, Recombinant Hormones, Erythropoietin, G-CSF, Others), By Indication (Oncology, Autoimmune Diseases, Blood Disorders, Diabetes, Others), By Manufacturing Type (In-House Manufacturing, Contract Manufacturing), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Gastrointestinal Endoscopy Market
Gastrointestinal Endoscopy Market By Type (Rigid Gastrointestinal Endoscope, Flexible Gastrointestinal Endoscopes, Disposable Gastrointestinal Endoscope), By Procedure Type (Colonoscopy, Gastroscopy, Duodenoscopy, Enteroscopy, Flexible Sigmoidoscopy, Others), By Application (Diagnosis, Treatment), By End-Users (Hospitals, Ambulatory Surgical Centers, Specialty Clinics, Laboratories, Others), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Apr 2026
Rare Inherited Metabolic Disorder Drug Market
Rare Inherited Metabolic Disorder Drug Market By Drug Class (Enzyme Replacement Drugs, Gene Therapy Drugs, Substrate Reduction Drugs, Small Module Drugs, Protein Drugs), By Route of Administration(Parenteral, Oral, Intrathecal), By Clinical Development (Marketed Drugs, Late Stage Clinical Phase III, Early Stage Clinical Phase I-II, Preclinical Candidates), By Indication (Lysosomal Storage Disorders, Urea Cycle Disorders, Amino Acid Metabolic Disorders, organic Acidemias, Peroximal Disorders), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033
-
Jan 2026
Pharmaceutical Cleanroom Technology Market
Pharmaceutical Cleanroom Technology Market By Product (Equipment, Consumables, Services); By Cleanroom Type (Standard Cleanrooms, Modular Cleanrooms); By End Use (Pharmaceutical Companies, Biotechnology Companies), By Industry Analysis, Size, Share, Growth, Trends, and Forecasts 2021-2033